| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| Other Sizes |
|
| 体外研究 (In Vitro) |
浓度为 100、300 和 1000 µM 的二甲嗪会强烈抑制 HepG2 细胞活力 [1]。二甲嗪(3、10、30 µM;24 h)促进 HepG2 细胞的能量代谢从有氧三羧酸循环(TCA)和氧化磷酸化转变为无氧糖酵解[1]。在 HepG2 细胞中,异丙嗪(3、10、30 µM;24 小时)可抑制复合物 IV 活性 [1]。
|
|---|---|
| 体内研究 (In Vivo) |
三氯芬酸(46.3、139、417 mg/kg;口服;单次)在肾脏(18.64%)、脑(23.58%)、胃(21.94%)和肝脏(35.84%)中蓄积[1]。异丙嗪(46.3、139、417 mg/kg;口服;单剂量)比任何其他器官都更能提高小鼠肝脏和大脑中的 MDA 水平[1]。
|
| 细胞实验 |
RT-PCR[1]
细胞类型: HepG2 细胞 测试浓度: 3、10、30 µM 孵育时间: 24 小时 实验结果: 30 µM 时,乳酸脱氢酶 B (LDHB) 水平显着增加,6-磷酸果糖-2-激酶/果糖-2,6-二磷酸酶略有增加3(PFKFB3)。在 3、10 和 30 µM 噻嗪酮处理下,ATP 水平以浓度依赖性方式分别降低至载体对照水平的 91.3%、87.9 和 67.2%。乳酸水平显着增加。 免疫荧光[1] 细胞类型: HepG2 细胞 测试浓度: 3、10、30 µM 孵育时间: 24 小时 实验结果: 在 3、10 和 30 ℃ 进行噻嗪酮处理后,复合物 IV 的活性显着抑制至载体对照水平的 82.2、69.2 和 63.4%分别为μM。 细胞活力测定[1] 细胞类型: HepG2 细胞 测试浓度: 3、10、30 µM <孵育时间: 24 小时 实验结果: 以不依赖浓缩物的方式显着提高细胞内 ROS 水平,并降低 mtDNA 含量。 |
| 动物实验 |
动物/疾病模型:雄性C57BL/6小鼠(6至8周龄)[1]。
剂量:46.3、139、417 mg/kg 给药途径:口服;单次 实验结果:所有器官的MDA水平均有升高趋势,其中肝脏和脑组织的MDA水平升高最为显著,且呈浓度依赖性。肝脏中MDA浓度最高(35.84%),其次是脑组织(23.58%)、胃(21.94%)和肾脏(18.64%),而小鼠脾脏和心脏中的MDA水平低于检测限。 |
| 药代性质 (ADME/PK) |
吸收、分布和排泄
(14)C-布洛芬嗪(放射化学纯度>97%)灌胃给予5只大鼠/每性别/每剂量,剂量分别为10和100 mg/kg;雄性大鼠48小时内消除90-91%的剂量(20-21%经尿液排出,69-71%经粪便排出);雌性大鼠48小时内消除87-89%的剂量(13-14%经尿液排出,73-76%经粪便排出);雄性大鼠在最初24小时内消除速度更快,但48小时后消除速度趋于一致;7天后体内残留剂量<1%;24小时后,雄性大鼠胆汁中回收剂量≥30%,雌性大鼠胆汁中回收剂量≥38%;尿液、胆汁和粪便的色谱分析表明存在广泛的结合反应;对 3M/3F 进行胆汁插管分析表明,粪便代谢物可能来源于胆汁。 将 [(14)C-苯基]-布洛芬嗪(2 或 22.5 mCi/mmol;放射化学纯度 >97%)悬浮于 1 mL 橄榄油中,通过灌胃法给予禁食的大鼠,剂量分别为 10 和 100 mg/kg(动物数量随实验而变化);两种浓度下,48 小时内均有超过 90% 的给药剂量排出;96 小时后,两种浓度下均有 70-74% 的剂量通过粪便排出(尽管在高剂量组相对于低剂量组在 24 小时内出现延迟),21-25% 通过尿液排出,极少量以呼出 (14)C-CO2 的形式排出;在 10 mg/kg 剂量下,12% 的母体化合物通过粪便排出;两种剂量下,血液中药物浓度峰值均在给药后 9 小时出现,之后呈双相下降(半衰期分别为 13 小时和 60 小时);放射性标记物在组织中的峰值水平出现在给药后 5-9 小时,之后组织水平呈双相下降,两个阶段的半衰期分别为 3.5-15 小时和 15-72 小时;96 小时后,组织残留水平较低。 