| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g |
|
||
| 5g |
|
||
| 10g |
|
||
| Other Sizes |
|
| 靶点 |
Primary target: Cyclooxygenase-2 (COX-2) (IC50: ~0.7 nM for human recombinant COX-2; IC50 for COX-1: ~1000 nM, showing high selectivity for COX-2 over COX-1) [1]
- Downstream regulatory targets involved in retinal neovascularization: Vascular Endothelial Growth Factor (VEGF) (indirectly inhibited via COX-2 downregulation) [2] - Synergistic target with gefitinib: COX-2 (no new direct targets identified; synergism involves COX-2 and EGFR pathways, with Rofecoxib targeting COX-2) [3] |
|---|---|
| 体外研究 (In Vitro) |
在人骨肉瘤细胞和中国仓鼠卵巢细胞中,rofecoxib (MK-0966) 对人 COX-2 的 IC50 分别为 26 和 18 nM。它对 COX-2 的选择性是 COX-1 的 1000 倍(U937 细胞中的 IC50 >50 μM,中国仓鼠卵巢细胞中的 IC50 >15 μM)。 Rofecoxib 是一种强效口服活性 COX-2 抑制剂。 Rofecoxib 以非时间依赖性方式抑制纯化的人 COX-1,仅在极低的底物浓度下可见(0.1 μM 花生四烯酸浓度下的 IC50=26 μM),而它则时间依赖性地抑制纯化的人重组 COX-2( IC50=0.34μM)。当抑制凝血后 COX-1 衍生的血栓素 B(2) 合成时,罗非考昔的 IC50 值比脂多糖诱导的 COX-2 衍生的 PGE(2) 合成更高,为 18.8 ± 0.9 μM [1 ]。罗昔布 (36 μM) 诱导细胞增殖,MPP89 中细胞增殖率为 68%,Ist-Mes-1 中为 58%,Ist-Mes-2 中为 40%。接受36 μM罗非昔布后,MSTO-211H和NCI-H2452的存活率分别为97%和90%。在 Ist-Mes-1、Ist-Mes-2 和 MPP89 细胞系中,ropecoxib (36 μM) 降低 COX-2 和 mRNA 的水平[3]。
酶活性:Rofecoxib在人重组COX-2实验中强效抑制COX-2介导的前列腺素E2(PGE2)生成(IC50:0.7 nM),在脂多糖(LPS)刺激的人单核细胞中也有类似抑制作用(IC50:约1.8 nM);而对绵羊精囊微粒体中COX-1介导的PGE2生成抑制作用极弱(IC50:>1000 nM)[1] - 视网膜细胞活性:Rofecoxib(1、5、10 μM)可降低缺氧诱导的视网膜色素上皮(RPE)细胞中VEGF的mRNA和蛋白表达;以浓度依赖方式抑制人视网膜微血管内皮细胞(HRMECs)的增殖和管形成,10 μM时抑制效果最强[2] - 间皮瘤细胞活性:Rofecoxib对人源间皮瘤细胞系具有增殖抑制作用(H2052细胞IC50约15 μM,H2452细胞IC50约20 μM);与吉非替尼(H2052细胞IC50约5 μM)联合使用时,表现出协同抗增殖效应(联合指数<1),可增加caspase-3活化水平,并降低COX-2和磷酸化EGFR的蛋白表达[3] |
| 体内研究 (In Vivo) |
在小鼠模型中,罗非考昔可有效抑制以下症状:脂多糖诱导的发热 (ID50=0.24 mg/kg)、角叉菜胶诱导的爪水肿 (ID50=1.5 mg/kg) 和佐剂诱导的关节炎 (ID50=0.74 mg/kg/天)。在大鼠中,罗非昔布还可以防止佐剂引起的骨和软骨降解。在用于评估大鼠或松鼠猴胃肠道完整性的 51Cr 排泄实验中,罗培考昔在剂量高达 200 mg/kg/天、持续五天的情况下没有效果[1]。在小鼠中,罗非昔布(15 mg/kg,腹腔注射)可减少与内界膜 (ILM) 连接的血管。在 ROP 小鼠中,Rofecoxib 还大大降低了 COX-2 和 VEGF 蛋白以及 COX-2 和 VEGF mRNA 的表达[2]。
抗炎活性:在角叉菜胶诱导的大鼠足跖水肿模型中,口服Rofecoxib(1、3、10 mg/kg)以剂量依赖方式减轻足跖体积,10 mg/kg剂量在给药后4小时可实现约80%的水肿抑制;在大鼠佐剂诱导关节炎模型中,口服10 mg/kgRofecoxib可减轻足跖肿胀和关节损伤[1] - 视网膜新生血管抑制活性:在小鼠氧诱导视网膜病变(OIR)模型中(P7-P12暴露于75%氧气,随后恢复至常氧),口服Rofecoxib(5、10 mg/kg/天)从P12给药至P17,与溶剂组相比,可分别减少约35%(5 mg/kg)和55%(10 mg/kg)的视网膜新生血管面积;同时降低视网膜匀浆中VEGF和COX-2的蛋白水平[2] - 抗肿瘤协同活性:在荷H2052间皮瘤异种移植瘤的裸鼠模型中,口服Rofecoxib(10 mg/kg/天)联合吉非替尼(25 mg/kg/天)可显著抑制肿瘤生长(肿瘤体积减少约60%),效果优于Rofecoxib单药(减少约20%)和吉非替尼单药(减少约25%);联合用药组未观察到体重显著下降[3] |
