| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|
| 体内研究 (In Vivo) |
多拉菌素 (10 mg/kg) 在体内有效,曼氏链球菌感染小鼠的蠕虫负担减少了 60.1%[3]。
|
|---|---|
| 动物实验 |
曼氏血吸虫小鼠模型:** 获得3周龄雌性NMRI小鼠,并在受控条件下(22°C,50%湿度,12小时光照/黑暗循环,自由摄食饮水)适应一周。小鼠皮下注射约100条从感染中间宿主螺类中获得的曼氏血吸虫尾蚴(利比里亚株)。感染7周后,4只小鼠接受多拉菌素治疗,8只小鼠作为对照组不接受治疗。多拉菌素溶于70:30的吐温/乙醇混合液中,最终浓度为10%。小鼠单次口服10 mg/kg,剂量根据小鼠体重调整。治疗3周后(感染后10周),用二氧化碳窒息法处死小鼠并进行解剖。收集肝门静脉和肠系膜静脉中的虫体,进行性别鉴定和计数。蠕虫负荷减少率(WBR)计算公式为:WBR (%) = 100% - (100% / 对照组平均蠕虫负荷 × 治疗组平均蠕虫负荷)。统计学比较采用 Kruskal-Wallis 检验和 Mann-Whitney U 检验,显著性水平为 p < 0.05。本研究已获得巴塞尔城市州兽医部门的批准(许可证号:2070)。[3]
|
| 药代性质 (ADME/PK) |
吸收、分布和排泄
在第一项研究中,10头荷斯坦奶牛接受了多拉菌素外用制剂治疗,剂量为0.58 mg/kg体重,并在56天后再次接受相同剂量的治疗。……在第一次和第二次治疗后,分别采集了49天和10天的牛奶样本。在第7天之前,每天采集两次样本;在第10、13、16、19、22、25、28、32、36、40和49天,每天采集一次样本。在再次治疗时,在第7天之前,每天采集两次样本;在第10天,每天采集一次样本。……牛奶中多拉菌素残留浓度在治疗后72小时达到平均值22 mg/kg的峰值。在给药后384小时(16天),多拉菌素残留的平均浓度降至定量限以下(3 mg/kg)。再次给药后,多拉菌素残留浓度逐渐升高,在给药后48小时达到平均最高值12 mg/kg;并在给药后240小时(10天)降至低于4 mg/kg。分别在给药后1、4和10天进行乳脂分析。在这些时间点,乳脂中多拉菌素残留的平均浓度分别为171 mg/kg、501 mg/kg和114 mg/kg。多拉菌素在乳脂中的残留浓度因子分别为29.6、32.2和24.7。 在第二项研究中,10头奶牛接受了多拉菌素滴剂局部治疗,剂量为0.58 mg/kg,并在56天后以相同剂量再次治疗。每天采集两次牛奶样本。治疗后45小时,牛奶中多拉菌素的浓度达到平均最大值9 mg/kg,并在治疗后237小时(10天)降至定量限以下。在第56天再次治疗后,残留浓度在93小时后达到平均最大值8 mg/kg,并在237小时(10天)后降至定量限以下。在第1、4和10天,乳脂中多拉菌素残留的平均浓度分别为91 mg/kg、142 mg/kg和55 mg/kg。乳脂中多拉菌素残留与乳中多拉菌素残留的浓缩倍数分别为14.2、20.9和14.1。 第三项研究测定了泌乳牛皮下注射0.23 mg/kg体重的多拉菌素制剂后,56天后以相同剂量再次给药后,多拉菌素残留的消耗情况。……乳中多拉菌素浓度逐渐升高,在67小时达到平均最大值45 mg/kg。随后,多拉菌素残留逐渐下降,在523小时(22天)时平均残留低于定量限(LOQ)。再次给药后,多拉菌素残留在56小时升高至平均最大值53 mg/kg。再次治疗后237小时(10天),残留浓度降至平均值25 mg/kg。在任何时间点,注射治疗产生的残留量均高于外用制剂治疗产生的残留量。分别于治疗后第1天、第4天和第10天早晨挤奶时采集乳脂样本进行乳脂分析。这些时间点乳脂中多拉菌素残留的平均浓度分别为557 mg/kg、1036 mg/kg和354 mg/kg。乳脂浓缩因子分别为24、24.2和23.4。 