| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 100mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|
| 靶点 |
Trehalase, tyrosinase. Validamycin A inhibits trehalase from Rhizoctonia solani with an IC₅₀ of 7.2 × 10⁻⁵ M (72 μM), and its aglycone validoxylamine A inhibits trehalase competitively with a Kᵢ of 1.9 × 10⁻⁹ M (1.9 nM). It inhibits mushroom tyrosinase with an IC₅₀ of 19.23 ± 0.26 mM, and a mixed-type inhibition constant Kᵢ of 5.893 ± 0.038 mM. [1,2,3]
Antibiotic[1], IC50: 72 μM (trehalase)[2], Ki: 5.893 mM (tyrosinase)[3] |
|---|---|
| 体外研究 (In Vitro) |
对立枯丝核菌海藻糖酶的抑制:Validamycin A对立枯丝核菌海藻糖酶表现出强效抑制活性,IC₅₀为7.2 × 10⁻⁵ M。其苷元validoxylamine A的抑制活性更强,Kᵢ为1.9 × 10⁻⁹ M。Validamycin A对来自立枯丝核菌的纤维素酶、果胶酶、几丁质酶、α-淀粉酶、α-葡萄糖苷酶和β-葡萄糖苷酶均无显著抑制作用。[2]
对酪氨酸酶的抑制:Validamycin A以剂量依赖性、可逆的方式抑制蘑菇酪氨酸酶,IC₅₀为19.23 ± 0.26 mM。抑制类型为混合型抑制,Kᵢ为5.893 ± 0.038 mM,α值为2.096 ± 0.546。失活动力学符合一级反应动力学,分为快相和慢相两个阶段,快相速率常数范围为1.54 × 10⁻³至3.60 × 10⁻³ s⁻¹,慢相速率常数范围为0.45 × 10⁻³至1.45 × 10⁻³ s⁻¹。[3] 对真菌生长和形态的影响:在“树枝状试验法”中,Validamycin A在低至0.01 μg/mL的浓度下即可引起立枯丝核菌菌丝异常分支。在富营养培养基上不显著抑制生长,但在水琼脂上特异性诱导菌丝异常分支并停止菌落发育。[2] 对黄曲霉的影响:Validamycin A在1 μg/mL浓度下显著增加黄曲霉分生孢子中海藻糖水平,延迟分生孢子萌发(特别是在10和12小时),并降低真菌黏附性。Validamycin A对黄曲霉ATCC204304的最低抑菌浓度(MIC)为1 μg/mL。[1] 与两性霉素B的联合作用:在棋盘格实验中,validamycin A与两性霉素B对黄曲霉ATCC204304表现出相加效应(FICI = 0.625)。在两性霉素B耐药(MIC > 4 μg/mL)的黄曲霉临床分离株中,两者联合表现出协同效应,FICI值为0.25–0.28,对应的validamycin A和两性霉素B浓度分别为0.125 μg/mL和2 μg/mL。[1] 对立枯丝核菌海藻糖代谢的影响:Validamycin A在0.1 μg/mL浓度下显著抑制立枯丝核菌菌丝内海藻糖的降解,表明其在体内抑制海藻糖酶活性。[2] 立枯丝核菌细胞摄取与代谢:Validamycin A可被立枯丝核菌菌丝摄取,并在细胞内被β-葡萄糖苷酶水解为具有更高抑制活性的validoxylamine A。Validamycin A(β-D-葡萄糖苷)比其苷元(validoxylamine A)或α-D-葡萄糖苷(validamycin D)更容易被摄取。[2] 井冈霉素 A(0.5–1 μg/mL;18 小时)可阻止 A 生长。分生孢子萌发并抑制黄霉菌[1]。对于人支气管上皮细胞,井冈霉素A没有细胞毒性[1]。 HIS85、HIS244、GLU256、HIS259 和 ASN260 属于有效霉素 A 直接结合的酪氨酸酶活性位点中的残基 [1]。 |
| 酶活实验 |
海藻糖酶抑制实验(立枯丝核菌):通过比色法测定从海藻糖释放的D-葡萄糖来检测海藻糖酶活性。反应混合物包含125 μL酶溶液、50 μL 0.4 M海藻糖、200 μL抑制剂溶液或水,以及125 μL 0.2 M磷酸盐缓冲液(pH 6.0)。在37°C孵育15分钟后,使用葡萄糖氧化酶法测定释放的葡萄糖。[2]
