| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
STING/stimulator of interferon genes
Stimulator of interferon genes (STING, also known as TMEM173) (IC50 for human STING activation inhibition: 0.8 μM; Ki for human STING binding: 0.5 μM) [1] - Stimulator of interferon genes (STING, TMEM173) [2] |
|---|---|
| 体外研究 (In Vitro) |
C-176 显着抑制 STING 介导的 IFNβ 报告基因活性,但不抑制 RIG-I 或 TBK1 介导的 IFNβ 报告基因活性。 C-176 预处理可显着抑制 CMA 介导的 I 型 IFN 和 IL-6 血清水平诱导[1]。
1. 强效抑制STING激活:C-176以剂量依赖性方式抑制环GMP-AMP(cGAMP)诱导的人THP-1单核细胞和稳定表达人STING的HEK293T细胞中STING激活,IC50为0.8 μM;它阻断STING介导的TBK1和IRF3磷酸化(western blot检测),在5 μM浓度下分别减少I型干扰素(IFN-β)和促炎细胞因子(TNF-α、IL-6)的分泌约85%、78%和72%[1] 2. 共价结合STING:质谱分析和X射线晶体学证实,C-176共价结合STING蛋白中的半胱氨酸残基(Cys148);这种结合诱导STING构象变化,阻止其寡聚化和下游信号激活[1] 3. 高STING选择性:C-176在高达20 μM的浓度下,对其他先天免疫信号通路(如TLR4、RIG-I、MDA5)无显著抑制作用;不与测试的其他含半胱氨酸蛋白(如GAPDH、肌动蛋白)结合,证实其STING特异性共价相互作用[1] 4. 抑制STING介导的NLRP3焦亡:在cGAMP刺激的小鼠原代小胶质细胞中,C-176(1-10 μM)剂量依赖性降低NLRP3炎症小体激活,表现为caspase-1(p20亚基)和gasdermin D(GSDMD)切割减少,IL-1β释放量在10 μM时减少约65%[2] |
| 体内研究 (In Vivo) |
C-176(每只小鼠 750/375 nmol C-176,溶于 200 μL 玉米油)不会引起相当大的毒性,可大大降低 CMA 介导的 I 型 IFN 和 IL-6 血液水平的激活[1]。 Trex1−/− 小鼠中没有明显的明显毒性症状,C-176 显着降低 I 型 IFN 的血清水平,并强烈抑制心脏中的炎症标志物[1]。在 Trex1−/− 小鼠中,C-176 显着降低了许多全身炎症指标 [
1. 减轻cGAMP诱导的小鼠全身性炎症:C57BL/6小鼠腹腔注射cGAMP(5 mg/kg)诱导STING依赖性炎症,C-176预处理(10 mg/kg,腹腔注射,cGAMP给药前1小时)较溶媒对照组显著降低血清IFN-β(约70%)、TNF-α(约62%)和IL-6(约58%)水平;肝和脾组织学分析显示炎症细胞浸润减少[1] 2. 改善创伤性脑损伤(TBI)小鼠的神经炎症和焦亡:遭受TBI的雄性C57BL/6小鼠,给予C-176(5 mg/kg,腹腔注射,TBI后1小时开始,每日1次,持续3天);C-176使大脑皮层中STING激活(p-STING/p-TBK1/p-IRF3水平)降低约55%,抑制NLRP3焦亡(caspase-1切割和GSDMD-N水平降低),脑匀浆中IL-1β和TNF-α水平分别减少约52%和48%;同时改善神经功能评分,减少脑水肿体积约35%[2] |
| 酶活实验 |
Competition assay/竞争分析[1]
