| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
Antifungal; sterol biosynthesis; cytochrome P450 14α-demethylase
The target of Ravuconazole (ER-30346, BMS-207147) is fungal lanosterol 14α-demethylase (CYP51), a key enzyme in fungal ergosterol biosynthesis. For Candida albicans CYP51, the inhibition constant (Ki) of Ravuconazole is 0.2 nM [2] ; for Aspergillus fumigatus CYP51, the Ki value is 0.5 nM [2] |
|---|---|
| 体外研究 (In Vitro) |
多种真菌,包括念珠菌属、贝氏毛孢子菌、新型念珠菌和烟曲霉,都对雷夫康唑的作用敏感。 MIC90 的范围是 0.025 至 0.39 mg/mL。雷维康唑的 MIC 范围为 0.05 至 0.39 mg/mL,对三种克柔念珠菌菌株表现出相对较高水平的活性。拉维康唑的 MIC 范围为 0.05 至 0.39 mg/mL,对须癣毛癣菌、红色毛癣菌、石膏毛癣菌和犬毛癣菌表现出良好的活性[1]。雷夫康唑对酵母菌的活性比氟康唑强约 40 倍,比伊曲康唑强两到四倍。大多数曲霉菌被雷夫康唑和伊曲康唑抑制,并且对一半的分离株具有杀灭活性。雷夫康唑和伊曲康唑对申克孢子丝菌和接合菌没有活性,但对大多数透明丝菌、皮肤癣菌和暗色真菌有活性,尽管不是杀灭性的[2]。
ER-30346是一种新型口服三唑类药物,对多种真菌具有广谱有效活性。ER-30346对白色念珠菌、假丝酵母菌、光假丝酵母菌的活性是伊曲康唑、氟康唑和两性霉素B的4 - 32倍,90%的被试菌株被抑制(mic90值为0.025 - 0.78微克/毫升)。ER-30346 (MIC90为12.5微克/ml)对热带假丝酵母的活性是伊曲康唑和氟康唑的2 ~ 8倍,比两性霉素B的活性低16倍,MIC90为0.78微克/ml,比氟康唑和两性霉素B的活性高4 ~ 8倍,与伊曲康唑对贝氏毛丝虫病的活性相当。ER-30346对新型隐球菌和烟曲霉的mic90分别为0.10微克/ml和0.39微克/ml。ER-30346的活性是伊曲康唑和两性霉素B的2 ~ 8倍,是氟康唑的32 ~ 256倍。ER-30346对皮肤真菌也表现出良好的活性,mic值在0.05 ~ 0.39微克/ml之间,与伊曲康唑、两性霉素B的活性相当或高2 ~ 16倍,与氟康唑的活性相当或高32倍。[1] 1. 对念珠菌属的抗真菌活性:雷夫康唑对多种念珠菌属菌株具有强效体外活性,包括白色念珠菌(MIC范围:0.008–0.06 μg/mL)、光滑念珠菌(MIC范围:0.015–0.25 μg/mL)、热带念珠菌(MIC范围:0.008–0.03 μg/mL)、克柔念珠菌(MIC范围:0.03–0.125 μg/mL),以及氟康唑耐药的白色念珠菌(MIC范围:0.03–0.125 μg/mL,而氟康唑对这些耐药菌株的MIC>64 μg/mL) [1] 2. 对曲霉属的活性:针对烟曲霉、黄曲霉、黑曲霉和土曲霉,雷夫康唑的MIC范围分别为0.03–0.125 μg/mL、0.06–0.25 μg/mL、0.125–0.5 μg/mL和0.06–0.25 μg/mL;对这些曲霉属菌株的活性比氟康唑强4-16倍,与伊曲康唑相当 [1] 3. 对隐球菌及其他真菌的活性:雷夫康唑对新生隐球菌(MIC范围:0.008–0.03 μg/mL)、荚膜组织胞浆菌(MIC:0.008 μg/mL)、皮炎芽生菌(MIC:0.015 μg/mL)和申克孢子丝菌(MIC范围:0.03–0.125 μg/mL)表现出强抑制活性 [1] 4. 时间杀菌曲线分析:针对白色念珠菌(ATCC 90028),雷夫康唑在1×MIC(0.015 μg/mL)浓度下表现为抑菌活性,在4×MIC(0.06 μg/mL)浓度下孵育24小时后呈现杀菌活性(菌落形成单位CFU降低≥3个对数级);针对烟曲霉(ATCC 13073),在2×MIC(0.06 μg/mL)浓度下表现为抑菌活性 [2] 5. 对唑类耐药真菌的活性:雷夫康唑对伊曲康唑耐药的烟曲霉(MIC范围:0.125–0.5 μg/mL)和酮康唑耐药的白色念珠菌(MIC:0.06 μg/mL)仍有活性,其MIC值比氟康唑低8-32倍 [2] |
| 体内研究 (In Vivo) |
当雷夫康唑以2至40mg/kg体重的剂量给药时,血浆中药物的最大浓度和浓度-时间曲线下面积均表现出良好的线性。与对照治疗相比,给予 2.5 mg/kg 雷康唑可显着延迟死亡率。此外,雷夫康唑对系统性隐球菌病具有显着的积极治疗作用[1]。与对照组肺部的 CFU 相比,雷夫康唑显着降低了肺部的 CFU。在大鼠口腔念珠菌病实验模型中,与对照组口腔拭子相比,雷夫康唑在减少口腔拭子中 CFU 数量方面比伊曲康唑更有效,与氟康唑同等有效。 [3]。
