| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 5g |
|
||
| Other Sizes |
|
| 靶点 |
THR-β (EC50 = 0.21 μM)
- Resmetirom (MGL-3196) selectively targets thyroid hormone receptor β (THR-β) with an EC50 of 0.21 μM, showing 28-fold selectivity over THR-α (EC50 = 3.74 μM) in reporter gene assays. [1] |
||
|---|---|---|---|
| 体外研究 (In Vitro) |
与 THR-α (EC50=3.74 μM) 相比,Resmetirom (MGL-3196) 对 THR-β (EC50=0.21 μM) 的选择性提高了 28 倍。 Resmetirom (MGL-3196) 用于抑制 hERG 通道,IC20 约为 30 μM。对CYP2C9的抑制作用相对温和(约22μM),而CYP3A4/5和CYP2C19的IC50>50μM[1]。
- 在受体激活实验中,瑞美替罗(Resmetirom,MGL-3196) 呈剂量依赖性激活THR-β介导的转录,在1 μM时达到最大激活效果;浓度高达10 μM时对THR-α无显著激活作用。[1] - 对CYP2C9表现出弱抑制(IC50≈22 μM),对CYP3A4/5或CYP2C19无显著抑制(IC50>50 μM)。[1] - 对hERG通道的IC20约为30 μM,提示心脏心律失常风险较低。[1] - 在肠道上皮细胞模型中,可调节胆汁酸转运体基因表达,包括上调ABCB11和下调ASBT,可能有助于减少肠道脂质吸收。[3] |
||
| 体内研究 (In Vivo) |
在大鼠中,瑞美罗 (MGL-3196) 显示出合理的口服生物利用度和良好的暴露量。分布量少,出清少。口服 Resmetirom (MGL-3196) 溶液后,DIO 小鼠中发现暴露量与剂量成比例增加 [1]。由于肝脏 TG,给予 Resmetirom (MGL-3196) 的大鼠胆固醇和肝脏大小有所减少。使用 Resmetirom (MGL-3196) 治疗的动物心脏或肾脏大小或骨矿物质密度 (BMD) 没有变化 [1]。
雷美替隆治疗不影响体重,但导致肝脏重量、肝脂肪变性、血浆丙氨酸氨基转移酶活性、肝脏和血浆胆固醇以及血糖显著降低。这些代谢效应转化为NAFLD活动评分的显著改善。此外,α-平滑肌肌动蛋白含量较低和参与纤维化的基因下调表明肝纤维化减少。[2] - 在高脂饮食诱导的非酒精性脂肪性肝炎(NASH)小鼠模型中,口服瑞美替罗(Resmetirom,MGL-3196)(3-10 mg/kg/天,持续23天)使肝脏甘油三酯水平降低32-45%,NAFLD活动评分(NAS)较载体对照组降低≥2分。[2] - 通过减少胶原沉积和下调肝组织中纤维化标志物(Col1a1、α-SMA)改善肝纤维化。[2] - 在大鼠中,瑞美替罗(Resmetirom,MGL-3196)(5-37.5 mg/kg)呈剂量依赖性降低血清胆固醇和LDL-C,且不影响骨密度或心脏参数。[1] - 在小鼠模型中,通过增加亲水性胆汁酸(如牛磺胆酸)和减少疏水性胆汁酸重构胆汁酸谱,这与肠道脂质吸收减少相关。[3] |
||
| 酶活实验 |
THR/RXR/GRIP1 Assay1]
将THR-β(H6-THR-β)的配体结合结构域(氨基酸148-410)和THR-α(H6-THR-α)的配体连接结构域(氨基202-461)克隆到含有N端六His序列的大肠杆菌表达载体pET28a中。在大肠杆菌BL21(DE3)细胞中产生了重组六His标记蛋白。使用摇瓶在25°C下在0.2 mM IPTG中诱导24小时,在Terrific Broth(内部制备的杆菌胰蛋白胨(3.3%,w/v)、Difico酵母提取物(2.0%,w/v”)和NaCl(0.5%,w/v“)培养基中培养细胞,收获细胞,并用五倍体积的缓冲液a(0.05 M Tris、0.3 M NaCl、1%w/v甜菜碱、0.01 M咪唑、0.02 Mβ-巯基乙醇,pH 8.0)裂解。将溶菌酶(1.0 mg/mL)和完全蛋白酶抑制剂混合物加入浆液中,在4°C下对溶液进行5次超声波处理1分钟。