酪氨酸酶抑制实验:通过监测L-DOPA氧化为多巴色素时475 nm处吸光度的增加,用分光光度法测定酪氨酸酶活性。测定混合物包含2 mM L-DOPA、1.0 μg/mL酪氨酸酶和不同浓度的validamycin A,溶于50 mM磷酸钠缓冲液(pH 7.0),在25°C下进行。[3] 抑制动力学分析:使用Lineweaver-Burk双倒数图确定抑制类型。绘制斜率与y截距相对于抑制剂浓度的二级图,通过线性拟合计算混合型抑制的Kᵢ和α值。[3] 失活动力学:通过将酶与不同浓度的validamycin A(3.125–50 mM)共孵育进行酪氨酸酶抑制的时间进程研究。在不同时间间隔收集等分试样,测量残留活性。半对数图显示双相一级失活动力学,从斜率确定速率常数。[3] 荧光光谱分析:在280 nm激发后测量酪氨酸酶的内源色氨酸荧光(发射波长300–400 nm),以监测validamycin A结合引起的三级结构变化。使用ANS结合荧光(激发390 nm,发射400–600 nm)评估表面疏水性的变化。从荧光猝灭数据计算结合常数(K = 0.06 ± 0.012 mM⁻¹)和结合位点数(n = 1.06 ± 0.37)。[3] 计算对接与分子动力学模拟:使用AutoDock Vina进行对接,CHARMM结合CGENFF参数进行分子动力学模拟,研究validamycin A与酪氨酸酶的结合模式。选择最低能量簇、最大簇和活性位点附近的簇进行10 ns模拟。鉴定出五个残基(HIS85、HIS244、GLU256、HIS259和ASN260)与validamycin A相互作用,距离小于4 Å。[3] |
| 细胞实验 |
黄曲霉活力测定(XTT):将黄曲霉分生孢子(10³个)接种于含或不含validamycin A的不同培养基的96孔板中,在37°C孵育18小时。加入XTT溶液(0.5 mg/mL溶于PBS),孵育15分钟,离心后测定上清液在490 nm的吸光度,评估真菌活力。[1]
真菌黏附性测定(结晶紫):将黄曲霉分生孢子(10⁵个/mL)接种于含或不含validamycin A(1 μg/mL)的沙氏葡萄糖肉汤的96孔板中,在37°C孵育24小时。轻轻洗涤后,用0.1%结晶紫染色,乙醇脱色,测定600 nm吸光度。[1] 黄曲霉分生孢子海藻糖测定:在含或不含validamycin A(1 μg/mL)的SDA上培养5天后收集黄曲霉分生孢子(2 × 10⁸个)。煮沸分生孢子,离心,使用葡萄糖氧化酶法测定上清液中的海藻糖含量。[1] 萌发实验:将黄曲霉分生孢子(1 × 10⁸个)接种于含或不含validamycin A(1 μg/mL)的沙氏葡萄糖肉汤中,在37°C下振荡培养(200 rpm)。在不同时间点,显微镜下计数100个分生孢子,计算萌发百分比。[1] 细胞毒性实验(LDH):将BEAS-2B人支气管上皮细胞(1 × 10⁴个)与不同浓度的validamycin A(1 μg/mL至1 mg/mL)共孵育24小时。在450 nm处测定LDH释放,计算相对于高对照和低对照的细胞毒性百分比。在高达1 mg/mL的浓度下未观察到显著细胞毒性。[1] 细胞增殖测定[1] 细胞类型: 黄曲霉 ATCC204304 测试浓度: 0.5 μg/mL、1 μg/mL 孵化时间:18小时 实验结果:抑制黄曲霉的生长。 |
| 药代性质 (ADME/PK) |
代谢/代谢产物
在动物体内,裂解为葡萄糖和胺残基。 生物半衰期 小于或等于5小时 |
| 毒性/毒理 (Toxicokinetics/TK) |
毒性数据
LC50(大鼠)> 5,000 mg/m3 非人类毒性值 LD50 大鼠口服 > 20,000 mg/kg LD50 小鼠口服 > 20,000 mg/kg LD50 大鼠经皮 > 5000 mg/kg |
| 参考文献 | |
| 其他信息 |
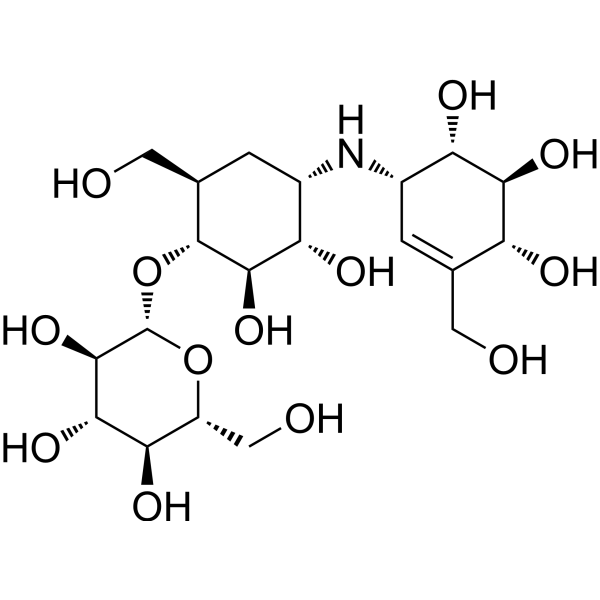
Validamycin A 属于 validamycin 类化合物,其结构为 (1R,2S,3S,4S,6R)-4-氨基-6-(羟甲基)环己烷-1,2,3-三醇,其中 1 位羟基被转化为 β-D-葡萄糖苷,且与氮原子相连的一个氢原子被 (1R,4R,5R,6S)-4,5,6-三羟基-3-(羟甲基)环己-2-烯-1-基取代。它是吸水链霉菌 (Streptomyces hygroscopicus) 产生的主要 validamycin。它是一种EC 2.4.1.231 [α,α-海藻糖磷酸化酶(构型保持型)]抑制剂、EC 2.4.1.64(α,α-海藻糖磷酸化酶)抑制剂、EC 3.2.1.28(α,α-海藻糖酶)抑制剂和抗真菌农药。它属于有效霉素类化合物,是一种仲氨基化合物、多元醇和抗生素杀菌剂。它是有效霉素A(1+)的共轭碱。