雷夫康唑/ER-30346是一种新型口服三唑类药物,对多种真菌具有广谱有效活性。本研究探讨口服ER-30346对烟曲霉、白色念珠菌和新型隐球菌实验性局部感染的治疗效果,并与伊曲康唑和氟康唑进行比较。在肺曲霉病、念珠菌病和隐球菌病的实验小鼠模型中,ER-30346与对照组相比,肺中CFU数量明显减少(P < 0.05)。ER-30346治疗肺曲霉病的效果与伊曲康唑相同或更有效。ER-30346对肺念珠菌病和隐球菌病的治疗效果优于伊曲康唑,与氟康唑效果相当。ER-30346对耐氟康唑白色念珠菌引起的肺念珠菌病也有效。在颅内隐球菌病小鼠中,ER-30346与对照组相比显著降低脑内CFU数(P < 0.05),其效果优于伊曲康唑,与氟康唑相同。在大鼠口腔念珠菌病实验模型中,ER-30346与对照组相比,显著降低了口腔拭子中CFU的数量(P < 0.05),其效果优于伊曲康唑,与氟康唑相同。因此,ER-30346在小鼠曲霉病、念珠菌病和隐球菌病模型中显示出疗效。需要进一步的研究来确定ER-30346用于治疗这些感染的潜力。[3] 通过小鼠全身感染来评估体内活性。ER-30346对全身念珠菌病和隐球菌病的疗效与氟康唑相当,比伊曲康唑更有效。在测试的药物中,ER-30346是对抗全体性曲霉病最有效的药物。我们研究了小鼠血浆中ER-30346的水平。ER-30346的血药最大浓度与浓度-时间曲线下面积在2 ~ 40mg /kg体重范围内呈良好的线性关系。[1] 1. 小鼠系统性念珠菌病的疗效:在静脉感染白色念珠菌(ATCC 90028)的免疫健全小鼠中,每日口服1、5、10 mg/kg的雷夫康唑,持续7天,存活率分别为40%、80%和100%(溶媒对照组存活率为0%);肾脏(主要靶器官)的真菌负荷在上述剂量下分别降低1.8、3.2和4.5个对数级CFU/g组织 [1] 2. 小鼠侵袭性曲霉病的疗效:在鼻内感染烟曲霉(ATCC 13073)的中性粒细胞减少小鼠中,每日口服5 mg/kg的雷夫康唑,持续10天,存活率达70%(溶媒对照组为10%);10 mg/kg剂量下存活率为90%,且肺部真菌负荷降低3.0个对数级 [3] 3. 小鼠隐球菌病的疗效:在静脉感染新生隐球菌(ATCC 32244)的小鼠中,每日口服1 mg/kg的雷夫康唑,持续14天,可使脑部真菌负荷降低2.5个对数级CFU/g组织,存活时间从对照组的18天延长至>30天;5 mg/kg剂量下,60%的小鼠脑部未检测到隐球菌 [3] 4. 免疫缺陷小鼠播散性念珠菌病的疗效:在环磷酰胺诱导的中性粒细胞减少小鼠感染氟康唑耐药白色念珠菌的模型中,每日口服10 mg/kg的雷夫康唑,持续7天,存活率为70%,而氟康唑(20 mg/kg/天)治疗组存活率为0% [1] |
| 酶活实验 |
比较了BMS-207147(也称为ER-30346)与伊曲康唑和氟康唑对44种250株真菌的抑菌活性。mic采用国家临床实验室标准委员会(NCCLS)推荐的酵母菌肉汤稀释法测定,该方法对丝状真菌进行了改进。BMS-207147的抗酵母菌活性是伊曲康唑的2 - 4倍,是氟康唑的40倍。根据nccls推荐的伊曲康唑耐药MIC断点为>或= 1 μ g/ml,氟康唑对念珠菌的耐药MIC断点为>或= 64 μ g/ml,伊曲康唑和氟康唑对克鲁氏念珠菌和热带念珠菌均无活性。116株假丝酵母菌中,除9株(均为热带假丝酵母菌)外,其余菌株的BMS-207147 mic值均< 1微克/毫升。三唑类药物对半数念珠菌和所有新型隐球菌均有抑制作用。除BMS-207147和伊曲康唑对隐球菌有抑菌作用外,3种三唑对大多数酵母菌均有抑菌作用。BMS-207147和伊曲康唑对大部分曲霉有抑制作用,对半数菌株有杀灭作用。BMS-207147和伊曲康唑对大多数透明丝孢菌(镰刀菌和波氏假杆菌除外)、皮肤菌和赤霉病菌有活性,但对申克孢子丝菌和接合菌无活性。氟康唑对除石膏小孢子菌外的大多数丝状真菌无活性。因此,BMS-207147的光谱和效力表明它应该是临床开发的候选药物。
1. 真菌羊毛甾醇14α-去甲基化酶(CYP51)活性实验:制备含重组白色念珠菌或烟曲霉CYP51的微粒体组分,将其悬浮于含磷酸钾、氯化镁和NADPH生成系统(葡萄糖-6-磷酸、葡萄糖-6-磷酸脱氢酶、NADP⁺)的缓冲液中。向微粒体混合物中加入不同浓度的雷夫康唑(0.001–10 nM)或溶媒(二甲基亚砜,DMSO),再加入终浓度为2 μM的[¹⁴C]-羊毛甾醇(底物)。反应混合物于30°C孵育60分钟后,加入氯仿-甲醇(2:1,v/v)终止反应。萃取有机层后,通过薄层层析(TLC)分离代谢产物,利用闪烁计数器定量产物(4,4-二甲基胆甾-8,14,24-三烯-3β-醇)的放射性,采用Lineweaver-Burk图从剂量抑制曲线中计算CYP51抑制的Ki值 [2] 2. 甾醇生物合成抑制实验:将白色念珠菌细胞接种于含[¹⁴C]-乙酸盐(1 μCi/mL)和不同浓度雷夫康唑(0.001–0.1 μg/mL)的沙氏葡萄糖肉汤中,37°C孵育18小时。收集细胞并经氢氧化钾乙醇溶液皂化,用石油醚萃取非皂化脂质,通过TLC分离甾醇,检测麦角甾醇(真菌主要甾醇)和羊毛甾醇(CYP51底物)的放射性。测得雷夫康唑对白色念珠菌麦角甾醇合成的半数抑制浓度(IC₅₀)为0.005 μg/mL [2] |
| 细胞实验 |
抗真菌药敏试验方法。[2]
所有分离株(马拉色菌除外)均采用NCCLS概述的参考肉汤大稀释法进行检测,并对其进行了改进,用于丝状真菌的抗真菌检测。BMS来自卫材公司,FLU来自辉瑞公司,ITR来自杨森制药,AMB来自百时美施贵宝公司。 FLU和ITR的解释性MIC断点来自ncls指南;这些断点可作为假丝酵母菌株的解释性指南。ncls推荐的流感断点如下:≤8 μg/ml,敏感;16 ~ 32 μg/ml,敏感剂量依赖性(S-DD);≥64 μg/ml耐药。对于ITR, nccls推荐的MIC断点为:≤0.13 μg/ml,敏感;0.25 ~ 0.5 μg/ml, S-DD;≥1 μg/ml耐药。在这一点上,没有为BMS建立解释性MIC断点。考虑到两种化合物在狗的血浆中达到相同的峰值水平,为了讨论本报告中的MIC结果,我们将使用ITR解释BMS的断点。至于AMB, NCCLS没有推荐可解释的MIC断点,尽管在动物模型中,AMB MIC为bb0.1 μg/ml的念珠菌分离株出现耐药性。