将悬浮液在Ti45 Beckmann转子中以127 300 RCF离心2小时,将上清液装载到NI_NTA琼脂糖(Quigen 30210)柱上。用缓冲液a洗涤后,用含有0.25M咪唑的缓冲液a洗脱H6-TRβ或H6-TRα。[1] 人维甲酸X受体(氨基酸225-462)(RxRα)的配体结合结构域用N端His6和EE(EFMPME)标签工程化,这是His6和EE序列之间的凝血酶切割位点,并克隆到pACYC载体中。在大肠杆菌细胞中产生了His6-EE标记的蛋白质。使用摇瓶在18°C下在0.1 mM IPTG中培养18小时,收获细胞,并用五倍体积的缓冲液B(0.025 M Tris、0.3 M NaCl、0.02 M咪唑、0.01 Mβ-巯基乙醇,pH 8.0)悬浮。加入溶菌酶(0.2 mg/mL)和完全蛋白酶抑制剂混合物,并在4°C下搅拌30分钟。将悬浮液在4°C下超声处理30秒,共五次。将悬浮液在12000 RCF下离心20分钟。上清液用0.45μm孔径的膜过滤,加入0.5%的NP-40。His6标记的蛋白与NiNTA金属亲和树脂结合并从中洗脱。将蛋白质浓缩并透析。 通过凝血酶消化从EE RxRα中去除His6标签,每毫克蛋白质使用10单位凝血酶(Pharmacia,Piscataway,NJ),在25°C下孵育2小时。使用苯甲脒琼脂糖6B分批去除凝血酶。将蛋白质浓缩并透析。该蛋白用于辅活化剂肽募集试验。[1] THR-β/RXR/GRIP1共激活肽募集试验[1] 将30μL H6-THR-β(50 nM)在50 mM Hepes、pH 7.0、1 mM DTT、0.05%NP40和0.2 mg/mL BSA(结合缓冲液)中与等体积的EE RxRα(50 nM)在结合缓冲液中混合。然后加入6μL的T3(0-14.8μM)或测试化合物(0-1.2 mM)的DMSO溶液,并在37°C下孵育30分钟。然后加入30μL的生物素-GRIP1肽(生物素-Aca-HGTSLKEKILHRLLQDSPSVDL-CONH2)(100 nM)的30μL结合缓冲液加5%的DMSO溶液并在37℃下孵育60分钟。加入30μL的溶液,其中含有12 nM铕偶联的抗hexa-His抗体和160 nM APC偶联的链霉抗生物素,溶于50 mM Tris、pH 7.4、100 mM NaCl和0.2 mg/mL BSA中,以及将溶液在4°C下孵育过夜。将等分试样(35μL/样品)转移到384孔黑色微量滴定板上。HTRF信号在Victor 5阅读器上读取。[1] THR-α/RXR/GRIP1共激活肽募集试验[1] 除了使用125 nM H6-THR-α、125 nM EE RXRα和250 nM生物素-GRIP1外,该测定方案与上述THR-β/RXR/GRIP1共激活肽募集测定基本相同。 - THR-β转录激活实验: - 向HEK293细胞共转染人THR-β表达质粒和含甲状腺激素反应元件(TREs)的荧光素酶报告质粒。 - 用系列稀释的瑞美替罗(Resmetirom,MGL-3196)(0.01-10 μM)处理细胞24小时。 - 检测荧光素酶活性并归一化至蛋白浓度,通过剂量-反应曲线计算EC50值。[1] - CYP抑制实验: - 将重组CYP酶(CYP3A4、CYP2C9、CYP2C19)与瑞美替罗(Resmetirom,MGL-3196)(0.1-100 μM)及特异性底物共同孵育。 - 通过HPLC定量代谢产物生成量以确定抑制效力(IC50)。[1] |
||
| 细胞实验 |
- 肝脏脂质蓄积实验:
- 原代肝细胞在含油酸的培养基中培养以诱导脂质蓄积。
- 用瑞美替罗(Resmetirom,MGL-3196)(0.1-10 μM)处理细胞48小时后进行油红O染色。
- 通过分光光度法定量脂滴,在浓度≥1 μM时脂质减少30-50%。[2]
- 基因表达分析: - 肝细胞经瑞美替罗(Resmetirom,MGL-3196)(1 μM)处理6小时后,通过qPCR检测脂质代谢基因(SREBP-1c、PPARα)的mRNA水平,结果显示生脂基因下调而脂肪酸氧化基因上调。[2] |
||
| 动物实验 |
|
||
| 药代性质 (ADME/PK) |
吸收
