| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
IOX1 is a selective inhibitor of the JMJD2 subfamily of Jumonji C-domain-containing histone demethylases (JmjC-KDMs). It exhibits high inhibitory activity against JMJD2A with an IC50 of 1.2 μM, JMJD2B (IC50 = 2.7 μM), and JMJD2D (IC50 = 5.8 μM). It shows minimal inhibition (IC50 >50 μM) against other JmjC-KDMs (e.g., JMJD1A, KDM5B) and non-JmjC enzymes (e.g., LSD1, HDAC1) [1,4]
- IOX1 does not inhibit m6A-RNA demethylases (e.g., ALKBH5) or methyltransferases (e.g., METTL3-METTL14 complex) at concentrations up to 20 μM, confirming its specificity for JMJD2 subfamily members [3] |
|---|---|
| 体外研究 (In Vitro) |
IOX1(0-200 μM;2 小时)以浓度依赖性方式抑制血管紧张素 II (Ang II) 诱导的血管平滑肌细胞 (VSMC) 的迁移和增殖[2]。 IOX1(200 μM;24 小时)增加处于 G0/G1 期的细胞比例,从而阻止血管紧张素 II (Ang II)-VSMC 进入细胞周期[2]。 IOX1(50-200 μM;2 小时)以浓度依赖性方式减弱细胞周期蛋白 D1 并上调 p21 mRNA 水平[2]。 IOX1(50-200 μM;2 小时)恢复 H3K9me3,介导细胞周期蛋白 D1 和 p21 表达 [2]。
抑制JMJD2A酶活性:IOX1(0.1-20 μM)可浓度依赖性抑制JMJD2A介导的H3K9me3(JMJD2A底物)去甲基化。基于放射性的去甲基化酶实验显示,5 μM浓度下,其较溶剂对照组降低JMJD2A活性82±6%[1] - 天然IOX1细胞渗透性差:在HeLa细胞中,10 μM IOX1处理24小时仅使细胞内H3K9me3水平(JMJD2A抑制标志物)升高15±3%,提示细胞膜渗透性差;其细胞渗透性酯衍生物(IOX1-ester)可改善细胞内蓄积,相同浓度下使H3K9me3水平升高65±7%[1] - 抑制Ang II诱导的VSMC增殖与迁移:在血管紧张素II(Ang II,100 nM)刺激的原代大鼠血管平滑肌细胞(VSMC)中,IOX1(0.5-10 μM)抑制细胞增殖的EC50为3.5 μM。5 μM浓度下,BrdU掺入率(增殖标志物)较Ang II处理组降低68±5%,Transwell迁移率降低55±6%[2] - 调节VSMC中细胞周期相关蛋白:Ang II刺激的VSMC经IOX1(5 μM)处理24小时后,Western blot显示Cyclin D1降低45±4%、CDK4降低38±3%,细胞周期依赖性激酶抑制剂p21升高2.3±0.2倍;流式细胞术证实细胞阻滞于G1期(G1期比例从45±3%升至68±4%)[2] - 抑制肝癌干细胞样细胞(LCSC)自我更新:在Huh7来源的LCSC中,IOX1(2-10 μM)浓度依赖性抑制球状体(sphere)形成,10 μM浓度下直径>50 μm的球状体数量较溶剂组减少72±8%。qPCR和Western blot显示IOX1使EpCAM的mRNA(降低65±7%)和蛋白(降低58±6%)、Sox9的mRNA(降低60±5%)和蛋白(降低52±4%)水平下调[4] - 对正常细胞低毒性:MTT实验显示,IOX1(0.1-20 μM)处理正常人肝星状细胞(NHSC)或原代人脐静脉内皮细胞(HUVEC)72小时后无显著细胞毒性,CC50 >20 μM[4] |