牛的舔舐行为最近被确定为局部给药伊维菌素动力学分布的决定因素。本研究记录了三种局部给药的内寄生虫杀灭剂因异体舔舐而在牛群间转移的发生情况和程度。四组荷斯坦奶牛,每组两头,分别接受多拉菌素、伊维菌素或莫昔菌素的外用制剂,或不进行任何处理。之后,这些奶牛被集中饲养在同一个围场中。在六头未接受处理的奶牛中,至少有五头观察到了全身暴露于每种外用驱虫药的情况。未接受处理的动物的血浆和粪便药物浓度曲线在不同动物之间以及同一动物的不同部位之间差异很大,有时甚至达到接受处理的动物的水平。通过同时静脉注射三种药物(混合剂)后测量血浆和粪便清除率来量化药物交换。多拉菌素、伊维菌素和莫昔菌素的血浆清除率分别为185±43、347±77和636±130 ml/kg/天,粪便清除率分别占血浆清除率的75±26%、28±13%和39±30%。未接受治疗的牛摄入的药物量分别占涂抹剂量(500微克/千克)的1.3%~21.3%(多拉菌素)、1.3%~16.1%(伊维菌素)和2.4%~10.6%(莫昔菌素)。所有未接受治疗的牛摄入的药物总量分别占涂抹于已治疗牛背部的每种药物总量的29%(多拉菌素)、19%(伊维菌素)和8.6%(莫昔菌素)。每头未接受治疗的牛摄入的内吸虫药累积量占涂抹剂量的1.3%~27.4%。由于舔舐行为摄入药物后,多拉菌素、伊维菌素和莫昔菌素的口服生物利用度分别为 13.5±9.4%、17.5±3.5% 和 26.1±11.1%。此处显示的药物交换程度引发了人们对药物疗效和安全性、耐药性的出现、已治疗和/或未治疗动物体内意外高残留水平以及高环境负担的担忧。 有关多拉菌素(共7种)的更多吸收、分布和排泄(完整)数据,请访问HSDB记录页面。 代谢/代谢物 将5位氚标记的多拉菌素以单剂量灌胃的方式给予Sprague-Dawley大鼠(2只雄性,以5 mg/kg体重溶于丙二醇:甘油中)、比格犬(1只雌性,以3.5 mg/kg体重溶于芝麻油中,通过灌胃方式)和牛(5头雄性,以0.2 mg/kg体重皮下注射)。在每种动物的肝脏和粪便以及牛的脂肪中鉴定出以下代谢物:未改变的多拉菌素、3"-O-去甲基多拉菌素、24-羟甲基多拉菌素和24-羟甲基-3"-O-去甲基多拉菌素。 在所有研究物种(大鼠、狗、猪、牛)中,多拉菌素的代谢产物相似。这些代谢产物比多拉菌素极性更强,是远端糖环O-去甲基化、24-甲基羟基化以及这两种生物转化共同作用的结果。 生物半衰期 采用原型商业制剂(75%芝麻油/25%油酸乙酯),以0.3 mg/kg体重肌注(3H)多拉菌素,对8头猪(4头去势雄性猪和4头雌性猪,每头体重约40 kg)进行血浆动力学测定。……总(3H)标记物质和未代谢多拉菌素从血浆中消除的表观末端半衰期分别为7.7天和6.4天。 |
| 毒性/毒理 (Toxicokinetics/TK) |
非人类毒性值
小鼠(CD-1)口服LD50 >2000 mg/kg 体重 /0.1% 甲基纤维素水溶液/ /取自表格/ 大鼠(Sprague-Dawley,雄性)腹腔注射LD50 >300 mg/kg 体重 /0.1% 甲基纤维素水溶液/ /取自表格/ |
| 参考文献 |
|
| 其他信息 |
多拉菌素是一种兽药,经美国食品药品监督管理局(FDA)批准,用于治疗牛的寄生虫,例如胃肠道线虫、肺线虫、眼线虫、蛆虫、吸虱和疥螨。
另见:多拉菌素;左旋咪唑(成分);多拉菌素;盐酸左旋咪唑(成分)。 作用机制 阿维菌素类药物可使线虫和节肢动物快速、非痉挛性麻痹。阿维菌素类药物的一个共同特征似乎是调节线虫神经细胞以及节肢动物神经和肌肉细胞中的跨膜氯离子(Cl-)通道活性。这些Cl-通道可能由多种神经递质受体激活,包括γ-氨基丁酸(GABA)、谷氨酸和乙酰胆碱。阿维菌素激活氯离子通道,导致氯离子电导增加,进而改变膜电位,最终抑制靶神经或肌肉细胞的电活动。γ-氨基丁酸(GABA)也是哺乳动物中枢神经系统(CNS)中的主要抑制性神经递质,阿维菌素对哺乳动物GABA受体/氯离子通道复合物具有内在活性。