雄性大鼠饲喂含 200 或 1000 ppm 布洛芬的饲料,持续长达 24 周;每剂量组分别于第 2 天和第 4 天以及第 1、2、4、8、12、16 和 24 周处死 3 只大鼠;从血液、脑、肝脏、肾脏、脂肪组织和肌肉中提取布洛芬,并通过气相色谱法测定其浓度;检测限:0.1 ppm; 200 ppm:仅脂肪组织达到足以持续检测的浓度,在 4 天至 24 周内保持稳定,平均浓度为 0.43-1.10 ppm;个别动物的肝脏中检测到浓度,而肾脏、肌肉和大脑中从未检测到浓度;1000 ppm:脂肪组织在第 4 天达到峰值 10.53 ppm,在 24 周时下降至 3.40 ppm;肝脏在整个期间保持稳定的浓度 0.21-0.96 ppm;肾脏在 8 周内接近或低于检测限,之后无法检测到;大脑在 1 周内接近或低于检测限,之后无法检测到;肌肉在 2 周内接近或低于检测限,之后无法检测到(4 周时未测量)。因此,在两种浓度下,测试物质均未在任何组织中积累。 代谢/代谢物 [(14)C-苯基]-布洛芬嗪(2 或 22.5 mCi/mmol;放射化学纯度 >97%)悬浮于 1 mL 橄榄油中,通过灌胃法给予禁食的 /大鼠/ 10 和 100 mg/kg(动物数量随实验而变化);代谢物研究揭示了苯环的羟基化、硫的氧化以及噻二嗪环的裂解,并有证据表明存在葡萄糖醛酸和硫酸结合。在反刍动物中:2-叔丁基亚氨基-5-(4-羟基苯基)-3-异丙基-1,3,5-噻二嗪-4-酮 (BF2) 是肝脏和肾脏中鉴定的主要残留物(脂肪和肌肉中的残留量≤0.020 ppm),而N-(4-羟基苯基)乙酰胺 (BF23) 是反刍动物代谢研究中牛奶中鉴定的主要残留物(所有其他残留物均<10%总放射性残留量 (TRR);3.5倍最大理论日粮负荷 (MTDB))。牛奶中需要关注的残留物是布洛芬嗪和BF23;反刍动物组织中需要关注的残留物是布洛芬嗪和BF2。 |
| 毒性/毒理 (Toxicokinetics/TK) |
毒性概述
鉴定和用途:噻嗪酮为固体。它用作杀虫剂(几丁质合成抑制剂)。人体暴露和毒性:在代谢活化或未活化的人类淋巴细胞培养物中,未发现与噻嗪酮相关的异常细胞或异常增加。动物研究:将大鼠皮肤每天用0、100、300或1000 mg/kg/天的噻嗪酮处理6小时,持续24天。1000 mg/kg剂量组的雌性大鼠出现局灶性肝细胞坏死的发生率增加。在大鼠的长期研究中,观察到6、12和24个月时甲状腺重量增加,12和24个月时肝脏重量增加。此外,高剂量组还观察到雌雄两性肝细胞坏死和增生性结节、雄性间质性肺炎、雌性间质性心脏水肿及其他心脏损害。发育研究发现,母体体重减轻和幼崽完全吸收。无论是否经过代谢活化,布洛芬净对五株鼠伤寒沙门氏菌测试菌株(TA98、TA100、TA1535、TA1537 和 TA1538)均无致突变性。生态毒性研究:布洛芬净急性暴露 48 小时后,水蚤在 EC50 为 0.44 mg/L 时出现活动障碍。在一项为期14天的布洛芬(0、0.025、0.05、0.10和0.15 mg/L)慢性暴露研究中,水蚤的发育和繁殖均受到显著影响,且体长比其他观察参数更为敏感。然而,布洛芬对亲代水蚤的不良影响会传递给其后代,且无法在短期内恢复。当非洲鲶(Clarias gariepinus)的胚胎和幼体暴露于浓度超过5 mg/L的布洛芬时,观察到畸形。 相互作用 ……在本研究中,我们评估了重金属镍(NiSO4)和昆虫生长调节剂布洛芬的联合作用对斑马鱼胚胎毒性的诱导作用。