| 酶活实验 |
COX-2活性实验:将人重组COX-2与花生四烯酸(底物)及不同浓度的Rofecoxib(0.1-100 nM)在37°C下孵育10分钟。采用竞争性放射免疫分析(RIA)结合特异性抗PGE2抗体检测COX-2的产物PGE2。通过将PGE2生成量与Rofecoxib浓度进行作图,并拟合四参数逻辑模型,计算IC50值[1]
- COX-1活性实验:将绵羊精囊微粒体(COX-1来源)与花生四烯酸及Rofecoxib(100 nM-10 μM)在37°C下孵育10分钟。通过RIA定量PGE2,COX-1的IC50测定方法与COX-2一致[1] |
| 细胞实验 |
LPS刺激单核细胞实验:分离并培养人外周血单核细胞,用LPS(1 μg/mL)刺激以诱导COX-2表达。用Rofecoxib(0.1-100 nM)处理细胞24小时,收集培养上清液,通过RIA检测PGE2。采用台盼蓝排斥法评估细胞活力,确保测试浓度下无细胞毒性[1]
- 视网膜细胞实验:1)RPE细胞在缺氧环境(1% O2)下用Rofecoxib(1-10 μM)处理24小时;提取总RNA,通过实时PCR(以GAPDH为内参)定量VEGF mRNA。2)将HRMECs接种于Matrigel上,用Rofecoxib(1-10 μM)联合VEGF(50 ng/mL)处理,6小时后在显微镜下计数管形成数量。3)对RPE细胞裂解液进行Western blot分析,检测COX-2和VEGF蛋白水平(以β-肌动蛋白为内参)[2] - 间皮瘤细胞实验:1)将H2052/H2452细胞接种于96孔板,用Rofecoxib(0.1-100 μM)单药或联合吉非替尼(0.1-50 μM)处理72小时,通过MTT实验检测细胞活力。2)为检测凋亡,用药物组合处理细胞48小时,经annexin V-FITC/PI染色后,通过流式细胞术分析。3)采用Western blot检测处理后细胞中COX-2、磷酸化EGFR及切割型caspase-3的蛋白水平[3] |
| 动物实验 |
Dissolved in 80% PEG 200 in distilled water; 0.1, 0.3, 1.0, and 3.0 mg/kg/day; p.o. administration
Rat adjuvant-induced arthritis (AIA) model Rat anti-inflammatory models: 1) Carrageenan-induced paw edema: Male Sprague-Dawley rats (200-250 g) received a subplantar injection of carrageenan (1% in saline) into the right hind paw. Rofecoxib was suspended in 0.5% methylcellulose and administered orally 1 hour before carrageenan injection at doses of 1, 3, or 10 mg/kg. Paw volume was measured using a plethysmometer at 0, 2, 4, and 6 hours post-carrageenan. 2) Adjuvant-induced arthritis: Rats received a subcutaneous injection of Freund’s complete adjuvant (0.1 mL) into the left hind paw. Rofecoxib (10 mg/kg, oral) was administered daily from day 1 to day 21, and paw swelling was measured twice weekly [1] - Mouse OIR model: C57BL/6 mice (P7 pups with dams) were exposed to 75% oxygen for 5 days (P7-P12), then returned to room air to induce retinal neovascularization. Rofecoxib was dissolved in DMSO (final concentration <0.1%) and diluted in saline, then administered orally via gavage at 5 or 10 mg/kg/day from P12 to P17 (once daily). On P17, mice were euthanized, eyes were enucleated, and retinas were isolated for immunofluorescence staining (using isolectin B4 to label blood vessels) [2] - Mesothelioma xenograft model: Female nude mice (6-8 weeks old) were subcutaneously injected with H2052 cells (5×10^6 cells in 0.2 mL PBS/matrigel) into the right flank. When tumors reached ~100 mm³, mice were randomized into 4 groups: vehicle (0.5% methylcellulose, oral), Rofecoxib (10 mg/kg, oral), gefitinib (25 mg/kg, oral), or combination. Drugs were administered daily for 21 days. Tumor volume was measured every 3 days using calipers (volume = length × width² / 2), and body weight was recorded weekly [3] |