据报道,阿维菌素还能与哺乳动物中枢神经系统特有的甘氨酸受体/氯离子通道复合物结合。阿维菌素极难穿透血脑屏障,这或许可以解释为何这些化合物在哺乳动物体内具有较高的安全性。/阿维菌素/ 治疗用途 兽用药物:抗寄生虫药 兽用:多拉菌素是一种用于牛和猪的体外和体内寄生虫杀灭剂。它是阿维菌素类半合成药物,结构与阿维菌素和伊维菌素相似。 兽医:多拉菌素(NADA 141-095)获准外用,用于治疗和控制各种蠕虫(蛔虫、肺虫和眼虫)、幼虫、虱子、角蝇和疥螨。它还获准用于控制感染,并可在治疗后21天内预防由库珀线虫(Cooperia oncophora)和牛肺线虫(Dictyocaulus viviparus)引起的再次感染,在28天内预防由奥斯特线虫(Ostertagia ostertagi)、点状库珀线虫(C. punctata)和放射状食道口线虫(Oesophagostomum radiatum)引起的再次感染,在35天内预防由血矛线虫(Haemonchus placei)引起的再次感染。 兽医:目的:确定多拉菌素外用制剂对牛达玛利线虫(Damalinia bovis)、宽腹血矛线虫(Haematopinus eurysternus)、牛细长线虫(Linognathus vituli)、毛状线虫(Solenopotes capillatus)、牛绒螨(Chorioptes bovis)、疥螨(Sarcoptes scabiei)、牛皮螨(Hypoderma bovis)和线状皮螨(Hypoderma lineatus)的有效性。动物:自然感染或人工感染一种或多种虱子、螨虫或幼虫的各种年龄的牛。方法:在10项虱子研究和6项螨虫研究中,牛在第0天接受多拉菌素(500微克/公斤,局部用药)治疗,并在治疗后28天再次接受治疗。自然感染牛的牛疥螨和疥座圈螨虫数量在第14至15天降至0,人工感染牛的牛疥螨数量也接近0。在幼虫研究中,136头对照组牛中有107头出现疥疮,而136头多拉菌素治疗组牛中仅有2头出现疥疮,治愈率为98.5%。 有关多拉菌素(共6项)的更多治疗用途(完整)数据,请访问HSDB记录页面。 |
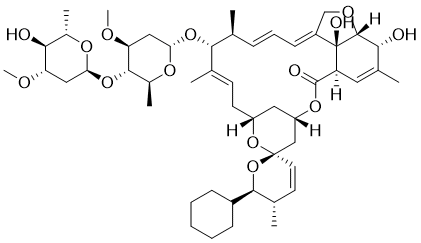
| 分子式 |
C50H74O14
|
|---|---|
| 分子量 |
899.12
|
| 精确质量 |
898.507
|
| 元素分析 |
C, 66.79; H, 8.30; O, 24.91
|
| CAS号 |
117704-25-3
|
| 相关CAS号 |
117704-25-3;
|
| PubChem CID |
9832750
|
| 外观&性状 |
White to off-white solid powder
|
| 密度 |
1.3±0.1 g/cm3
|
| 沸点 |
967.4±65.0 °C at 760 mmHg
|
| 熔点 |
116 - 119ºC
|
| 闪点 |
274.4±27.8 °C
|
| 蒸汽压 |
0.0±0.6 mmHg at 25°C
|
| 折射率 |
1.580
|
| LogP |
7.16
|
| tPSA |
170.06
|
| 氢键供体(HBD)数目 |
3
|
| 氢键受体(HBA)数目 |
14
|
| 可旋转键数目(RBC) |
7
|
| 重原子数目 |
64
|
| 分子复杂度/Complexity |
1790
|
| 定义原子立体中心数目 |
19
|
| SMILES |
C[C@H]1/C=C/C=C/2\CO[C@H]3[C@@]2([C@@H](C=C([C@H]3O)C)C(=O)O[C@H]4C[C@@H](C/C=C(/[C@H]1O[C@H]5C[C@@H]([C@H]([C@@H](O5)C)O[C@H]6C[C@@H]([C@H]([C@@H](O6)C)O)OC)OC)\C)O[C@]7(C4)C=C[C@@H]([C@H](O7)C8CCCCC8)C)O