| 体内研究 (In Vivo) |
在体内,IOX1 (5-c-8HQ)(口服灌胃;10–20 mg/kg;12 天)减弱肝癌干样细胞 (LCSC) 的自我更新并抑制肿瘤生长[1]。
抑制大鼠颈动脉损伤模型的新生内膜增生:雄性SD大鼠(250-300 g)行颈动脉球囊损伤术后,随机分为2组(n=8/组):溶剂组(10% DMSO/PBS)和IOX1组(5 mg/kg,溶于10% DMSO/PBS)。两组均每日腹腔注射一次,连续21天。实验结束时,H&E染色显示IOX1使新生内膜面积减少52±7%(新生内膜/中膜比值:0.8±0.1 vs 溶剂组1.7±0.2);PCNA(增殖标志物)免疫组化显示新生内膜中PCNA阳性VSMC减少48±6%[2] - 裸鼠肝癌异种移植模型的抗肿瘤疗效:6-8周龄雌性裸鼠皮下注射1×106个Huh7来源的LCSC,当肿瘤体积达100-150 mm³时,分为2组(n=6/组):溶剂组(10% DMSO/PBS)和IOX1组(10 mg/kg,腹腔注射,每日一次,连续28天)。IOX1的肿瘤生长抑制率(TGI)达65±8%(肿瘤体积:320±40 mm³ vs 溶剂组910±80 mm³);肿瘤组织分析显示球状体形成单位(SFU)减少55±6%,EpCAM/Sox9蛋白水平分别降低50±5%和45±4%[4] - 保护异种移植小鼠正常肝功能:IOX1处理组小鼠的血清丙氨酸转氨酶(ALT)、天冬氨酸转氨酶(AST)和白蛋白水平与溶剂组无显著差异,提示无肝毒性[4] |
| 酶活实验 |
JMJD2A活性测定(放射性法):将重组人源JMJD2A(与辅因子α-酮戊二酸形成复合物)与含50 mM Tris-HCl(pH 7.6)、0.1 mM FeSO4、2 mM抗坏血酸和10 μM [3H]标记H3K9me3肽段(组蛋白H3的1-20位氨基酸)的反应缓冲液混合。加入浓度范围为0.01-50 μM的IOX1,37°C孵育120分钟。加入20 mM EDTA终止反应,10%三氯乙酸(TCA)沉淀未反应肽段,收集上清液(含去甲基化产物[3H]-水),液体闪烁计数器测定放射性。相对于溶剂组计算抑制率,非线性回归法求得IC50[1]
- JMJD2D活性测定(荧光法):重组人源JMJD2D与50 mM Tris-HCl(pH 7.5)、0.1 mM FeSO4、2 mM α-酮戊二酸和10 μM荧光标记H3K9me3肽段混合,加入IOX1(0.1-50 μM),37°C孵育90分钟。20 mM EDTA终止反应,检测荧光强度(激发光485 nm,发射光520 nm)。荧光强度与剩余H3K9me3量成正比(与JMJD2D活性成反比),据此计算IC50[4] - ALKBH5/METTL3-METTL14选择性测定:采用[3H]标记m6A-RNA底物测试20 μM IOX1对ALKBH5(m6A-RNA去甲基化酶)的抑制活性,采用S-腺苷甲硫氨酸(SAM)底物测试其对METTL3-METTL14(m6A-RNA甲基转移酶)的活性。放射性检测显示两种酶的抑制率均<8%,证实无交叉反应[3] |
| 细胞实验 |
VSMC增殖实验(BrdU掺入法):原代大鼠VSMC以5×103个/孔接种于96孔板,血清饥饿24小时。Ang II(100 nM)刺激前1小时加入IOX1(0.5-10 μM),48小时后加入BrdU试剂孵育4小时。4%甲醛固定细胞,0.1% Triton X-100通透,加入抗BrdU抗体,检测荧光强度(激发光488 nm,发射光530 nm),相对于Ang II处理组计算增殖率[2]
- VSMC迁移实验(Transwell法):VSMC血清饥饿24小时后消化,重悬于含IOX1(0.5-10 μM)的无血清培养基。将1×105个细胞加入Transwell小室(8 μm孔径)上室,下室加入含10% FBS和Ang II(100 nM)的培养基。24小时后去除上室细胞,下室细胞经固定、0.1%结晶紫染色后计数,迁移率以相对于Ang II处理组的百分比表示[2] - 肝癌干细胞样细胞(LCSC)球状体形成实验:Huh7细胞在干细胞培养基(DMEM/F12 + 20 ng/mL EGF + 20 ng/mL bFGF + B27添加剂)中培养以富集LCSC,加入IOX1(2-10 μM)后,将1×103个细胞接种于超低吸附6孔板。