每日多次服用80 mg或100 mg瑞美替罗后,中位达峰时间(Tmax)约为4小时。与空腹状态相比,与食物同服导致Cmax降低33%,AUC降低11%,中位Tmax延迟约2小时。 消除途径 口服100 mg放射性标记的瑞美替罗后,约67%的总放射性剂量从粪便中回收,主要以代谢物的形式存在;24%的总放射性剂量从尿液中回收。未在粪便中检测到未改变的标记瑞美替罗,其占尿液回收剂量的1%。代谢物MGL-3623分别占粪便和尿液回收剂量的3.3%和16%。草酸代谢物在血浆中检测到,但在尿液中未检测到。 分布容积 瑞美替罗稳态表观分布容积 (Vd/F) 为 68 (227%) L。 清除率 稳态表观清除率 (CL/F) 为 17.5 (56.3%) L/h。 蛋白结合率 瑞美替罗的蛋白结合率大于 99%。 代谢/代谢物 瑞美替罗经 CYP2C8 代谢。MGL-3623 是其主要代谢物,对 THR-β 的抑制效力比瑞美替罗低 28 倍。每日一次服用 100 mg 瑞美替罗后,MGL-3623 占稳态瑞美替罗 AUC 的 33% 至 51%。 生物半衰期 终末血浆半衰期中位数为 4.5 小时。 - 在大鼠中,Resmetirom (MGL-3196) 显示出良好的口服生物利用度(未具体说明),给药后 2 小时内达到血浆峰浓度。[1] - 在临床前动物模型中,其清除率低,分布容积中等。[1] - 在人体中,其血浆蛋白结合率高(原始参考文献中未具体说明),主要 CYP 酶的代谢作用极弱。[1] |
||
| 毒性/毒理 (Toxicokinetics/TK) |
肝毒性
接受瑞美替罗治疗的患者中,相当一部分会出现轻度、短暂的血清转氨酶升高,通常在治疗的前4周内发生。这些升高通常较轻,具有自限性,且不伴有症状或黄疸。此外,这些早期变化之后,血清酶通常会下降,并在3至6个月后恢复正常。肝脏相关酶的改善与肝脏脂肪减少和脂肪性肝炎的组织学证据在一定程度上相关。治疗52周后,肝活检显示26%至30%的患者非酒精性脂肪性肝炎(NASH)得到缓解。这些变化是否能持续或随着治疗的进行而加重尚不清楚。该疗法不会导致体重减轻,且一旦停药,肝脏组织学和纤维化的改善可能会消失。 对1300多名接受瑞美替罗(resmetirom)治疗(每日剂量80或100毫克,疗程长达一年)的非酒精性脂肪性肝炎(NASH)成年患者的肝功能检查结果进行分析,发现2例患者出现肝损伤,至少可能与瑞美替罗有关。首次发病潜伏期分别为2个月和3个月[ALT分别为236 U/L和578 U/L,碱性磷酸酶(ALP)未知和64 U/L,胆红素分别为0.6 mg/dL和1.1 mg/dL]。两名患者在停药后1至2个月内完全康复。一名患者重新开始治疗后,28 天内再次出现肝损伤(ALT 3226 U/L,碱性磷酸酶 140 U/L,胆红素 10.9 mg/dL),且比初次发作更为严重,但停药后 2 个月内自行缓解。在这两个病例中,仍存在其他诊断的可能性。 可能性评分:D(可能是临床上明显的肝损伤的罕见病因)。 妊娠和哺乳期影响 ◉ 哺乳期用药概述 目前尚无关于瑞美替罗在哺乳期临床应用的信息。由于瑞美替罗与血浆蛋白的结合率超过 99%,因此其在乳汁中的含量可能很低。如果母亲需要服用瑞美替罗,则不应因此停止哺乳。在获得更多数据之前,哺乳期妇女使用瑞美替罗时应密切监测婴儿情况,尤其是在哺乳新生儿或早产儿时。 ◉ 对母乳喂养婴儿的影响 截至修订日期,未找到相关的已发表信息。 ◉ 对泌乳和母乳的影响 截至修订日期,未找到相关的已发表信息。 - 在急性毒性研究中,小鼠在剂量高达 200 mg/kg 时未观察到死亡。[1] - 在大鼠中,长期给药(10 mg/kg/天,持续 28 天)未引起肝肾功能指标的显著变化。[1] - 在临床前模型中,未检测到对骨矿物质密度或心脏结构的不良影响。[1] - 在临床试验中,常见不良事件包括短暂性腹泻(33%)和恶心(14%),程度为轻度至中度。[7] |
||
| 参考文献 |
[1]. Discovery of 2-[3,5-dichloro-4-(5-isopropyl-6-oxo-1,6-dihydropyridazin-3-yloxy)phenyl]-3,5-dioxo-2,3,4,5-tetrahydro[1,2,4]triazine-6-carbonitrile (MGL-3196), a Highly Selective Thyroid Hormone Receptor β agonist in clinical trials for the treatment of dyslipidemia. J Med Chem. 2014 May 22;57(10):3912-23.