|
| InChi Key |
QLFZZSKTJWDQOS-CYWJOYLHSA-N
|
| InChi Code |
InChI=1S/C50H74O14/c1-27-13-12-16-34-26-57-47-42(51)30(4)21-37(50(34,47)54)48(53)60-36-22-35(63-49(25-36)20-19-29(3)45(64-49)33-14-10-9-11-15-33)18-17-28(2)44(27)61-41-24-39(56-8)46(32(6)59-41)62-40-23-38(55-7)43(52)31(5)58-40/h12-13,16-17,19-21,27,29,31-33,35-47,51-52,54H,9-11,14-15,18,22-26H2,1-8H3/b13-12+,28-17+,34-16+/t27-,29-,31-,32-,35+,36-,37-,38-,39-,40-,41-,42+,43-,44+,45-,46-,47+,49+,50+/m0/s1
|
| 化学名 |
(1'R,2S,4'S,5S,6R,8'R,10'E,12'R,13'S,14'E,20'R,21'R,24'S)-6-Cyclohexyl-21',24'-dihydroxy-12'-{[(2R,4S,5S,6S)-5-{[(2S,4S,5S,6S)-5-hydroxy-4-methoxy-6-methyloxan-2-yl]oxy}-4-methoxy-6-methyloxan-2-yl]oxy}-5,11',13',22'-tetramethyl-5,6-dihydro-3',7',19'-trioxaspiro[pyran-2,6'-tetracyclo[15.6.1.14,8.020,24]pentacosane]-10',14',16',22'-tetraen-2'-one
|
| 别名 |
Dectomax;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 本产品在运输和储存过程中需避光。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : 50~100 mg/mL (55.61~111.22 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (2.78 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: 2.5 mg/mL (2.78 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 悬浊液; 超声助溶。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.5 mg/mL (2.78 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 配方 4 中的溶解度: 10% DMSO+40% PEG300+5% Tween-80+45% Saline: ≥ 2.5 mg/mL (2.78 mM) 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.1122 mL | 5.5610 mL | 11.1220 mL | |
| 5 mM | 0.2224 mL | 1.1122 mL | 2.2244 mL | |
| 10 mM | 0.1112 mL | 0.5561 mL | 1.1122 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
|