| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|
| 靶点 |
Fluorescent dye for monitoring glucose uptake in living cells and tissues
Intracellular chloride ions ([Cl⁻]i) (no traditional "target" like enzyme/receptor; binds [Cl⁻]i via electrostatic interaction for quantitative fluorescence detection). - Dissociation constant (Kd) for [Cl⁻]i: ~140 mM (determined by fluorescence titration in vitro, consistent with physiological [Cl⁻]i range (10-60 mM))[3] |
|---|---|
| 体外研究 (In Vitro) |
1. MQAE 工作液配制
用 Krebs-Hepes 缓冲液(20 mM HEPES、128 mM NaCl、2.5 mM KCl、2.7 mM CaCl₂、1 mM MgCl₂、16 mM 葡萄糖,pH 7.4)稀释储备液,配制成终浓度为 5–10 mM 的 MQAE 工作液。 注:建议根据具体实验需求优化工作液浓度,并现配现用。 2. 细胞染色步骤 2.1 用 Krebs-HEPES 缓冲液洗涤细胞三次,每次 2 分钟。 2.2 加入 1 mL MQAE 工作液,室温避光孵育 30–60 分钟。 2.3 用 PBS 洗涤细胞两次,每次 5 分钟。 2.4 使用荧光显微镜观察或流式细胞仪检测染色效果。 3. 保存条件 -20℃ 避光保存,有效期为一年。 4. 注意事项 4.1 推荐用于染色的细胞密度为 8–10×10⁶ 个/mL。 4.2 请根据实际实验体系优化 MQAE 工作液浓度及孵育时间。 4.3 本品仅限科研专业人员使用,严禁用于临床诊断、治疗、食品或药品。 4.4 操作时请穿实验服并佩戴一次性手套,以确保安全与健康。 在体外(步骤1-2)或体内(步骤3-7)进行MQAE染色,然后进行成像(步骤8)。 MQAE对培养细胞或组织切片的染色 该方案允许对切片上部70-120µm进行高质量染色,以便根据其形态识别不同类型的神经元(Marandi等人,2002;参见图1)。 1. 将MQAE溶解于小鼠标准生理盐水中至终浓度6mM。 2. 将培养的细胞或脑切片与该溶液在37℃下孵育10分钟,然后用无染料生理盐水冲洗10 - 15分钟。 1. 神经元与HEK293细胞的双光子[Cl⁻]i成像(文献[1]): - 原代小鼠海马神经元(P0-P2幼鼠,培养7-10天):负载MQAE(5-10 mM,37℃孵育30分钟)。双光子显微镜(激发光720 nm,发射光460±20 nm)显示,当[Cl⁻]i从10 mM升高至40 mM时,荧光强度降低~40%;对生理浓度的Na⁺、K⁺、Ca²⁺无交叉反应。 - HEK293细胞:10 mM MQAE可稳定报告[Cl⁻]i变化>2小时,荧光信号波动<5%(无光漂白相关干扰)[1] 2. 肾近端小管上皮细胞的氯离子外流检测(文献[2]): - 大鼠肾近端小管上皮细胞(DMEM/F12 + 10% FBS培养):负载MQAE(5 mM,Krebs-Ringer缓冲液中37℃孵育45分钟)。加入10 μM呋塞米(氯离子外流诱导剂)后,15分钟内MQAE荧光强度升高~35%(vs基线),与X射线微分析结果(氯离子含量降低~32%)一致,证实[Cl⁻]i定量的可靠性[2] 3. 主动脉平滑肌细胞的[Cl⁻]i测量(文献[3]): - 培养大鼠主动脉平滑肌细胞:负载MQAE(5 mM,HBS缓冲液pH 7.4中37℃孵育60分钟)。1 μM去甲肾上腺素处理使[Cl⁻]i从~35 mM升高至~55 mM,伴随MQAE荧光强度降低~30%。负载2小时后细胞存活率>90%(台盼蓝排斥法)[3] 4. 气道纤毛细胞不受体积影响的[Cl⁻]i检测(文献[4]): - 小鼠气道纤毛细胞(气管分离):负载MQAE(5 mM,含25 mM甘露醇的HBS中37℃孵育30分钟)。双光子显微镜(激发光730 nm,发射光460 nm)显示,即使细胞因渗透压冲击体积变化±15%,MQAE荧光仍能准确反映[Cl⁻]i(与氯离子电极数据相关系数R²=0.92)。纤毛摆动频率无变化(~12 Hz vs对照组~11.8 Hz)[4] [1][2][3][4] |