因此,当采用NCCLS RPMI 1640方法时,本研究将AMB耐药定义为AMB mic≥2 μg/ml。 酵母菌按NCCLS指南进行肉汤稀释,丝状真菌按Espinel-Ingroff法和Kerkering法进行改良。琼脂稀释法用于毛马拉色菌已在前面描述。 根据NCCLS推荐的方法,用肉汤稀释法测定MIC终点。AMB mic被定义为抑制所有可见生长的最低药物浓度(即100%抑制)。FLU、ITR和BMS mic被定义为抑制生长对照管中80%生长的最低药物浓度(通过与生长对照的1:5稀释率进行比较确定),但毛马拉色菌除外,其100%生长抑制是终点。 Minimum fungicidal concentrations (MFCs)最低杀真菌浓度。[2] 最低杀真菌浓度(mfc)是通过从每管中取出0.1 ml的MIC肉汤大稀释系列中未见生长的培养基传代到无药SDA板上来测定的,如前所述。测定菌落计数,并根据每毫升菌落数减少的程度定义mfc,即MFC99表示每毫升最终接种量的菌落数减少99%,MFC95表示减少95%,MFC90表示减少90%。 1. 微量肉汤稀释法测定MIC:将真菌菌株接种于酵母氮源肉汤(用于酵母)或RPMI 1640培养基(用于霉菌),最终菌浓度为1×10⁶ CFU/mL(酵母)或1×10⁴孢子/mL(霉菌)。在96孔微量滴定板中,将雷夫康唑在培养基中进行系列稀释(0.001–64 μg/mL),并向各孔加入真菌菌液。平板于35°C孵育24小时(酵母)或48–72小时(霉菌),MIC定义为抑制真菌可见生长的最低雷夫康唑浓度;对于曲霉属菌株,MIC为与对照组相比浊度降低50%的药物浓度 [1] 2. 时间杀菌实验:将白色念珠菌或烟曲霉接种于RPMI 1640培养基中,菌浓度为1×10⁶ CFU/mL,加入浓度为0.5×MIC、1×MIC、2×MIC和4×MIC的雷夫康唑。分别在0、6、12、24和48小时取样,用无菌生理盐水系列稀释后,涂布于沙氏葡萄糖琼脂(SDA)平板。35°C孵育24–48小时后计数CFU,计算与初始菌液相比的CFU对数降低值,以判断抑菌或杀菌活性 [2] 3. 琼脂稀释法:制备含系列稀释雷夫康唑(0.001–64 μg/mL)的SDA平板,将真菌悬液(酵母为1×10⁴ CFU/点,霉菌为1×10³孢子/点)点种于平板上,35°C孵育24–72小时。MIC为阻止菌落形成的最低雷夫康唑浓度,该实验用于验证微量肉汤稀释法的结果 [1] |
| 动物实验 |
Mouse
To prepare ravuconazole, mix 10% DMSO with 0.5% CMC.For 48 hours, C. neoformans No. 3 is grown on an SDA plate at 30°C. Sterile saline is used to prepare the challenge organisms. The tail vein is the route of infection in mice (n = 5; p. 5). Oral administration of ravuconazole is initiated 1 hour after infection, twice a day for 5 days in a volume of 0.2 mL per dose. In 0.5% CMC, controls are given 10% DMSO. The dosages of ravuconazole are 8 and 32 mg/kg. Every day after the infection for 21 days, mortality is recorded. Determining the delay in mortality allows for the evaluation of drug efficacy. 1. Mouse model of systemic candidiasis: Female ICR mice (6–8 weeks old) are infected intravenously via the tail vein with 5×10⁶ CFU of C. albicans (ATCC 90028) in 0.2 mL of sterile saline. Treatment with Ravuconazole is initiated 2 hours post-infection; the drug is formulated as a suspension in 0.5% carboxymethylcellulose (CMC) and administered orally by gavage at doses of 1, 5, or 10 mg/kg once daily for 7 days (vehicle control receives 0.5% CMC only). Survival is monitored daily for 14 days, and on day 7, mice are euthanized to collect kidneys for fungal burden determination (kidney tissue is homogenized, diluted, plated on SDA, and CFU are counted after incubation) [1] 2. Neutropenic mouse model of invasive aspergillosis: Female BALB/c mice are rendered neutropenic