将表达Flag-STING的HEK293T细胞与指定化合物一起孵育,1小时后加入C-176-AL 1小时。将细胞收集在PBS中,通过C-176-AL-介导的STING标记的凝胶内分析进行分析(见“基于凝胶的化合物与STING结合的分析”)。 基于凝胶的化合物与STING结合分析[1] 表达Flag-STING的HEK293T细胞在无血清培养基中与C-176-AL、C-175-AZ、叠氮碘乙酰胺或H-151-AL一起孵育,收集在PBS中,通过反复冷冻和解冻裂解。用新制备的“点击试剂”混合物处理43微升裂解细胞,该混合物含有三(苄基三唑甲基)胺(TBTA)(每个样品3μl,3 mM在1:4 DMSO:t-ButOH中)、四甲基罗丹明(TAMRA)叠氮化物、SiR叠氮化物或SiR炔(每个样品2μl,1.25 mM在DMSO中),以及新制得的CuSO4(每个样品1μl)和三-(2-羧乙基)盐酸膦(TCEP)(每个样本1μl,在室温下孵育30分钟。通过加入还原性样品缓冲液淬灭反应。使用Fusion-FX可视化凝胶内荧光,并使用Fusion-capt高级采集软件进行分析。 与亚琥珀酸二琥珀酰亚胺酯交联[1] 表达Flag-mmSTING的HEK293T细胞在有或没有C-176(1μM)的情况下孵育1小时,并用DMSO或CMA(250μg ml-1)处理2小时。在室温下,在PBS中用DMSO中新鲜制备的1 mM二琥珀酰亚胺基琥珀酸盐(DSS)进行交联1小时。 [3H]-棕榈酸酯代谢标记[1] 指示标志-STING构建体在HEK293T细胞中表达。对于代谢标记,细胞在用10mM Hepes缓冲的格拉斯哥最低必需培养基中饥饿1小时,pH 7.4,用C-178或C-176(1μM)缓冲。然后在C-178或C-176存在下,在200μCi ml−1[3H]-棕榈酸(9,10-3H(N)) 的IM中孵育细胞2小时,并用CMA(250μg ml−1)刺激或不刺激。对于免疫沉淀,细胞在PBS中洗涤三次,在4°C下在以下缓冲液(0.5%Nonidet P-40、500 mM Tris pH 7.4、20 mM EDTA、10 mM NaF、2 mM苯甲脒和蛋白酶抑制剂混合物)中裂解30分钟,并在5000 rpm下离心3分钟。上清液在4°℃下与适当的抗体(抗Flag、抗转铁蛋白受体和抗钙蛋白酶)和G琼脂糖珠一起孵育过夜。对于放射性标记实验,在免疫沉淀后,在4-20%梯度SDS-PAGE之前,将洗涤过的珠子在90°C的还原样品缓冲液中孵育5分钟。SDS-PAGE后,将凝胶在固定剂溶液(25%异丙醇、65%H2O、10%乙酸)中孵育,并与信号增强剂Amplify NAMP100一起孵育30分钟。使用放射自显影显示放射性标记产物,并使用台风成像仪进行定量。 试试AI翻译 笔记 1. STING激活抑制实验(HTRF基于): - 细胞制备:稳定表达人STING和IFN-β荧光素酶报告基因的HEK293T细胞以2×10⁴个细胞/孔接种到96孔板,孵育过夜[1] - 药物与激动剂处理:向细胞中加入系列稀释的C-176(0.01 μM-20 μM),随后用cGAMP(1 μM)刺激16小时[1] - 荧光素酶检测:裂解细胞,用发光仪测量荧光素酶活性,相对于溶媒对照组计算STING介导的荧光素酶活性抑制百分比,通过剂量-反应曲线推导IC50值[1] 2. SPR-based STING结合实验: - 传感器芯片制备:通过胺偶联法将重组人STING(1-341位残基)固定在CM5传感器芯片上[1] - 结合反应:将系列稀释的C-176(0.05 μM-10 μM)以恒定流速注入STING包被的芯片(运行缓冲液中);选择性对照实验中,芯片固定TLR4或RIG-I并重复上述流程[1] - 数据分析:记录传感图,采用共价结合模型计算解离常数(Ki);未检测到与TLR4或RIG-I的显著结合[1] 3. 质谱法验证共价结合: - 反应设置:重组人STING蛋白(10 μM)与C-176(20 μM)在结合缓冲液中37°C孵育2小时;对照反应不含C-176[1] - 样本制备:反应混合物经胰蛋白酶消化,肽段通过反相色谱纯化[1] - 质谱分析:通过LC-MS/MS分析肽段片段以鉴定修饰残基,在含Cys148的肽段中检测到与C-176结合对应的质量偏移[1] |
| 细胞实验 |
免疫沉淀[1]