| 体内研究 (In Vivo) |
In Vivo Staining of Neurons and Glia Using Multicell Bolus Loading [1]
这种方法允许在直径约200µm的球形体积内染色神经元和胶质细胞(见图2)。 3. 根据《使用多细胞丸加载的体内双光子钙成像》(Garaschuk and Konnerth 2010)对小鼠大脑进行手术 4. 使用Ultrafree-MC离心过滤器过滤新制备的移液染色液。 5. 用染色溶液填充类似于膜片移液器的移液管(填充的移液管的阻力为3-6 MΩ),并使用LN-Mini机械手将其放置在脑组织内所需深度的光学控制下。使用成像系统连续监测移液器位置。 6. 使用短暂(仅500毫秒)的弹射脉冲(弹射压力34.5千帕)将MQAE压射到大脑中。以1-3分钟的脉冲间隔重复喷射2 - 4次。 7. 在最后一次喷射脉冲后10分钟检查获得的染色质量。 这个短暂的等待时间是允许MQAE从细胞外空间冲洗出来所必需的(很可能是因为微循环)。与膜渗透钙指示染料相比,MQAE在细胞内不进行脱酯化;因此,无需额外的等待时间。 MQAE染色细胞的双光子成像 8. 进行双光子成像 在单光子激发下,MQAE的激发波长为320-400 nm,最大发射波长为460 nm (Verkman et al. 1989)。通过双光子成像,MQAE在~ 740-770 nm处被有效激发。也可以在更长的波长(高达800纳米)激发MQAE,但发射光的强度较低(Marandi et al. 2002)。利用我们的成像系统,在960 ~ 990 nm的激发波长处无法激发MQAE。 MQAE的胞内校正 用Cl -淬灭喹啉基Cl -指示剂的效率取决于溶剂的粘度和/或极性(Jayaraman和Verkman 2000),因此,与试管试验相比,在细胞内可能不同。Krapf et al.引入的校准方案可用于校准切片神经元中的Cl -水平(Krapf et al. 1988;Marandi et al. 2002). 9. 制备含有不同量Cl−的体外校准溶液(例如,0、10、20、30和40 mM)。分别加入三丁基氯化锡(10µM)和尼日利亚菌素(10µM) 这种处理将破坏跨细胞膜的Cl -梯度,并将确定细胞质Cl -浓度([Cl -]i)等于相应的校准溶液的浓度。 10. 依次应用体外校准液,测量细胞内稳态荧光水平。无Cl−溶液中的平均荧光水平定义为F0。将每个校准溶液的F值绘制为F0/F相对于相应的[Cl−]i(所谓的Stern-Volmer图)。回归线的斜率(Stern-Volmer常数KSV)是表观解离常数Kd的倒数。 在我们的校准实验中,MQAE在试管内的Kd为13 mM,在体外脑片神经元内的Kd为40 mM (KSV = 24.7 M-1) (Marandi et al. 2002)。在其他组织中,KSV值在3 - 26 M-1之间变化(Lau等,1994;Bevensee et al. 1997;Maglova et al. 1998;Eberhardson et al. 2000;Gilbert et al. 2007;Hille et al. 2009)。 故障排除 问题(步骤2和7):观察到染色不良。 解决方案:MQAE染色本身比较简单可靠。然而,它需要高质量的切片/体内制备。考虑以下内容: 1. 为了获得高质量的体内制剂,请遵循使用多细胞丸加载的体内双光子钙成像(Garaschuk and Konnerth 2010)中描述的建议。 2. 我们不建议一次交付大量的MQAE。看起来MQAE从细胞外间隙被冲洗的效果不如膜渗透钙指示染料,大量染料的积累导致模糊(低对比度-高亮度)染色。 1. 小鼠海马体内[Cl⁻]i双光子成像(文献[1]): - 动物:雄性C57BL/6小鼠(8-10周龄)。 - 给药:MQAE溶解于0.9%生理盐水(50 mg/kg,单次腹腔注射,给药体积10 μL/g体重)。 - 检测:给药30分钟后,小鼠用1.5%异氟烷(100%氧气载气)麻醉;通过开颅术暴露海马CA1区,双光子显微镜(激发光720 nm,发射光460±20 nm)成像。MQAE可穿透神经元,静息状态[Cl⁻]i定量为~25±5 mM。未观察到异常行为(共济失调/嗜睡)或TUNEL阳性神经元(凋亡)[1] |