by intraperitoneal injection of cyclophosphamide (150 mg/kg) on day -4 and day -1 before infection, and cortisone acetate (100 mg/kg) on day -1. Mice are infected intranasally with 1×10⁶ conidia of A. fumigatus (ATCC 13073) in 20 μL of saline under light anesthesia. Ravuconazole (formulated in 0.5% CMC) is administered orally at 5 or 10 mg/kg/day starting 24 hours post-infection for 10 days. Survival is recorded for 21 days, and lung tissue is collected on day 7 to quantify fungal burden by CFU counting [3] 3. Mouse model of cryptococcosis: Male ICR mice are infected intravenously with 1×10⁷ CFU of C. neoformans (ATCC 32244) in 0.2 mL of saline. Ravuconazole is given orally at 1 or 5 mg/kg/day for 14 days, starting 2 hours post-infection. Brain and lung tissues are collected on day 14, homogenized, and plated on SDA for CFU determination; survival is monitored for 30 days [3] 4. Immunocompromised mouse model of fluconazole-resistant candidiasis: Neutropenic mice (cyclophosphamide-treated) are infected intravenously with 1×10⁷ CFU of fluconazole-resistant C. albicans (MIC >64 μg/mL for fluconazole). Ravuconazole (10 mg/kg/day) or fluconazole (20 mg/kg/day) is administered orally for 7 days, and survival is monitored for 14 days; kidney fungal burden is measured on day 7 [1] |
| 药代性质 (ADME/PK) |
ADME/Pharmacokinetics
1. Oral absorption: Lavoconazole has excellent oral bioavailability in animals: 92% in rats, 85% in dogs, and 78% in monkeys after oral administration of 10 mg/kg[1] 2. Plasma half-life: After a single oral dose of 10 mg/kg in rats, the plasma half-life (t₁/₂) of lavonazole was 12 hours; the t₁/₂ in dogs was 24 hours, and the t₁/₂ in monkeys was 36 hours, supporting once-daily oral administration[1] 3. Tissue distribution: Lavoconazole has extensive tissue distribution in rats; after oral administration of 10 mg/kg, the tissue to plasma concentration ratios 24 hours after administration were: liver 5.2, lung 8.5, kidney 3.8, brain 2.1[3] 4. Metabolism: Lavoconazole is metabolized in the liver by cytochrome P450 3A4 (CYP3A4) to produce hydroxylated metabolites, which are inactive against fungi; less than 5% of the original drug is excreted unchanged in urine and feces [1] 5. Plasma protein binding: Lavoconazole is highly bound to plasma proteins in humans (98.5%), rats (97.8%) and dogs (98.2%), mainly to albumin and α₁-acid glycoprotein [2] |
| 毒性/毒理 (Toxicokinetics/TK) |