多西环素在HEK293T细胞中诱导Flag-STING表达过夜。细胞与或不与C-178或C-176(1μM)一起孵育1小时,并用DMSO或CMA(250μg ml−1)处理2小时。细胞在PBS中洗涤,在裂解缓冲液(50 mM HEPES、150 mM NaCl、10%甘油、1 mM MgCl、1 mM CaCl、1%Brij-58和蛋白酶抑制剂混合物)中裂解30分钟。在4°C下使用抗Flag M2亲和凝胶琼脂糖凝胶对Flag-STING进行免疫沉淀2小时。在裂解缓冲液和PBS中严格洗涤后,完全去除上清液,在进行SDS-PAGE之前,将树脂在样品缓冲液中煮沸。为了免疫沉淀内源性STING,将脾细胞在上述裂解缓冲液中裂解,并与抗STING(RD System AF6516)和G琼脂糖珠一起孵育过夜。在PBS中洗涤珠子,并进行基于凝胶的C-176-AL与STING结合分析。 mmSTING的完整质量测量[1] 多西环素在HEK293T细胞中诱导Flag-mmSTING或Flag-mmS廷G(C91S)表达过夜。细胞用或不用C-178或C-176(1μM)处理30分钟,并在裂解缓冲液(20 mM Hepes、150 mM NaCl、10%甘油和1%DDM)中裂解。Flag-STING在4°C下用抗Flag M2亲和凝胶琼脂糖凝胶免疫沉淀2小时。根据制造商的说明,使用Flag肽洗脱沉淀的蛋白质。在岛津MS2020上进行蛋白质质谱分析,该MS2020连接到Nexerra UHPLC系统,配备Waters ACQUITY UPLC BEH C4 1.7-μm,2.1×50mm柱。缓冲液A为0.05%甲酸水溶液,缓冲液B为0.05%甲酸乙腈溶液。在6.0分钟内,缓冲液B的分析梯度为10%至90%,流速为0.75 ml min−1。从300-2000 Da收集质谱,并使用MagTran软件对光谱进行解卷积。 |
| 动物实验 |
动物/疾病模型:野生型小鼠。
剂量:每只小鼠750/375 nmol C-176,溶于200 μL玉米油中(约1.34/0.67 mg/mL)。 给药途径:腹腔注射,单次。 实验结果:血清I型干扰素和IL-6水平显著降低。 小鼠和体内研究[1] C57BL/6J小鼠(品系编号000664)购自Jackson Laboratories。TREX1缺陷小鼠由T. Lindahl31惠赠,并已回交至C57BL/6NJ超过10代。小鼠饲养于瑞士洛桑联邦理工学院(EPFL)的特定病原体清除(SPF)条件下。在药代动力学研究中,每只野生型小鼠腹腔注射750 nmol C-176(溶于200 μl玉米油中)。分别于注射后30分钟、2小时和4小时采集血液样本,并通过液相色谱-高分辨率质谱法(LC-HRMS)测定血清中C-176的浓度。为评估C-176的体内抑制作用,将8-12周龄的野生型小鼠分别注射溶剂或C-176。1小时或4小时后,给予浓度为224 mg/kg的CMA。4小时后,处死小鼠并采集血清,以测定CMA诱导的细胞因子水平。为了评估 H-151 的体内抑制作用,将 750 nmol H-151(溶于 200 μl 含 10% Tween-80 的 PBS 溶液中)腹腔注射到野生型小鼠体内。1 小时后,给予 CMA(112 mg kg−1),4 小时后处死小鼠并收集血清。Trex1−/− 小鼠的疗效研究如下:将 7.5 μl C-176 或 DMSO(溶于 85 μl 玉米油中)注射到 2-5 周龄的小鼠体内,连续 11 天,每天两次。小鼠在 CO2 麻醉箱中麻醉后,通过颈椎脱臼处死。毒理学研究中,将 562.5 nmol C-176(溶于 8 周龄小鼠体内)每日注射到 8 周龄小鼠体内,持续 2 周。本研究分两个阶段进行。实验共使用了198只雄性大鼠(体重300-350克,9-11周龄)(第一阶段90只,第二阶段108只)(如图1所示)。实验过程中(造模后24小时内)共发现7只大鼠死亡,另有6只大鼠因行为学异常被排除(造模后30天)。所有动物均接受健康状况监测,并采用颈椎脱臼后断头处死。[2] 第一阶段[2] 为了研究STING的作用,在第一阶段,通过腹腔注射选择性STING拮抗剂C-176或STING激动剂ADU-S100,以研究TBI后STING信号通路的变化。大鼠被随机分为四组(每组 n = 18 只):假手术 + 载体1 组、TBI + 载体1 组、TBI + ADU-S100 组和 TBI + C-176 组。大鼠的随机分组采用基于网络的随机分组生成器(www.pubmed.de/tools/zufallsgenerator)。数据采集和分析过程对实验人员不知情。