| 细胞实验 |
本方案描述了一种使用基于喹啉的氯化物(Cl−)指示染料MQAE (N-[6-甲氧基喹啉]乙酰乙酯)对活细胞进行高分辨率氯化物成像的技术。应用于急性脑切片,MQAE提供了神经元细胞及其过程的高质量标记。在活麻醉小鼠中,使用多细胞丸加载程序标记皮质细胞。结合双光子显微镜,该方法可以在体内可视化神经元和星形胶质细胞的细胞体以及一些星形胶质细胞的过程,并允许人们监测几十个单个细胞内细胞内氯化物浓度的变化。[1]
材料 你们务必查阅相应的物质安全数据表和你们机构的环境健康与安全办公室,以正确处理本方案中使用的设备和危险材料 试剂 体外定标液 MQAE (体外染色) MQAE试管校准溶液 尼日利亚素(K+/H+离子载体) 移液染色液,新鲜配制(用于体内染色) 标本 培养小鼠细胞或脑切片(用于体外染色) 所需菌株小鼠(用于体内染色) 小鼠标准外用生理盐水(体外染色用) 手术和麻醉试剂,如《使用多细胞丸加载的体内双光子钙成像》(Garaschuk和Konnerth 2010)(用于体内染色)中所述。 氯化三丁基锡(Cl−/OH−反转运物) 设备 玻璃毛细血管(用于体内染色) 图像的设置 任何市售的双光子成像系统都可以使用。这样的系统可以从几个供应商处获得。我们目前使用的是一个定制的装置,该装置基于一个锁模Ti:蓝宝石激光器,具有自动色散补偿和一个激光扫描系统,耦合到一个垂直显微镜,并配备了一个60×, 1.0数值孔径(NA)的水浸物镜(Fluor 60x;尼康)。这种定制系统可以按照Majewska等人(2000)和Nikolenko和Yuste(2005)所描述的说明进行组装。我们在750-770 nm处激发MQAE,在400 - 720 nm处采集荧光。然后使用ImageJ程序(http://rsb.info.nih.gov/ij/)和基于labview的软件包对获取的图像进行背景校正和离线分析。 培养箱,预设到37°C(用于体外染色) LN-Mini机械手(用于体内染色) 移液器(用于体内染色) 压力应用系统(用于体内染色) 带有中央通道开口的记录室:由标准组织培养皿(直径35毫米;Garaschuk et al. 2006)。 手术和麻醉设备,描述在体内双光子钙成像使用多细胞丸加载(Garaschuk和Konnerth 2010)(用于体内染色) Ultrafree-MC离心滤芯,孔径0.45µm(体内染色) 1. 原代海马神经元[Cl⁻]i成像实验(文献[1]): - 细胞分离与培养:解剖P0-P2小鼠海马,用0.25%胰蛋白酶(37℃,15分钟)消化,吹打成单细胞,接种于多聚赖氨酸包被的盖玻片。用含B27添加剂的神经基础培养基(37℃,5% CO₂)培养7-10天。 - MQAE负载:培养基替换为含5-10 mM MQAE的HEPES缓冲盐水(HBS:140 mM NaCl、5 mM KCl、2 mM CaCl₂、10 mM HEPES,pH 7.4),37℃孵育30分钟。盖玻片用HBS洗涤3次,去除细胞外游离MQAE。 - 成像与分析:双光子显微镜(激发光720 nm,发射光460±20 nm)每2分钟捕获荧光图像。[Cl⁻]i通过标准曲线(5-60 mM Cl⁻的HBS溶液,R²=0.95)计算[1] 2. 肾上皮细胞氯离子外流实验(文献[2]): - 细胞培养:大鼠肾近端小管上皮细胞用DMEM/F12 + 10% FBS培养,传代至24孔板(1×10⁵个/孔),过夜培养。 - MQAE负载:细胞用PBS洗涤后,在Krebs-Ringer缓冲液(115 mM NaCl、5 mM KCl、2 mM CaCl₂、1 mM MgSO₄、25 mM NaHCO₃、10 mM葡萄糖,pH 7.4)中加入5 mM MQAE,37℃孵育45分钟。 - 荧光检测:洗涤后加入10 μM呋塞米,酶标仪(激发光360 nm,发射光460 nm)每5分钟测量一次荧光强度,持续30分钟。