| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| Other Sizes |
|
| 靶点 |
Antineoplastic and anti-vasculogenic mimicry
|
|---|---|
| 体外研究 (In Vitro) |
采用NCI60筛选法检测CVM-1118的抗癌活性。结果表明,CVM-1118在平均GI50值<100 nM的细胞株中,约87%的细胞株具有抑制癌细胞生长的作用。还使用NCI的GI50结果进行了比较分析。有趣的是,它与NCI60筛选数据库中出现的任何标准抗癌药物都没有密切的相关性,这表明CVM-1118在癌细胞中的细胞毒性作用可能涉及一种新的作用机制。[2]
先前对2-苯基喹啉-4-酮(2- pqs)的探索已经产生了一种抗癌候选药物2-(2-氟苯基)-6,7-亚甲基二氧喹啉-4-一磷酸一钠(CHM-1-P-Na)。为了开发新的候选药物,设计、合成了新的2-PQs,并对其细胞毒性活性进行了评估。大多数类似物,包括1b、2a、b、3a、b、4a、b和5a、b,对所有测试的肿瘤细胞系均表现出显著的抑制活性(IC(50)为0.03 ~ 8.2 μM)。作为最有效的类似物之一,2-(3-氟苯基)-5-羟基-6-甲氧基喹啉-4- 1 (3b)在美国国家癌症研究所(NCI)的评估中选择性地抑制了60种癌细胞系中的14种。初步作用机制研究提示3b对胰岛素样生长因子-1受体(IGF-1R)酪氨酸自磷酸化有显著影响。3b的安全药理学分析显示,对大多数酶的正常生物学功能没有显著影响。此外,3b的单磷酸盐2-(3-氟苯基)-6-甲氧基-4-氧-1,4-二氢喹啉-5-酰基磷酸钠(15)在Hep3B异种移植裸鼠模型中超过了阿霉素的活性,与CHM-1-P-Na相当。综上所述,15是一个很有前景的临床候选药物,目前正在进行临床前研究。[1] |
| 体内研究 (In Vivo) |
Foslinanib是一种口服生物利用药物,具有潜在的抗肿瘤和抗血管生成模拟活性。口服后,foslinanib靶向并抑制血管源性拟态(VM;血管拟态)。通过破坏VM通道和网络,使癌细胞缺乏灌注,从而诱导癌细胞凋亡,抑制癌细胞增殖。VM与肿瘤转移有关。[3]
|
| 细胞实验 |
CVM-1118对肿瘤细胞增殖、存活率和凋亡率的影响[2]
用流式细胞术分析了CVM-1118对未处理(对照)或1、10、50和100 nM CVM-1118处理的人黑色素瘤细胞的增殖、存活率、凋亡和细胞碎片(代表死细胞)的影响使用Guava viaccount和Nexin试剂。将黑色素瘤细胞(1 × 105)用不同浓度的CVM-1118处理24、48和72小时,随后与未处理的细胞进行三次重复,测定其增殖、存活率和凋亡率。如图2所示的代表性结果显示,用50和100 nM的CVM-1118处理24小时后细胞的增殖显著(p<0.05)降低,用10、50和100 nM处理48和72小时后细胞的增殖显著(p<0.05)降低。虽然CVM-1118在24、48和72小时后,在10、50和100 nM处显著降低细胞活力,但所有浓度的CVM-1118(包括1 nM)在24小时后,在48和72小时后,在50和100 nM的CVM-1118处理的细胞中,凋亡百分比都增加了。 CVM-1118对血管拟态的抑制作用[2] 为了评估CVM-1118对体外VM的影响,在12孔培养皿中使用Matrigel 制备的三维基质进行标准VM测定。将人黑色素瘤细胞(1 × 105个细胞/孔)不加(对照)或加CVM-1118,浓度分别为1、10或50 nM,分别镀在基质上。24小时后观察VM管状网络的形成,使用10倍物镜(100倍终放大)的倒置显微镜和日立HV-C20 CCD相机 进行数字图像捕获。然后使用AngioSys软件包分析来自对照和处理培养的四个不同领域的图像,评估每个领域的连接和小管数量以及总小管长度,并使用Excel确定平均值,标准误差和显著性。图3显示了对照组和每个CVM-1118处理组的代表性图像,以及为后续分析生成的结果二值图像。数据显示,与对照样品相比,用1,10和50 nM的CVM-1118处理的样品中,结数和平均总管长(每个观察场)在剂量依赖性,统计学上显着减少。虽然CVM-1118抑制VM的能力在图像中清晰可见,但详细的分析支持这样的概念,即这种抑制是剂量依赖性的,并且可能与细胞形成VM特征分支管状网络的能力的破坏有关。 CVM-1118影响的通路[2] 在用CVM-1118治疗人类黑色素瘤细胞后,对上述和图1中突出显示的VM的关键信号通路进行了进一步研究。结合qRT-PCR分析和蛋白质阵列,测量了人类磷酸化mapk /磷酸化激酶和细胞应激检查点以及凋亡调节因子的变化。图4显示了响应CVM-1118处理产生的主要结果的汇编,显示为级联和重叠的信号通路。在mRNA水平上,CVM-1118对人类黑色素瘤细胞的主要总体影响是下调干细胞相关基因Nodal(及下游pSMAD2)、Notch4 ICD和HES1,以及血管信号相关基因VEGF-A(见表1),从而抑制VM。对CVM-1118在蛋白质水平上的处理效果的进一步分析显示,(磷酸化的,指定为p-) p- hif -1α, p-p27和p-c- jun的显著增加,共同证明了缺氧反应,细胞周期阻滞,生长抑制和凋亡事件背后的蛋白质稳定。 |
| 参考文献 |
|
| 其他信息 |