载体1(玉米油,货号 ST1177;碧云天)用于溶解 ADU-S100 和 C-176。载体1(玉米油)在 TBI 或假手术模型建立后相同时间点通过腹腔注射给药。注射后 24 小时收集脑组织进行蛋白质印迹分析(每组 n = 6 只)。在采用重物坠落模型诱导TBI后30天,对大鼠进行神经行为学测试,包括旷场测试、强迫游泳测试和新物体识别测试(每组n=12只大鼠)。此外,还收集了脑组织进行尼氏染色和免疫荧光染色(每组n=6只大鼠)以及ELISA检测(每组n=6只大鼠)(图1a)。 第二阶段[2] 在第二阶段,通过脑室内注射给予NLRP3选择性激活剂(尼日利亚菌素),以阐明NLRP3作为TBI下游因子的作用。将大鼠分为五组(每组 n = 18 只):TBI + ADU-S100 + MCC950 组、TBI + ADU-S100 + VX765 组、TBI + C-176 + 尼日利亚菌素组、TBI + C-176 + 载体2 组和 TBI + ADU-S100 + 载体2 组。载体2用于溶解尼日利亚菌素、MCC950 和 VX-765,并通过脑室内注射给药。在 TBI 后 24 小时收集组织进行蛋白质印迹分析(每组 n = 6 只)。之后,在造模后30天进行行为学测试(每组12只大鼠)、尼氏染色和免疫荧光染色(每组6只大鼠)以及ELISA检测(每组6只大鼠)(图1b)。其余大鼠通过颈椎脱臼处死。 给药在造模前30分钟,使用连接微量注射器(5 μl)的针头进行脑室内注射,分别给予尼日利亚菌素、MCC950或VX765。注射点坐标为:前囟后方0.3 mm,侧方1.0 mm,腹侧2.5 mm。使用微量输注泵(TJ-4A/SL0107-1A,LongerPump),以 0.667 μl/min 的速率将尼日利亚菌素(2 μg/2 μl)、MCC950(2 μg/2 μl)或 VX765(2 μg/2 μl)注入脑组织。尼日利亚菌素、MCC950 或 VX765 溶解于 10% 乙醇和 90% 玉米油的混合溶液中。针头在脑内停留 10 分钟后缓慢拔出。最后,用骨蜡封闭钻孔。在脑外伤后立即腹腔注射 STING 激动剂 ADU-S100(0.5 mg/kg)和拮抗剂 C-176(10 mg/kg)。本研究中使用的药物剂量是根据先前发表的研究(Lee et al., 2021; Peng et al., 2020; Tsuchiya et al., 2019)在预实验中确定的。 1. cGAMP诱导的全身炎症小鼠模型: - 动物准备:6-8周龄雄性C57BL/6小鼠适应环境1周[1] - 分组和给药:小鼠随机分为载体对照组、cGAMP单药组和C-176+cGAMP组(每组n=6)。C-176溶解于DMSO和玉米油(1:9 v/v)的混合溶液中。治疗组在腹腔注射cGAMP(5 mg/kg,腹腔注射)前1小时腹腔注射10 mg/kg C-176。对照组接受等体积的载体或单独的cGAMP注射[1] - 样本采集:小鼠在注射cGAMP 6小时后处死。采集血清用于细胞因子检测(ELISA)。取肝脏和脾脏组织,用福尔马林固定,石蜡包埋,用于组织学分析[1] 2. 创伤性脑损伤(TBI)小鼠模型: - 动物准备:8-10周龄雄性C57BL/6小鼠适应环境1周[2] - TBI诱导:小鼠麻醉后进行可控皮层冲击(CCI)以诱导TBI[2] - 分组和给药:小鼠随机分为假手术组、TBI+载体组和TBI+C-176组(每组n=8)。将C-176溶解于DMSO/玉米油(1:9 v/v)混合溶液中。治疗组在TBI后1小时开始,每天腹腔注射5 mg/kg C-176,连续3天。假手术组和TBI+载体组注射等体积的载体[2]。 - 结果检测:在TBI后24小时、48小时和72小时,采用改良神经功能严重程度评分(mNSS)评估神经功能。小鼠在TBI后72小时处死,取出脑组织测量脑水肿体积。将脑皮层组织匀浆用于细胞因子检测(ELISA)和蛋白质印迹分析。制备脑组织切片用于p-STING和Iba1(小胶质细胞标志物)的免疫组织化学染色[2]。 |
| 毒性/毒理 (Toxicokinetics/TK) |
1. 体外毒性:C-176在浓度高达20 μM(体外实验中使用的最高浓度)时,对THP-1巨噬细胞、HEK293T细胞或原代小胶质细胞均未显示出显著的细胞毒性,细胞存活率与对照组相比>90% [1, 2]
