| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g |
|
||
| 5g |
|
||
| 10g |
|
||
| Other Sizes |
|
| 体外研究 (In Vitro) |
体外活性:氯诺昔康在缓解妇科或骨科手术后的术后疼痛方面与阿片类镇痛药吗啡、哌替啶(哌替啶)和曲马多一样有效,在口腔手术后与其他非甾体抗炎药一样有效。氯诺昔康在缓解骨关节炎、类风湿性关节炎、强直性脊柱炎、急性坐骨神经痛和腰痛方面也与其他非甾体抗炎药一样有效。细胞测定:对完整人体细胞的研究表明,氯诺昔康强烈抑制 COX-1 和 COX-2,在大量测试的 NSAID 中 IC50 最低。在全血 COX-1/-2 中也获得了类似的结果。此外,氯诺昔康以剂量依赖性方式抑制 NO 形成,IC50 为 65 μM。
|
||
|---|---|---|---|
| 体内研究 (In Vivo) |
氯诺昔康剂量相关地减少了 c-Fos-LI 神经元的总数,其中最高剂量 9 mg/kg 减少 75%,低剂量 0.3 mg/kg 减少 45%,效果最强。氯诺昔康(0.1、0.3 mg/kg、1 mg/kg、3 mg/kg 和 9 mg/kg,静脉注射)显着减少浅表处 c-Fos-LI 神经元的数量(24%、33%、53%、脊髓背角的椎板分别减少 54% 和 63%)和深部(分别减少 28%、48%、62%、69% 和 79%)。氯诺昔康以有效剂量减少痛觉过敏,在慢性关节炎大鼠模型中分别提供 0.083 mg/kg、3.9 mg/kg 和 4.3 mg/kg 的 50% 抑制 (ED50)。氯诺昔康显着降低大鼠爪子渗出液和脑脊液中 PGE2 的水平。氯诺昔康 0.16 mg/kg、塞来昔布 4 mg/kg 和洛索洛芬 2.4 mg/kg 显着降低急性水肿大鼠的痛觉过敏,程度相似。
|
||
| 动物实验 |
|
||
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
Lornoxicam is absorbed rapidly and almost completely from the GI tract (90-100%). Metabolism / Metabolites Lornoxicam is metabolized completely by cyp 2C9 with the principal metabolite being 5'-hydroxy-lornoxicam and only negligible amounts of intact lornoxicam are excreted unchanged in the urine. Approximately 2/3 of the drug is eliminated via the liver and 1/3 via the kidneys in the active form. Lornoxicam has known human metabolites that include 5'-Hydroxylornoxicam. Biological Half-Life 3-5 hours |
||
| 毒性/毒理 (Toxicokinetics/TK) |
Protein Binding
Lornoxicam is 99% bound to plasma proteins (almost exlusively to serum albumin). |
||
| 参考文献 |
Drugs.1996 Apr;51(4):639-57;Inflammopharmacology.1997;5(4):331-41.
|
||
| 其他信息 |
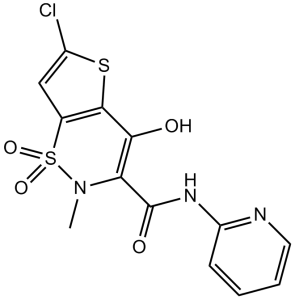
Lornoxicam is a thienothiazine-derived monocarboxylic acid amide obtained by formal condensation of the carboxy group of 6-chloro-4-hydroxy-2-methylthieno[2,3-e][1,2]thiazine-3-carboxylic acid 1,1-dioxide with the amino group of 2-aminopyridine. Used for the treatment of pain, primarily resulting from inflammatory diseases of the joints, osteoarthritis, surgery, sciatica and other inflammations. It has a role as a non-steroidal anti-inflammatory drug, a non-narcotic analgesic and an antipyretic. It is a thienothiazine, a member of pyridines, a monocarboxylic acid amide, an organochlorine compound and a heteroaryl hydroxy compound.