1. Acute toxicity: In acute toxicity studies, the oral LD₅₀ of lavonazole was >2000 mg/kg in mice and rats; in dogs, no acute lethal toxicity was observed at doses up to 5000 mg/kg[1]
2. Subchronic toxicity: In a 28-day oral toxicity study in rats, lavonazole at doses up to 100 mg/kg/day did not cause significant changes in body weight, food consumption, or hematologic/biochemical parameters; mild hepatocyte vacuolation was observed at a dose of 200 mg/kg/day, which was reversible upon discontinuation of the drug[2] 3. Chronic toxicity: In a 6-month canine study, a dose of lavonazole at 50 mg/kg/day (10 times the therapeutic dose) resulted in a slight increase in liver enzyme levels (ALT, AST), but no histopathological changes were observed in the liver or other organs[1] 4. Drug interactions: Lavoconazole It is a moderate inhibitor of human CYP3A4 (Ki = 0.5 μM), and when used in combination with CYP3A4 substrates (e.g., midazolam), it can increase plasma concentrations in healthy volunteers by 2-fold; no significant interactions with warfarin or digoxin were observed [2]. 5. Reproductive toxicity: In teratogenicity studies in rats and rabbits, no evidence of fetal malformation or embryotoxicity was shown at doses up to 50 mg/kg/day of lavoconazole [1]. |
| 参考文献 |
|
| 其他信息 |
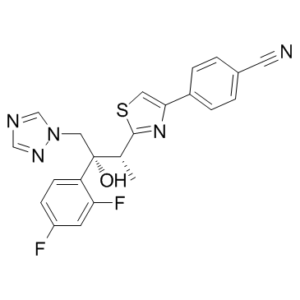
Lavoconazole belongs to the triazole class of compounds, with the chemical name 1-butyl-1H-1,2,4-triazole, where the butyl group is substituted at positions 2, 2, and 3 with hydroxyl, 2,4-difluorophenyl, and 4-(p-cyanophenyl)-1,3-thiazolyl-2-yl groups, respectively (R,R stereoisomers). It exerts its antifungal activity by inhibiting 14α-demethylase, an enzyme involved in sterol synthesis, leading to fungal cell wall lysis and cell death. (NCIO4) Lavoconazole can be used as an ergosterol biosynthesis inhibitor, an antifungal drug, an EC 1.14.14.154 (sterol 14α-demethylase) inhibitor, and an anti-leishmaniasis drug. It belongs to the triazole, fluorobenzene, tertiary alcohol, 1,3-thiaazole, and nitrile classes of compounds. Lavoconazole is a triazole compound with antifungal activity. Lavoconazole inhibits 14α-demethylase (an enzyme involved in sterol synthesis), leading to fungal cell wall lysis and cell death. (NCI04)