[2]. Activation of thyroid hormone receptor-β improved disease activity and metabolism independent of body weight in a mouse model of non-alcoholic steatohepatitis and fibrosis. Br J Pharmacol. 2021 Jun;178(12):2412-2423. [3]. A new mechanism of thyroid hormone receptor β agonists ameliorating nonalcoholic steatohepatitis by inhibiting intestinal lipid absorption via remodeling bile acid profiles. Acta Pharmacologica Sinica. 2024 Oct;45(10):2134-2148. |
||
| 其他信息 |
MGL-3196 已用于研究非酒精性脂肪性肝炎和杂合子家族性高胆固醇血症的治疗试验。
Resmetirom 是一种甲状腺激素受体β (THR-β) 激动剂,与饮食和运动联合用于治疗中重度纤维化的非酒精性脂肪性肝炎 (NASH)。Resmetirom 治疗在治疗的第一个月会出现轻度且短暂的血清转氨酶升高,罕见情况下会出现急性肝损伤,这些损伤可能很严重,但停药后可逆转。 药物适应症 治疗非酒精性脂肪性肝炎 (NASH) Resmetirom 是一种甲状腺激素受体β (THR-β) 激动剂。 2024年3月14日,美国食品药品监督管理局(FDA)批准瑞美替罗(Resmetirom)作为首个用于治疗非酒精性脂肪性肝炎(NASH)引起的肝纤维化的药物。NASH是一种非酒精性脂肪肝(NAFLD)。甲状腺激素直接调节肝脏的脂质代谢;因此,NAFLD患者常出现甲状腺功能受损,例如血清甲状腺激素水平降低。瑞美替罗通过刺激脂肪酸降解和氧化来减少肝脏脂肪。 瑞美替罗是一种甲状腺激素受体β激动剂。其作用机制包括作为甲状腺激素受体β激动剂、细胞色素P450 2C8抑制剂、有机阴离子转运多肽1B1抑制剂、有机阴离子转运多肽1B3抑制剂和乳腺癌耐药蛋白抑制剂。 瑞美替罗(Resmetirom)是一种小分子药物,目前已完成最多IV期临床试验(涵盖所有适应症),于2024年首次获批,用于治疗非酒精性脂肪肝和非酒精性脂肪性肝炎,并有3项在研适应症。 甲状腺激素受体β激动剂 药物适应症 瑞美替罗适用于与饮食和运动联合治疗,用于治疗伴有中度至重度肝纤维化(符合F2至F3期纤维化)的非肝硬化性非酒精性脂肪性肝炎(NASH)成人患者。失代偿期肝硬化患者应避免使用。该适应症基于NASH和纤维化的改善而获得加速批准。该适应症的持续批准可能取决于在验证性试验中对临床获益的验证和描述。 药效学 Resmetirom 是甲状腺激素受体β (THR-β) 的部分激动剂。在体外 THR-β 激活功能试验中,Resmetirom 的疗效达到三碘甲状腺原氨酸 (T3) 的 83.8%,EC50 为 0.21 µM。在相同的甲状腺激素受体α (THR-α) 激动功能试验中,Resmetirom 相对于 T3 的疗效为 48.6%,EC50 为 3.74 µM。Resmetirom 可降低肝脏脂肪含量和游离甲状腺素 (FT4)(一种甲状腺激素原)的浓度。尽管尚无定论,但文献中已有报道表明,雷斯美替罗可能将甲状腺素 (T4) 转化为三碘甲状腺原氨酸 (T3)。它还能提高性激素结合球蛋白的浓度。 作用机制 甲状腺激素,例如游离甲状腺素 (FT4) 和游离三碘甲状腺原氨酸 (FT3),是肝脏脂质代谢的关键调节因子。甲状腺激素受体β (THR-β) 是肝脏中主要的甲状腺激素受体,刺激该受体可降低肝内甘油三酯水平。许多非酒精性脂肪性肝病 (NAFLD) 患者存在甲状腺功能障碍,例如甲状腺功能减退,这使其成为 NAFLD 的一个重要危险因素。甲状腺功能减退还与脂肪组织脂解失调和脂肪组织向肝脏释放的游离脂肪酸增加有关,从而促进肝脏胰岛素抵抗。循环中促炎性脂肪因子水平升高,可导致肝脏炎症和纤维化,也可观察到。Resmetirom 是 THR-β 的部分激动剂,可促进脂肪自噬和肝脏脂肪酸 β-氧化,从而减少肝脏脂肪。它对 THR-β 的选择性比 FT3 高约 28 倍,而对甲状腺激素受体 α (THR-α) 的选择性则较低,THR-α 主要表达于心脏和骨骼。 - Resmetirom (MGL-3196) 是一种靶向肝脏的 THR-β 激动剂,旨在避免 THR-α 对心脏和骨骼的影响。[1] - 其作用机制包括促进脂肪酸氧化、减少从头脂肪生成以及调节胆汁酸代谢,从而改善非酒精性脂肪性肝炎 (NASH) 的病理。 [2,3] - 该药物于2024年获得FDA加速批准,用于治疗中重度纤维化(F2-F3)的非酒精性脂肪性肝炎(NASH)。[1,7] - 在3期临床试验中,25.9%至29.9%的患者在接受52周治疗后NASH得到缓解,且纤维化程度未加重。[7] |
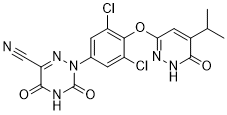
| 分子式 |
C17H12CL2N6O4
|
|
|---|---|---|
| 分子量 |
435.22
|
|
| 精确质量 |
434.03
|
|