氯离子外流以荧光强度较基线的升高百分比量化[2] 3. 主动脉平滑肌细胞[Cl⁻]i测量实验(文献[3]): - 细胞分离与培养:解剖大鼠主动脉,去除外膜,中膜用胶原酶(0.1%)消化30分钟。平滑肌细胞接种于DMEM + 10% FBS,培养至70%融合度后,接种于96孔板(5×10³个/孔)。 - MQAE负载:细胞在含5 mM MQAE的HBS(pH 7.4)中37℃孵育60分钟,用HBS洗涤3次以消除背景荧光。 - 分析:荧光分光光度计(激发光380 nm,发射光460 nm)测量荧光强度。[Cl⁻]i通过公式计算:F = F₀ / (1 + Kd/[Cl⁻]i)(F:样品荧光,F₀:0 mM Cl⁻时荧光,Kd=140 mM)[3] 4. 气道纤毛细胞体积无关[Cl⁻]i实验(文献[4]): - 细胞分离:解剖小鼠气管,用蛋白酶(0.2%)消化20分钟分离上皮层,纤毛细胞接种于胶原包被的盖玻片(培养24小时)。 - MQAE负载:细胞在含5 mM MQAE和25 mM甘露醇(稳定渗透压)的HBS中37℃孵育30分钟。 - 双光子成像:洗涤后,用低渗(250 mOsm)或高渗(350 mOsm)缓冲液诱导细胞体积±15%变化,记录荧光(激发光730 nm,发射光460 nm)。通过与氯离子电极数据的相关性(R²=0.92)验证[Cl⁻]i检测准确性[4] [1][2][3][4] |
| 动物实验 |
雌性小鼠(C57BL/6J,5周龄)自由摄取标准颗粒饲料和水。气道纤毛细胞的分离方法如前所述。简而言之,小鼠先用3%异氟烷(吸入)麻醉,然后腹腔注射戊巴比妥钠(40 mg/kg)和肝素(1000 U/kg)15分钟,进一步麻醉。之后,用高剂量戊巴比妥钠(100 mg/kg,腹腔注射)处死小鼠,并用弹性蛋白酶处理分离气道纤毛细胞。[4]
\nMQAE荧光强度测定[4] \nMQAE溶解于乙腈和水(1:1;储备液)中,储备液(500 mM)储存于−20 °C。将分离的气道纤毛细胞与 10 mM MQAE 在 37 °C 下孵育 60 分钟。5 mM 的 MQAE 常用于多种细胞类型的胞内标记,但在此浓度下,本研究中的气道纤毛细胞在 60 分钟内 MQAE 染色不足。因此,我们使用 10 mM 的 MQAE 以获得足够的染色,从而测量气道纤毛细胞中 MQAE 的荧光强度。同样浓度 (10 mM) 也用于测量 A6 细胞中的 [Cl−]i。将 MQAE 标记的细胞置于预先涂有 Cell-Tak 的盖玻片上,然后将盖玻片置于安装在倒置光学显微镜上的微灌注室 (20 µl) 中,该显微镜配备有共聚焦激光扫描系统。使用双光子激发激光系统在 780 nm 处激发 MQAE,并在 510 nm 处发射 MQAE。计算归一化荧光强度值 (Ft/F0);下标“0”或“t”分别表示施加渗透压应激开始前或开始后的时间。\n \n使用共聚焦激光扫描显微镜观察光学切片(厚度 0.9 μm)中的气道纤毛细胞。通过在每个光学切片的相差图像上描绘纤毛细胞的轮廓来测量细胞体积,并测量面积 (An µm2);“n”表示光学切片的数量。我们还测量了每个光学切片中细胞内区域的 MQAE 荧光强度 (Fn)。图像分析系统将 Fn 报告为每微米² 的强度。细胞体积 (V) 通过对每个切片中的 An 值求和来计算。我们还通过将每个切片中的总荧光强度 (An × Fn) 相加来计算所有细胞区域的总 MQAE 荧光强度。如果MQAE分子数量不变,则所有细胞区域的总MQAE荧光强度指示[Cl⁻]i。我们从每个细胞中获得了18-22个光学切片。细胞体积的归一化值(Vt/V0)也使用An的总和计算,其中下标“0”或“t”分别表示施加渗透压应激之前或之后的时间。我们还使用整个实验过程中相同的焦平面测量了所选细胞选定局部区域的MQAE荧光强度变化(Ft/F0);下标“0”或“t”分别表示施加渗透压应激之前或之后的时间。 \n1. 小鼠海马[Cl⁻]i体内成像(文献[1]):\n - 动物:雄性C57BL/6小鼠(8-10周龄),在实验室适应5天(12小时光照/黑暗循环,自由摄食/饮水)。 - MQAE 制备:用无菌 0.9% 生理盐水溶解至 50 mg/mL 的浓度(超声处理 5 分钟以确保完全溶解)。- 给药:单次腹腔注射(10 μL/g 体重)50 mg/kg MQAE;对照组注射等体积的生理盐水。- 成像设置:注射后 30 分钟,用 1.5% 异氟烷(以 1 L/min 的氧气为载气)麻醉小鼠,并将头部固定于立体定位仪上。在海马 CA1 区上方(坐标:前囟后方 -2.0 mm,侧方 +1.5 mm)做一个直径 1 mm 的小开颅术。 \n - 检测:使用双光子显微镜(20×物镜,NA 0.95)采集荧光图像(激发波长720 nm,发射波长460±20 nm)。[Cl⁻]i采用体外标准曲线(5-60 mM Cl⁻,R²=0.95)进行定量。成像后,处死小鼠,检查脑组织,确认无出血或神经元损伤。[1] |