Forslini disodium is the disodium salt form of forslini, a highly bioavailable oral drug with potential antitumor and anti-angiogenic mimicry activities. After oral administration, forslini targets and inhibits the formation of angiogenic mimics (VMs). By disrupting VM channels and networks, cancer cells lose perfusion, thereby inducing apoptosis and inhibiting cancer cell proliferation. VMs are associated with tumor metastasis. Forslini is currently being investigated in the clinical trial NCT03600233 (CVM-1118 study for the treatment of patients with advanced neuroendocrine tumors). In 1999, the American Journal of Pathology published an article by Maniotis et al. entitled "Vascular channel formation by human melanoma cells in vivo and in vitro: vasculogenic mimicry," which sparked years of heated discussion and was hailed by the journal as a "classic cited article" (Maniotis et al., 1999). Tumor cell angiogenic mimicry (VM), also known as vascular mimicry, describes the plasticity of invasive cancer cells in forming new blood vessel networks and is associated with malignant phenotypes and poor clinical prognosis. Tumor cells capable of VM share common characteristics of a stem cell-like transendothelial phenotype, which may be induced by hypoxia. Since VM was proposed as a novel paradigm for melanoma tumor perfusion, numerous studies have revealed potential molecular pathways supporting VM in various tumors, including carcinoma, sarcoma, glioblastoma, astrocytoma, and melanoma, and have yielded new discoveries. Of particular note is that angiogenesis inhibitors are ineffective against tumor cell VMs, suggesting that this phenotype is selectively resistant to conventional therapies. Key proteins that promote the functional plasticity of tumor cell VMs are closely associated with angiogenesis, stem cells, extracellular matrix and hypoxia-related signaling pathways—all of which deserve further investigation as potential therapeutic targets and diagnostic markers of aggressive metastatic phenotypes. This review highlights groundbreaking findings related to angiogenesis mimicry (VMs), including the role of a novel small molecule compound, CVM-1118 (currently in clinical development to target VMs), and elucidates key molecular pathways that inhibit this highly plastic and aggressive phenotype using melanoma as a model. [2] This review covers VMs in a variety of cancers, but highlights only key findings related to their functional and transformative significance, and their association