Lornoxicam (chlortenoxicam) is a new nonsteroidal anti-inflammatory drug (NSAID) of the oxicam class with analgesic, anti-inflammatory and antipyretic properties. Lornoxicam differs from other oxicam compounds in its potent inhibition of prostaglandin biosynthesis, a property that explains the particularly pronounced efficacy of the drug. Lornoxicam is approved for use in Japan. Lornoxicam is an orally bioavailable oxicam and non-steroidal anti-inflammatory drug (NSAID), with analgesic, anti-pyretic, anti-thrombotic and anti-inflammatory activities. Upon oral administration, lornoxicam binds to and inhibits the activity of the cyclooxygenase enzymes (COX) type 1 (COX-1) and type 2 (COX-2). This blocks COX-mediated signaling pathways, which leads to reduced prostaglandin and thromboxane production and decreased pain, fever and inflammation. Drug Indication For the treatment of acute mild to moderate pain, as well as pain and inflammation of the joints caused by certain types of rheumatic diseases. Mechanism of Action Like other NSAIDS, lornoxicam's anti-inflammatory and analgesic activity is related to its inhibitory action on prostaglandin and thromboxane synthesis through the inhibition of both COX-1 and COX-2. This leads to the reduction of inflammation, pain, fever, and swelling, which are mediated by prostaglandins. However, the exact mechanism of lornoxicam, like that of the other NSAIDs, has not been fully determined. Pharmacodynamics Lornoxicam is a non-steroidal anti-inflammatory drug (NSAID) that belongs to the oxicam class. As with other NSAIDS, lornoxicam is a potent inhibitor of the cyclooxgenase enzymes, which are responsible for catalyzing the formation of prostaglandins (act as messenger molecules in the process of inflammation) and thromboxane from arachidonic acid. Unlike some NSAIDS, lornoxicam's inhibition of cyclooxygenase does not lead to an increase in leukotriene formation, meaning that arachidonic acid is not moved to the 5-lipoxygenase cascade, resulting in the minimization of the risk of adverse events. |
| 分子式 |
C13H10CLN3O4S2
|
|
|---|---|---|
| 分子量 |
371.82
|
|
| 精确质量 |
370.98
|
|
| CAS号 |
70374-39-9
|
|
| 相关CAS号 |
Lornoxicam-d4;1216527-48-8
|
|
| PubChem CID |
54690031
|
|
| 外观&性状 |
Light yellow to yellow solid powder
|
|
| 密度 |
1.7±0.1 g/cm3
|
|
| 熔点 |
225-230°C (dec.)
|
|
| 折射率 |
1.741
|
|
| LogP |
2.18
|
|
| tPSA |
136.22
|
|
| 氢键供体(HBD)数目 |
2
|
|
| 氢键受体(HBA)数目 |
7
|
|
| 可旋转键数目(RBC) |
2
|
|
| 重原子数目 |
23
|
|
| 分子复杂度/Complexity |
634
|
|
| 定义原子立体中心数目 |
0
|
|
| InChi Key |
WLHQHAUOOXYABV-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C13H10ClN3O4S2/c1-17-10(13(19)16-9-4-2-3-5-15-9)11(18)12-7(23(17,20)21)6-8(14)22-12/h2-6,18H,1H3,(H,15,16,19)
|
|
| 化学名 |
6-chloro-4-hydroxy-2-methyl-1,1-dioxo-N-pyridin-2-ylthieno[2,3-e]thiazine-3-carboxamide
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|---|
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6895 mL | 13.4474 mL | 26.8947 mL | |
| 5 mM | 0.5379 mL | 2.6895 mL | 5.3789 mL | |
| 10 mM | 0.2689 mL | 1.3447 mL | 2.6895 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT05679453 | Completed | Drug: Lornoxicam 8 Mg Oral Tablet | Pain, Acute Edema Trismus |
Kutahya Health Sciences University | July 20, 2022 | Phase 4 |
| NCT00997750 | Completed | Drug: Lornoxicam | Acute Coronary Syndrome | Central Clinical Hospital w/Outpatient Health Center of Business Administration for the President of Russian Federation | March 2007 | Phase 4 |
| NCT02750917 | Completed | Drug: Lornoxicam Drug: Etoricoxib |
Postoperative Pain | Foisor Orthopedics Clinical Hospital | September 2014 | Phase 3 |
| NCT01117948 | Terminated Has Results | Drug: Lornoxicam Drug: Placebo |
Alzheimer´s Disease | JSW Lifesciences | September 2009 | Phase 2 |
| NCT01055470 | Completed | Drug: Diclofenac Drug: Lornoxicam |
Osteoarthritis of Knee Joint | Government Medical College, Bhavnagar | December 2008 | Not Applicable |