Drug Indications Studied for the treatment of fungal infections, aspergillosis, candidiasis, and onychomycosis. In this study, BMS and ITR at a concentration of 1 μg/ml showed inhibitory activity against all but one of the 16 Aspergillus strains. Similarly, Hata et al. also observed consistent activity of BMS and ITR against Aspergillus strains. The anti-Aspergillus efficacy of BMS and ITR was comparable. Fluconazole (FLU) was ineffective against Aspergillus fungi. Buthiacromegaly toxin (BMS) and itraconazole (ITR) also showed fungicidal activity against 50% to 74% of the tested Aspergillus strains. Compared to fluconazole, buthiacromegaly toxin and itraconazole showed significantly different activities against other filamentous fungi, while fluconazole was ineffective against most filamentous fungi. Itraconazole and buthiacromegaly toxin were effective against dermatophytes, while fluconazole showed lower activity against Microsporum gypseum. Fusarium stenocladus, Penicillium mutagenesis, and Penicillium fungi are sensitive to thiodicarboxamide and itraconazole. While both itraconazole and thiodicarboxamide are effective against most dark-skinned fungi, itraconazole appears to be slightly more active than fluconazole. Both thiodicarboxamide and itraconazole show low activity against most Pseudomonas boydii, Sporothrix schenckii, and Zygomycetes, and are generally ineffective against Fusarium fungi. Unlike Aspergillus, BMS and ITR have no fungicidal activity against other filamentous fungi. In summary, BMS is a novel triazole antifungal drug with 2 to 4 times the potency of ITR and 40 times the activity of fluconazole (FLU) against a wide range of fungi. Its antibacterial spectrum includes some fluconazole-resistant yeast strains. Similar to ITR, BMS has fungicidal activity against Cryptococcus and many Aspergillus strains. BMS’s in vitro antibacterial spectrum makes it a promising candidate for human treatment. [2] 1. Lavoconazole (ER-30346, BMS-207147) is a novel triazole antifungal drug jointly developed by Eisai Co., Ltd. and Bristol-Myers Squibb, aiming to overcome the limitations of existing azole drugs (e.g., narrow antibacterial spectrum, drug resistance)[1] 2. Mechanism of action: Lavoconazole inhibits fungal lanosterol 14α-demethylase (CYP51), which is a key enzyme in the biosynthesis of ergosterol (a major component of fungal cell membranes). Inhibition of CYP51 leads to ergosterol depletion and accumulation of toxic methylated sterols, which disrupt the integrity and function of fungal cell membranes, ultimately resulting in growth inhibition or cell death [2]. 