| 毒性/毒理 (Toxicokinetics/TK) |
1. 体外毒性: - 细胞活力:用 5-10 mM MQAE 处理 0.5-2 小时的原代海马神经元、HEK293 细胞、肾上皮细胞和主动脉平滑肌细胞的活力 >90%(台盼蓝排除试验或 MTT 试验);无形态学变化(细胞收缩、膜起泡或脱落)[1]
[2][3] - 气道纤毛功能:小鼠气道纤毛细胞经 5 mM MQAE 处理 30 分钟后,纤毛摆动频率无变化(~12 Hz vs. 对照组 ~11.8 Hz,通过高速显微镜测量),表明无功能损伤[4] 2. 体内毒性:- 小鼠腹腔注射 50 mg/kg MQAE 后 24 小时内未出现异常行为(共济失调、嗜睡或食物摄入减少)。海马组织切片(成像后)未见 TUNEL 阳性神经元(凋亡)或胶质细胞活化(GFAP 免疫染色:无上调)[1] |
| 参考文献 |
|
| 其他信息 |
氯离子通道对细胞的重要性已由多种严重的人类疾病所证实,这些疾病均由氯离子通道基因突变引起。其中最著名的疾病是囊性纤维化。研究该疾病的机制和可能的治疗方法需要对单个细胞水平的氯离子流进行研究。本研究比较了两种氯离子转运的研究方法:X射线微分析和MQAE荧光图像分析。实验系统选择培养的呼吸道上皮细胞中的cAMP激活氯离子通道。两种方法均显示,用cAMP升高剂福斯克林和IBMX刺激可使细胞内氯离子含量降低约20-27%。用硝酸盐替代细胞外氯离子以诱导氯离子外流,结果显示,在福斯克林和IBMX存在的情况下,氯离子外流显著增加。本研究表明,X射线微分析和MQAE荧光是测量单个细胞中cAMP依赖性氯离子转运的充分且可比的方法。[2]
一种新型荧光指示剂N-[乙氧羰基甲基]-6-甲氧基喹啉溴化物(MQAE)被用于测量大鼠主动脉平滑肌细胞(VSMC)原代培养物中的细胞内氯离子浓度([Cl-]i)。对该染料的水解和荧光特性进行了表征。计算得出细胞内Stern-Volmer常数为25 M-1。Cl-外排曲线具有饱和型动力学特征,表观米氏常数为11 ± 4.8 (SD) mM,最大流速为0.038 ± 0.021 mM/s,半衰期(t1/2)为9.0 ± 3.7 min。在130 μM 4,4'-二异硫氰酸根-二氢芪-2,2'-二磺酸 (H2DIDS) (0.014 ± 0.006,P = 0.02) 或40 μM呋塞米 (0.017 ± 0.004,P = 0.04) 存在下,前10分钟的平均外排速率 (0.023 ± 0.004 mM/s) 均降低。在氯离子浓度降至零后,恢复生理细胞外氯离子浓度 ([Cl-]o) 导致净氯离子内流,半衰期为3.6 ± 1.0分钟。呋塞米处理后,初始Cl⁻内流速率从0.069 ± 0.006 mM/s降低至0.046 ± 0.008 mM/s(P < 0.002);H₂DIDS处理后,初始Cl⁻内流速率从0.102 ± 0.013 mM/s降低至0.033 ± 0.003 mM/s(P < 0.001)。呋塞米使稳态[Cl⁻]i从31.6 ± 3.2 mM降低至26.1 ± 2.4 mM(P < 0.01),而H₂DIDS对[Cl⁻]i的影响很小。我们的结果表明,MQAE可用于测量原代培养的VSMC中的[Cl⁻]i。[3]优点和局限性[1] MQAE可对体外和体内神经元进行简便快速的染色,并在细胞体中获得令人满意的荧光强度。在脑切片中,该方法还可以对神经元树突进行成像,而在活体中,只能清晰地分辨出胶质细胞突起。这种差异很可能是由于活体条件下染料从细胞外空间缓慢或不完全清除所致。 与其他Cl⁻指示剂相比,MQAE的优势包括对Cl⁻具有相对较高的灵敏度和选择性,对碳酸氢根浓度和pH值的变化不敏感,以及在使用双光子激发时可以进行长时间连续测量。