通过使用Hill和Langmuir函数对每种化学物质的浓度-效应数据进行非线性回归,并利用浓度加和(CA)模型计算预测值,我们证实了NiSO4和噻嗪酮共同作用对斑马鱼胚胎具有协同毒性。随后,我们进一步发现NiSO4和噻嗪酮形成复合物,促进胚胎对镍(Ni)和噻嗪酮的吸收。随后,我们阐明了该复合物的氧化机制可能是NiSO4和丁丙诺啡协同胚胎毒性的潜在机制。 非人类毒性值 小鼠经口LD50 >5 g/kg 大鼠经皮LD50 >5 g/kg 雄性大鼠经口LD50 2198 mg/kg 雌性大鼠经口LD50 2355 mg/kg 大鼠吸入LC50 >2.2 mg/L (4 小时) /Applaud 70 DF/ |
| 参考文献 |
|
| 其他信息 |
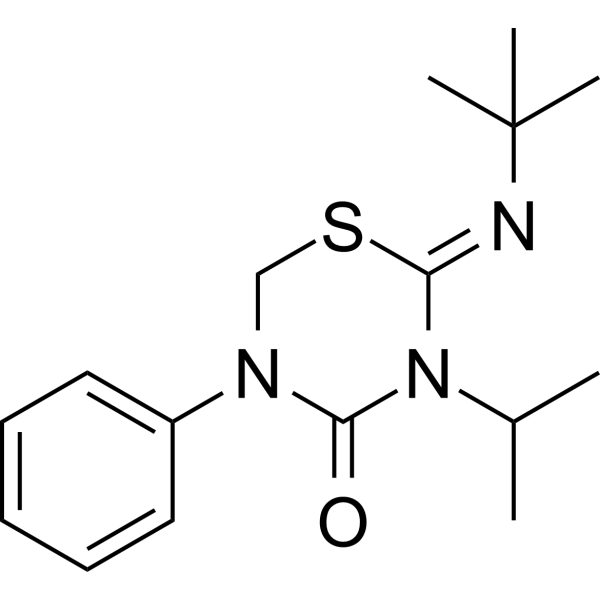
噻嗪酮是一种2-(叔丁基亚氨基)-5-苯基-3-(丙-2-基)-1,3,5-噻二嗪烷-4-酮,其中C=N双键为Z构型。它是一种杀虫剂,也是同翅目昆虫几丁质生物合成抑制剂之一。
|
| 分子式 |
C16H23N3OS
|
|---|---|
| 分子量 |
305.44
|
| 精确质量 |
305.156
|
| CAS号 |
69327-76-0
|
| 相关CAS号 |
Buprofezin-d6;2140803-94-5
|
| PubChem CID |
50367
|
| 外观&性状 |
White to off-white solid powder
|
| 密度 |
1.18
|
| 沸点 |
273°C (12 torr)
|
| 熔点 |
104-106°C
|
| 闪点 |
176-178°C
|
| 折射率 |
1.52-1.522
|
| LogP |
4.185
|
| tPSA |
61.21
|
| 氢键供体(HBD)数目 |
0
|
| 氢键受体(HBA)数目 |
3
|
| 可旋转键数目(RBC) |
3
|
| 重原子数目 |
21
|
| 分子复杂度/Complexity |
408
|
| 定义原子立体中心数目 |
0
|
| SMILES |
CC(C)N1C(=NC(C)(C)C)SCN(C1=O)C2=CC=CC=C2
|
| InChi Key |
PRLVTUNWOQKEAI-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C16H23N3OS/c1-12(2)19-14(17-16(3,4)5)21-11-18(15(19)20)13-9-7-6-8-10-13/h6-10,12H,11H2,1-5H3
|
| 化学名 |
2-tert-butylimino-5-phenyl-3-propan-2-yl-1,3,5-thiadiazinan-4-one
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO: 100 mg/mL (327.40 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (8.18 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (8.18 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.5 mg/mL (8.18 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2740 mL | 16.3698 mL | 32.7397 mL | |
| 5 mM | 0.6548 mL | 3.2740 mL | 6.5479 mL | |
| 10 mM | 0.3274 mL | 1.6370 mL | 3.2740 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。