3. Antifungal properties: Lavoconazole has a broader antifungal spectrum and higher efficacy than fluconazole and itraconazole, especially effective against Aspergillus and azole-resistant Candida strains; it has a long plasma half-life and high oral bioavailability, making it suitable for once-daily oral administration [3]. 4. Clinical development: Lavoconazole was evaluated in a phase II clinical trial for the treatment of invasive fungal infections (aspergillosis, candidiasis, cryptococcosis) in immunocompromised patients, and the results showed good efficacy and safety; however, due to the strategic decisions of pharmaceutical companies, its development has not entered phase III [1]. |
| 分子式 |
C22H17N5OF2S
|
|---|---|
| 分子量 |
437.46508
|
| 精确质量 |
437.112
|
| 元素分析 |
C, 60.40; H, 3.92; F, 8.69; N, 16.01; O, 3.66; S, 7.33
|
| CAS号 |
182760-06-1
|
| 相关CAS号 |
Ravuconazole-d4;1329499-27-5
|
| PubChem CID |
467825
|
| 外观&性状 |
Off-white to yellow solid powder
|
| 密度 |
1.4±0.1 g/cm3
|
| 沸点 |
674.9±65.0 °C at 760 mmHg
|
| 闪点 |
362.0±34.3 °C
|
| 蒸汽压 |
0.0±2.2 mmHg at 25°C
|
| 折射率 |
1.666
|
| LogP |
3.89
|
| tPSA |
115.86
|
| 氢键供体(HBD)数目 |
1
|
| 氢键受体(HBA)数目 |
8
|
| 可旋转键数目(RBC) |
6
|
| 重原子数目 |
31
|
| 分子复杂度/Complexity |
657
|
| 定义原子立体中心数目 |
2
|
| SMILES |
FC1=CC(F)=CC=C1[C@](CN2C=NC=N2)(O)[C@@H](C)C3=NC(C4=CC=C(C#N)C=C4)=CS3
|
| InChi Key |
OPAHEYNNJWPQPX-RCDICMHDSA-N
|
| InChi Code |
InChI=1S/C22H17F2N5OS/c1-14(21-28-20(10-31-21)16-4-2-15(9-25)3-5-16)22(30,11-29-13-26-12-27-29)18-7-6-17(23)8-19(18)24/h2-8,10,12-14,30H,11H2,1H3/t14-,22+/m0/s1
|
| 化学名 |
p-(2-((alphaR,betaR)-2,4-Difluoro-beta-hydroxy-alpha-methyl-beta-(1H-1,2,4-triazol-1-ylmethyl)phenethyl)-4-thiazolyl)benzonitrile
|
| 别名 |
BMS 207147; BMS207147; BMS-207147; 182760-06-1; Ravuconazole [INN]; Benzonitrile, 4-[2-[(1R,2R)-2-(2,4-difluorophenyl)-2-hydroxy-1-methyl-3-(1H-1,2,4-triazol-1-yl)propyl]-4-thiazolyl]-; ER-30346 ;ER 30346 ; ER30346.
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ≥ 50 mg/mL (~114.29 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (5.71 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (5.71 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.5 mg/mL (5.71 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 配方 4 中的溶解度: 10% DMSO+40% PEG300+5% Tween-80+45% Saline: ≥ 2.5 mg/mL (5.71 mM) 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2859 mL | 11.4294 mL | 22.8587 mL | |
| 5 mM | 0.4572 mL | 2.2859 mL | 4.5717 mL | |
| 10 mM | 0.2286 mL | 1.1429 mL | 2.2859 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。