此外,值得一提的是,MQAE会被Cl⁻快速猝灭(<1毫秒;Verkman等人,1989),因此非常适合监测[Cl⁻]i的生理变化,这些变化通常发生在毫秒到秒的范围内。而且,MQAE的猝灭机制是碰撞猝灭,不涉及Cl⁻与指示染料分子的结合(Verkman,1990)。因此,MQAE 不缓冲 Cl⁻,细胞内染料浓度的增加可以提高信噪比,而不会干扰 Cl⁻ 瞬态的时间进程。此外,在使用荧光寿命成像时,MQAE 成为一种比率型染料,可以进行 Cl⁻ 的定量测量。 MQAE 的主要局限性在于细胞内染料的泄漏损失。泄漏率似乎与制备方法有关,从脂质体中的 3%/h(Verkman 等,1989)到脑切片中的 30%/h(Marandi 等,2002)不等。正如亲脂性化合物的预期,其泄漏率与温度相关。因此,染料在体内的泄漏非常显著。这使得体内Cl⁻测量的持续时间限制在染色后约2小时。MQAE是一种“非比率型”氯离子(Cl⁻)猝灭荧光指示剂,用于测定细胞内Cl⁻浓度([Cl⁻]i)。据报道,基于MQAE的双光子显微镜是测量[Cl⁻]i的有效方法,但仍存在争议,因为细胞体积的变化可能会改变MQAE浓度,从而导致荧光强度发生变化,而[Cl⁻]i却没有变化。为了阐明细胞体积对MQAE浓度的影响(或无影响),我们研究了通过施加不同程度的渗透压应激而引起的细胞体积变化对气道纤毛细胞中MQAE荧光强度的影响。为了单独研究细胞体积变化对MQAE荧光强度的影响,即排除细胞内氯离子浓度[Cl⁻]i变化的影响,我们首先在不含氯离子的硝酸根(NO₃⁻)溶液中进行实验,以用NO₃⁻(一种不会猝灭MQAE荧光的阴离子)替代细胞内液中的氯离子。低渗(-30 mM NaNO₃)或高渗(+30 mM NaNO₃)均会引起细胞体积的变化,但并未导致MQAE荧光强度发生显著变化。我们也在含氯溶液中进行了实验。低渗(-30 mM NaCl)会增加MQAE荧光强度和细胞体积,而高渗(+30 mM NaCl)则会降低这两项指标。这些结果表明,渗透压应激引起的MQAE荧光强度变化是由细胞内氯离子浓度[Cl⁻]i的变化引起的,而非MQAE浓度的变化。此外,MQAE 的细胞内分布呈异质性,且不受渗透压应激诱导的细胞体积变化的影响,这表明 MQAE 与未知的亚细胞结构结合。这些结合的 MQAE 似乎使得即使在细胞体积变化的情况下,也能测量气道纤毛细胞中的 [Cl⁻]i。[4] 1. 作用机制:MQAE 是一种基于喹啉的荧光探针,它通过带正电荷的喹啉部分与细胞内 [Cl⁻]i 发生静电相互作用。结合的 MQAE 会发生荧光猝灭(荧光强度与 [Cl⁻]i 浓度呈负相关):[Cl⁻]i 越高,荧光越低,反之亦然。其 Kd 值(~140 mM)与生理 [Cl⁻]i 范围(10-60 mM)相匹配,从而能够在生物系统中进行精确定量。[3] 2.技术优势: - 双光子兼容性:MQAE 可被双光子显微镜(720-730 nm)激发,从而降低光毒性(对长期细胞成像至关重要)并提高组织穿透深度(高达 300 μm),使其适用于体内成像(例如,小鼠海马)和较厚的离体样本[1] [4] - 离子选择性:在生理浓度下,MQAE 与 Na⁺、K⁺、Ca²⁺ 或 Mg²⁺ 无显著结合(当这些离子浓度调整 ±20% 时,荧光变化 <5%),确保特异性检测 [Cl⁻]i[1] [3] 3.应用场景: - 神经科学:研究神经元中的氯离子稳态(例如,依赖于细胞内氯离子浓度梯度的GABA能信号传导)[1] - 肾脏生理学:研究肾小管中的氯离子转运(例如,呋塞米敏感的氯离子外流)[2] - 血管生物学:分析血管收缩过程中平滑肌细胞内氯离子浓度的变化(例如,去甲肾上腺素诱导的反应)[3] - 呼吸生理学:定量气道纤毛细胞中的氯离子浓度以研究黏液纤毛清除[4] [1][2][3][4] |
| 分子式 |
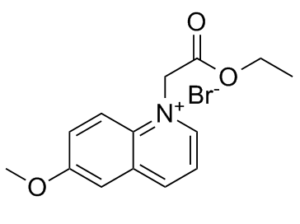
C14H16BRNO3
|
|
|---|---|---|
| 分子量 |
326.19
|
|
| 精确质量 |
325.031
|
|
| 元素分析 |
C, 51.55; H, 4.94; Br, 24.50; N, 4.29; O, 14.71
|
|
| CAS号 |
162558-52-3
|
|