7天后计数直径>50 μm的球状体,球状体形成效率(SFE)计算公式为:(球状体数量/接种细胞数)×100%[4] - 细胞周期分析(流式细胞术):Ang II刺激的VSMC经IOX1(5 μM)处理24小时后收集,-20°C下70%乙醇固定过夜,加入含RNase A(100 μg/mL)的碘化丙啶(PI)染色。流式细胞术分析细胞周期分布(G0/G1、S、G2/M期),ModFit软件计算各期细胞百分比[2] - LCSC中EpCAM/Sox9的Western blot检测:LCSC经IOX1(2-10 μM)处理48小时后,用含蛋白酶抑制剂的RIPA缓冲液裂解,30 μg蛋白进行10% SDS-PAGE电泳。膜与抗EpCAM、抗Sox9或抗GAPDH(内参)抗体孵育,再与HRP偶联二抗孵育。ECL化学发光显影,ImageJ软件定量条带强度[4] |
| 动物实验 |
动物/疾病模型: 6周龄雄性BALB/c裸鼠[4]
剂量: 10 mg/kg,20 mg/kg 给药途径: 12天 实验结果: 治疗后未观察到小鼠体重减轻和主要器官毒性,表明未出现明显的不良反应。抑制LCSC原位移植瘤的生长。显著降低LCSC原位移植瘤中EpCAM和Sox9的蛋白水平,抑制LCSC原位移植瘤的生长。减少Ki67阳性肿瘤细胞,并以剂量依赖的方式显著降低LCSC的肿瘤球形成能力。 大鼠颈动脉球囊损伤模型:雄性SD大鼠(250-300 g)用异氟烷麻醉。将2F Fogarty球囊导管插入左侧颈总动脉,充气至2个大气压,并回撤3次以诱导内皮损伤。大鼠随机分为两组(每组n=8):1. 对照组:腹腔注射0.2 mL 10% DMSO PBS溶液,每日一次,连续21天;2. IOX1组:腹腔注射IOX1(5 mg/kg,溶于10% DMSO PBS溶液),每日一次,连续21天。第21天,处死大鼠,切取左侧颈动脉,用4%甲醛固定,石蜡包埋,切片(5 μm)。切片经苏木精-伊红染色(H&E染色)用于新生内膜测量,或进行PCNA免疫组化染色[2] - 裸鼠肝癌异种移植模型:雌性裸鼠(6-8周龄)适应环境1周。将Huh7来源的肝癌干细胞(LCSCs,1×10⁶个细胞)悬浮于50% Matrigel中,皮下注射至小鼠右侧腹部。当肿瘤体积达到100-150 mm³时,将小鼠分为两组(每组n=6):1. 载体组:腹腔注射0.2 mL 10% DMSO PBS溶液,每日一次,连续28天;2. IOX1组:腹腔注射IOX1(10 mg/kg,溶于10% DMSO PBS溶液),每日一次,连续28天。每隔3天测量一次肿瘤体积(V = L×W²/2,L=最长直径,W=最短直径)。第28天,处死小鼠,切除肿瘤进行球体形成实验和蛋白质印迹分析,并收集血清进行肝功能检测[4]。 |
| 药代性质 (ADME/PK) |
细胞渗透性:天然IOX1的细胞膜渗透性较差。在Caco-2细胞单层渗透试验中,IOX1的表观渗透系数(Papp)为0.3×10⁻⁶ cm/s,比其酯衍生物(IOX1酯,Papp = 3.2×10⁻⁶ cm/s)低10倍[1]。文献中未提供IOX1的口服吸收、分布、代谢、排泄或药代动力学参数(例如,半衰期、口服生物利用度、清除率)的数据[1,2,4]。
|
| 毒性/毒理 (Toxicokinetics/TK) |
体外细胞毒性:IOX1 对正常细胞毒性较低。在 NHSC、HUVEC 和原代大鼠肝细胞中,处理 72 小时后 CC50 > 20 μM。浓度高达 10 μM(功能测定中使用的最高浓度)时,未观察到细胞形态或活力的显著变化 [2,4]。体内急性毒性:在 SD 大鼠腹腔注射 IOX1(15 mg/kg/天,连续 7 天)后,未观察到死亡或严重毒性(例如,嗜睡、体重减轻)。体重变化为+2±1%(对照组为+3±1%),血清ALT、AST、BUN和肌酐水平均在正常范围内[2]
- 异种移植小鼠的慢性毒性:在接受IOX1(10 mg/kg/天,持续28天)治疗的裸鼠中,未观察到肝脏、肾脏、脾脏或心脏组织的组织病理学损伤。外周血细胞计数(白细胞、血小板、红细胞)正常,证实无骨髓抑制[4] - 血浆蛋白结合率:文献中未提供IOX1血浆蛋白结合率的数据[1,2,4] - 药物相互作用:文献中未提供涉及IOX1的药物相互作用的数据[1,2,4] |