据报道,有效霉素A存在于花青链霉菌(Streptomyces anthocyanicus)、吸湿链霉菌(Streptomyces hygroscopicus)和利迪链霉菌(Streptomyces lividans)中,并有相关数据。 |
| 分子式 |
C20H35NO13
|
|---|---|
| 分子量 |
497.49
|
| 精确质量 |
497.21
|
| 元素分析 |
C, 48.29; H, 7.09; N, 2.82; O, 41.81
|
| CAS号 |
37248-47-8
|
| PubChem CID |
443629
|
| 外观&性状 |
White to off-white solid powder
|
| 密度 |
1.7±0.1 g/cm3
|
| 沸点 |
813.7±65.0 °C at 760 mmHg
|
| 熔点 |
130-135ºC
|
| 闪点 |
445.9±34.3 °C
|
| 蒸汽压 |
0.0±6.6 mmHg at 25°C
|
| 折射率 |
1.689
|
| LogP |
-5.43
|
| tPSA |
253.02
|
| 氢键供体(HBD)数目 |
12
|
| 氢键受体(HBA)数目 |
14
|
| 可旋转键数目(RBC) |
7
|
| 重原子数目 |
34
|
| 分子复杂度/Complexity |
697
|
| 定义原子立体中心数目 |
14
|
| SMILES |
C1[C@@H]([C@H]([C@@H]([C@H]([C@H]1N[C@H]2C=C([C@H]([C@@H]([C@H]2O)O)O)CO)O)O)O[C@H]3[C@@H]([C@H]([C@@H]([C@H](O3)CO)O)O)O)CO
|
| InChi Key |
JARYYMUOCXVXNK-CSLFJTBJSA-N
|
| InChi Code |
InChI=1S/C20H35NO13/c22-3-6-1-8(12(26)15(29)11(6)25)21-9-2-7(4-23)19(17(31)13(9)27)34-20-18(32)16(30)14(28)10(5-24)33-20/h1,7-32H,2-5H2/t7-,8+,9+,10-,11-,12+,13+,14-,15+,16+,17-,18-,19-,20+/m1/s1
|
| 化学名 |
(2R,3R,4S,5S,6R)-2-[(1R,2R,3S,4S,6R)-2,3-dihydroxy-6-(hydroxymethyl)-4-[[(1S,4R,5S,6S)-4,5,6-trihydroxy-3-(hydroxymethyl)cyclohex-2-en-1-yl]amino]cyclohexyl]oxy-6-(hydroxymethyl)oxane-3,4,5-triol
|
| 别名 |
Validamycin; jinggangmycin; VALIDAMYCIN A; 37248-47-8; Valimon;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: (1). 本产品在运输和储存过程中需避光。 (2). 请将本产品存放在密封且受保护的环境中(例如氮气保护),避免吸湿/受潮。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
H2O : 125 mg/mL (251.26 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 100 mg/mL (201.01 mM) in PBS (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶。
请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0101 mL | 10.0505 mL | 20.1009 mL | |
| 5 mM | 0.4020 mL | 2.0101 mL | 4.0202 mL | |
| 10 mM | 0.2010 mL | 1.0050 mL | 2.0101 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。