2. 体内毒性:小鼠单次腹腔注射C-176(10 mg/kg)或连续3天腹腔注射(5 mg/kg/天)后,未观察到体重、食物摄入量或器官重量(肝脏、肾脏、脑)的显著变化。未检测到急性毒性症状(例如嗜睡、行为异常、出血)或主要器官的组织学异常 [1, 2] |
| 参考文献 | |
| 其他信息 |
先天免疫通路异常激活与多种疾病相关。对先天免疫通路分子机制的深入理解为靶向治疗方法带来了希望,但开发能够特异性作用于目标分子的药物仍然面临挑战。本文报道了高效且选择性的小分子干扰素基因刺激因子(STING)拮抗剂的发现和表征。STING是细胞内DNA感应通路的核心信号组分1,2。从机制上讲,这些化合物共价靶向预测的跨膜半胱氨酸残基91,从而阻断STING的激活诱导棕榈酰化。利用这些抑制剂,我们发现STING的棕榈酰化对于其在Golgi体组装成多聚体复合物至关重要,进而影响下游信号因子的募集。这些化合物及其衍生物能够降低人和小鼠细胞中STING介导的炎症细胞因子的产生。此外,我们发现这些小分子拮抗剂能够减轻小鼠自身炎症性疾病的病理特征。总之,我们的研究揭示了STING可被药理学抑制的机制,并证明了靶向STING的疗法在治疗自身炎症性疾病方面的潜力。[1]
严重创伤性脑损伤(TBI)后的长期神经功能缺损,包括认知功能障碍和情绪障碍,会显著影响康复。炎症反应诱导的胶质细胞活化与TBI后的神经功能缺损有关。本研究旨在探讨干扰素基因刺激因子(STING)-核苷酸结合寡聚化结构域样受体含吡啶结构域3(NLRP3)信号通路在严重TBI啮齿动物模型中的作用。采用坠落加失血再输注模型建立严重TBI模型。造模后,单次给予选择性STING激动剂ADU-S100或拮抗剂C-176。此外,在建立脑损伤模型前30分钟,分别脑室内注射NLRP3抑制剂MCC950、激活剂尼日利亚菌素或caspase-1抑制剂VX765。之后,采用新物体识别测试、旷场测试、强迫游泳测试、蛋白质印迹法和免疫荧光法评估重度TBI的行为和病理变化。C-176的给药减轻了TBI引起的认知功能障碍和情绪障碍、神经元丢失以及胶质细胞的炎症激活。然而,STING激动剂ADU-S100的给药加剧了TBI引起的行为和病理变化。此外,STING激活通过促进胶质细胞活化加剧了细胞焦亡相关的神经炎症,表现为裂解型caspase-1和GSDMD N端表达增加。相反,C-176的给药则表现出抗细胞焦亡作用。 C-176 的神经保护作用可被 NLRP3 激活剂尼日利亚菌素部分逆转。总的来说,神经胶质细胞 STING 是 TBI 后神经炎症的主要因素。然而,STING 的药理学抑制可显著改善神经炎症,部分原因在于抑制了 NLRP3 信号通路。STING-NLRP3 信号通路是 TBI 诱导的神经功能障碍的潜在治疗靶点。[2] 1. C-176 是一种首创的共价小分子 STING 抑制剂,靶向先天免疫信号通路[1] 2. 其核心作用机制是与 STING 的 Cys148 共价结合,从而阻断 STING 寡聚化——这是下游 TBK1-IRF3 激活和细胞因子产生的关键步骤。这使其区别于非共价 STING 抑制剂[1] 3. C-176对STING具有高度选择性,优于其他固有免疫受体和含半胱氨酸蛋白,从而最大限度地减少了脱靶效应[1] 4. 该化合物通过抑制STING-NLRP3细胞焦亡轴,在STING介导的炎症性疾病(包括全身炎症和与创伤性脑损伤相关的神经炎症)中显示出治疗潜力[2] 5. STING是固有免疫的核心介质,由受损DNA释放的环状二核苷酸(例如cGAMP)激活,其过度激活会导致多种炎症和自身免疫性疾病[1, 2] |
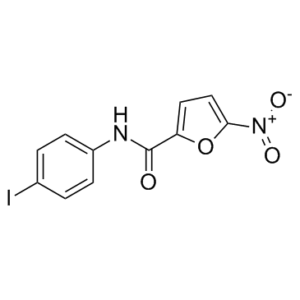
| 分子式 |
C11H7IN2O4
|
|
|---|---|---|
| 分子量 |
358.09
|
|
| 精确质量 |
357.945
|
|
| 元素分析 |
C, 36.90; H, 1.97; I, 35.44; N, 7.82; O, 17.87
|
|
| CAS号 |
314054-00-7
|
|
| 相关CAS号 |
|
|
| PubChem CID |
1103958
|
|