| 参考文献 |
|
| 其他信息 |
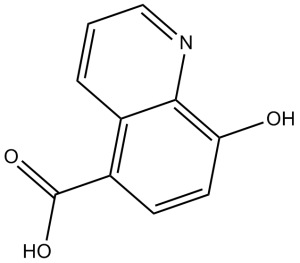
8-羟基-5-喹啉羧酸属于喹啉类化合物。
作用机制:IOX1通过抑制JMJD2亚家族成员(JMJD2A、JMJD2B、JMJD2D)发挥其生物学效应。这些酶可使H3K9me3/H3K9me2(抑制性组蛋白标记)去甲基化;IOX1的抑制作用可增加细胞内H3K9me3水平,从而导致靶基因(例如,VSMC中的Cyclin D1、LCSC中的EpCAM/Sox9)的转录抑制,并随后抑制细胞增殖、迁移或自我更新[2,4]。 -酯衍生物的开发原理:天然IOX1的细胞渗透性差,限制了其细胞内活性。细胞渗透性酯衍生物(IOX1-酯)旨在增强其膜渗透性;进入细胞后,IOX1 水解为活性 IOX1,从而提高靶向结合能力 [1] - 治疗潜力:IOX1 在两种疾病模型中显示出临床前潜力:1) 通过抑制血管平滑肌细胞 (VSMC) 增殖/迁移治疗血管内膜增生(例如支架植入后);2) 通过靶向癌干细胞样细胞治疗肝癌。其对正常细胞的低毒性支持其作为靶向疗法的进一步开发 [2,4] - 局限性:IOX1 细胞渗透性差,且未见口服生物利用度报道,因此在临床前模型中需要肠外给药。目前尚无临床数据(例如,人体疗效、药代动力学),也未评估其在大型动物中的长期毒性[1,4] - 选择性优势:与泛JmjC抑制剂不同,IOX1特异性靶向JMJD2亚家族,从而减少对其他组蛋白去甲基化酶的脱靶效应,并最大限度地降低潜在的不良反应[1,3] |
| 分子式 |
C10H7NO3
|
|
|---|---|---|
| 分子量 |
189.17
|
|
| 精确质量 |
189.042
|
|
| CAS号 |
5852-78-8
|
|
| 相关CAS号 |
|
|
| PubChem CID |
459617
|
|
| 外观&性状 |
White to gray solid powder
|
|
| 密度 |
1.5±0.1 g/cm3
|
|
| 沸点 |
464.5±30.0 °C at 760 mmHg
|
|
| 闪点 |
234.7±24.6 °C
|
|
| 蒸汽压 |
0.0±1.2 mmHg at 25°C
|
|
| 折射率 |
1.730
|
|
| LogP |
1.81
|
|
| tPSA |
70.42
|
|
| 氢键供体(HBD)数目 |
2
|
|
| 氢键受体(HBA)数目 |
4
|
|
| 可旋转键数目(RBC) |
1
|
|
| 重原子数目 |
14
|
|
| 分子复杂度/Complexity |
231
|
|
| 定义原子立体中心数目 |
0
|
|
| InChi Key |
JGRPKOGHYBAVMW-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C10H7NO3/c12-8-4-3-7(10(13)14)6-2-1-5-11-9(6)8/h1-5,12H,(H,13,14)
|
|
| 化学名 |
8-Hydroxyquinoline-5-carboxylic acid
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.08 mg/mL (11.00 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 20.8 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.08 mg/mL (11.00 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.2863 mL | 26.4313 mL | 52.8625 mL | |
| 5 mM | 1.0573 mL | 5.2863 mL | 10.5725 mL | |
| 10 mM | 0.5286 mL | 2.6431 mL | 5.2863 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。