with aggressive and metastatic phenotypes. The molecular pathways of VMs provide new candidate targets for developing innovative therapeutic strategies that target tumor cell plasticity and metastatic characteristics associated with disease recurrence and drug resistance. The presence of heterogeneous subpopulations in tumors and the complex and diverse vascular supply further increase the difficulty of effectively eradicating cancer. In addition, unintended consequences such as rapid tumor growth or hypoxia caused by certain conventional therapies may act as catalysts for VM and cancer stem cell phenotypes. Therefore, it seems wise and timely to consider using novel drugs (such as CVM-1118) to target VM pathways that are associated with stem cell phenotypes and resistant to most conventional drugs. Further research is needed to evaluate the efficacy of CVM-1118 in other aggressive cancers that express VM, so as to better understand its full potential. Combining specific molecular compounds that target VM with first-line therapies may be the most promising approach to fighting cancer. [2]
|
| 分子式 |
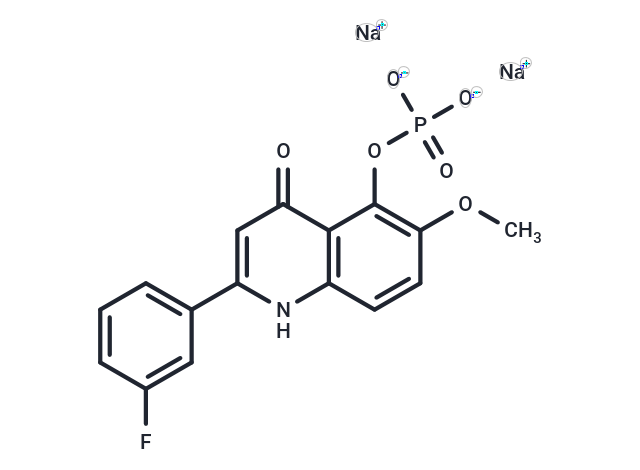
C16H11FNNA2O6P
|
|---|---|
| 分子量 |
409.21
|
| 精确质量 |
409.0103
|
| CAS号 |
1256037-62-3
|
| 相关CAS号 |
1256037-60-1;Foslinanib (CVM-1118);
|
| PubChem CID |
49840583
|
| 外观&性状 |
Typically exists as solid at room temperature
|
| LogP |
0
|
| tPSA |
111Ų
|
| 氢键供体(HBD)数目 |
1
|
| 氢键受体(HBA)数目 |
8
|
| 可旋转键数目(RBC) |
3
|
| 重原子数目 |
27
|
| 分子复杂度/Complexity |
582
|
| 定义原子立体中心数目 |
0
|
| SMILES |
COC1=C(C2=C(C=C1)NC(=CC2=O)C3=CC(=CC=C3)F)OP(=O)([O-])[O-].[Na+].[Na+]
|
| InChi Key |
TWMCXXQLLQDSTN-UHFFFAOYSA-L
|
| InChi Code |
InChI=1S/C16H13FNO6P.2Na/c1-23-14-6-5-11-15(16(14)24-25(20,21)22)13(19)8-12(18-11)9-3-2-4-10(17)7-9;;/h2-8H,1H3,(H,18,19)(H2,20,21,22);;/q;2*+1/p-2
|
| 化学名 |
disodium;[2-(3-fluorophenyl)-6-methoxy-4-oxo-1H-quinolin-5-yl] phosphate
|
| 别名 |
Foslinanib disodium; 1256037-62-3; TRX818 Sodium; CVM-1118 disodium; TRX-818 (disodium); 8X5F5LAF7B; UNII-8X5F5LAF7B; disodium;[2-(3-fluorophenyl)-6-methoxy-4-oxo-1H-quinolin-5-yl] phosphate;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO: ~7.86 mg/mL (~19.2 mM )
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4437 mL | 12.2187 mL | 24.4373 mL | |
| 5 mM | 0.4887 mL | 2.4437 mL | 4.8875 mL | |
| 10 mM | 0.2444 mL | 1.2219 mL | 2.4437 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。