| 相关CAS号 |
|
|
| PubChem CID |
2762651
|
|
| 外观&性状 |
Light yellow to khaki solid powder
|
|
| 熔点 |
177-179ºC(lit.)
|
|
| tPSA |
39.41
|
|
| 氢键供体(HBD)数目 |
0
|
|
| 氢键受体(HBA)数目 |
4
|
|
| 可旋转键数目(RBC) |
5
|
|
| 重原子数目 |
19
|
|
| 分子复杂度/Complexity |
282
|
|
| 定义原子立体中心数目 |
0
|
|
| SMILES |
CCOC(=O)C[N+]1=CC=CC2=C1C=CC(=C2)OC.[Br-]
|
|
| InChi Key |
DSLLHVISNOIYHR-UHFFFAOYSA-M
|
|
| InChi Code |
InChI=1S/C14H16NO3.BrH/c1-3-18-14(16)10-15-8-4-5-11-9-12(17-2)6-7-13(11)15;/h4-9H,3,10H2,1-2H3;1H/q+1;/p-1
|
|
| 化学名 |
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 请将本产品存放在密封且受保护的环境中(例如氮气保护),避免吸湿/受潮和光照。 |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 2.08 mg/mL (6.38 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 悬浮液;超声助溶。
例如,若需制备1 mL的工作液,可将100 μL 20.8 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.08 mg/mL (6.38 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: 100 mg/mL (306.57 mM) in PBS (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶 (<60°C). 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0657 mL | 15.3285 mL | 30.6570 mL | |
| 5 mM | 0.6131 mL | 3.0657 mL | 6.1314 mL | |
| 10 mM | 0.3066 mL | 1.5328 mL | 3.0657 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT02254551 | Terminated Has Results | Drug: LDE225 Drug: Bortezomib |
Multiple Myeloma | SCRI Development Innovations, LLC | January 2015 | Phase 2 |
| NCT04066504 | Active, not recruiting | Drug: sonidegib | Basal Cell Carcinoma | Sun Pharmaceutical Industries Limited | March 11, 2019 | |
| NCT02086513 | Terminated | Drug: LDE225 | Graft Versus Host Disease | Massachusetts General Hospital | April 2014 | Phase 1 |
| NCT04007744 | Recruiting | Biological: Pembrolizumab Drug: Sonidegib |
Clinical Stage III Cutaneous Melanoma AJCC v8 Clinical Stage III Gastric Cancer AJCC v8 |
Mayo Clinic | February 13, 2020 | Phase 1 |