| 外观&性状 |
Light yellow to yellow solid powder
|
|
| 密度 |
1.9±0.1 g/cm3
|
|
| 沸点 |
361.2±37.0 °C at 760 mmHg
|
|
| 闪点 |
172.3±26.5 °C
|
|
| 蒸汽压 |
0.0±0.8 mmHg at 25°C
|
|
| 折射率 |
1.714
|
|
| LogP |
3.38
|
|
| tPSA |
88.1
|
|
| 氢键供体(HBD)数目 |
1
|
|
| 氢键受体(HBA)数目 |
4
|
|
| 可旋转键数目(RBC) |
2
|
|
| 重原子数目 |
18
|
|
| 分子复杂度/Complexity |
326
|
|
| 定义原子立体中心数目 |
0
|
|
| SMILES |
IC1C=CC(=CC=1)N([H])C(C1=CC=C([N+](=O)[O-])O1)=O
|
|
| InChi Key |
JBIKQXOZLBLMKI-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C11H7IN2O4/c12-7-1-3-8(4-2-7)13-11(15)9-5-6-10(18-9)14(16)17/h1-6H,(H,13,15)
|
|
| 化学名 |
N-(4-iodophenyl)-5-nitrofuran-2-carboxamide
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.03.00
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 1.67 mg/mL (4.66 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 悬浮液; 超声和加热处理
例如,若需制备1 mL的工作液,可将100 μL 16.7 mg/mL澄清的DMSO储备液加入到400 μL PEG300中,混匀;再向上述溶液中加入50 μL Tween-80,混匀;然后加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 0.25 mg/mL (0.70 mM) (饱和度未知) in 10% EtOH + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 2.5 mg/mL 澄清 EtOH 储备液加入到400 μL PEG300 中,混匀;再向上述溶液中加入50 μL Tween-80,混匀;然后加入450 μL 生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 0.25 mg/mL (0.70 mM) (饱和度未知) in 10% EtOH + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 配方 4 中的溶解度: 0.25 mg/mL (0.70 mM) in 10% EtOH + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 悬浊液; 超声助溶。 例如,若需制备1 mL的工作液,您可以将 100 μL 2.5 mg/mL 澄清 EtOH 储备液添加到 900 μL 玉米油中并充分混合。 配方 5 中的溶解度: 10 mg/mL (27.93 mM) in Cremophor EL (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶. 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7926 mL | 13.9630 mL | 27.9259 mL | |
| 5 mM | 0.5585 mL | 2.7926 mL | 5.5852 mL | |
| 10 mM | 0.2793 mL | 1.3963 mL | 2.7926 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。