| 元素分析 |
C, 46.92; H, 2.78; Cl, 16.29; N, 19.31; O, 14.70
|
|
| CAS号 |
920509-32-6
|
|
| 相关CAS号 |
|
|
| PubChem CID |
15981237
|
|
| 外观&性状 |
Typically exists as Yellow to orange solids at room temperature
|
|
| 密度 |
1.65±0.1 g/cm3 (20 °C, 760 mmHg)
|
|
| LogP |
2.098
|
|
| tPSA |
146.52
|
|
| 氢键供体(HBD)数目 |
2
|
|
| 氢键受体(HBA)数目 |
7
|
|
| 可旋转键数目(RBC) |
4
|
|
| 重原子数目 |
29
|
|
| 分子复杂度/Complexity |
878
|
|
| 定义原子立体中心数目 |
0
|
|
| SMILES |
N#CC1C(=O)NC(=O)N(C2C=C(Cl)C(OC3C=C(C(C)C)C(=O)NN=3)=C(Cl)C=2)N=1
|
|
| InChi Key |
FDBYIYFVSAHJLY-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C17H12Cl2N6O4/c1-7(2)9-5-13(22-23-15(9)26)29-14-10(18)3-8(4-11(14)19)25-17(28)21-16(27)12(6-20)24-25/h3-5,7H,1-2H3,(H,23,26)(H,21,27,28)
|
|
| 化学名 |
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 3.75 mg/mL (8.62 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 37.5 mg/mL 澄清的 DMSO 储备液加入到400 μL PEG300中,混匀;再向上述溶液中加入50 μL Tween-80,混匀;然后加入450 μL 生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 3.75 mg/mL (8.62 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 37.5 mg/mL 澄清 DMSO 储备液加入 900 μL 20% SBE-β-CD 生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 3.75 mg/mL (8.62 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2977 mL | 11.4884 mL | 22.9769 mL | |
| 5 mM | 0.4595 mL | 2.2977 mL | 4.5954 mL | |
| 10 mM | 0.2298 mL | 1.1488 mL | 2.2977 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
Model of53(MGL-3196, magenta) bound to THR-β (1N46) with the T3 geometry (cyan) from3GWSsuperimposed. Polar interactions of53in the anion binding site are highlighted.J Med Chem.2014 May 22;57(10):3912-23. |
|---|
(A) 2D description of the binding site for T3 (PDB code3GWS). (B) 2D description of the binding site for the53model (MOE).J Med Chem.2014 May 22;57(10):3912-23. |
Left panel: cardiac α-MHC hnRNA relative levels (arbitrary units) in untreated thyroidectomized rats (control), euthyroid rats, and thyroidectomized rats 6 h after exposure to53dosed intraperitoneally at the specified doses.Right panel: activities of tested compounds relative to full activity (euthyroid or T3-treated) and exposure of the compound 6 h after dose.J Med Chem.2014 May 22;57(10):3912-23. |
Effects of53(MGL-3196) vs T3 on cholesterol and BMD in DIO mice.J Med Chem.2014 May 22;57(10):3912-23. |
|---|