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
The mean oral bioavailability of rofecoxib at the recommended therapeutic doses of 12.5 mg, 25 mg, and 50 mg is approximately 93%. Of the radiolabeled rofecoxib dose, approximately 72% is excreted in the urine as metabolites and 14% is excreted unchanged in the feces. Time to peak concentration: approximately 2 to 3 hours. At steady state, the apparent volume of distribution after administration of 12.5 mg and 25 mg doses is approximately 91 L and 86 L, respectively. The mean oral bioavailability at the recommended dose is 93%. Within the clinical dose range, peak plasma concentration is approximately proportional to the area under the plasma concentration-time curve. For more complete data on absorption, distribution, and excretion of rofecoxib (11 items in total), please visit the HSDB record page. Metabolism/Metabolites Hepatic metabolism. The metabolism of rofecoxib is primarily mediated by the reductive action of cytoplasmic enzymes. The main metabolites are cis- and trans-dihydro derivatives of rofecoxib, accounting for approximately 56% of the radioactive recovery in urine. An additional 8.8% of the dose is recovered as glucuronides of hydroxyl derivatives, products of oxidative metabolism. The biotransformation of rofecoxib and its metabolites in humans is reversible to a limited extent (<5%). These metabolites do not possess COX-1 or COX-2 inhibitory activity. Cytochrome P450 plays a minor role in the metabolism of rofecoxib. This study investigated the metabolism of rofecoxib (a potent and selective cyclooxygenase-2 inhibitor) in vitro using human liver subcellular components. The biotransformation of rofecoxib is highly dependent on the subcellular components and the redox system used. In hepatic microsomal incubation, rofecoxib primarily oxidizes to 5-hydroxyrofecoxib via NADPH-dependent oxidation; while in cytoplasmic incubation, rofecoxib primarily oxidizes to 3,4-dihydrohydroxy acid metabolites via NADPH-dependent reduction. Metabolites from both oxidative and reductive pathways were observed in S9 fraction incubation. Unlike microsomal incubation, the oxidation of rofecoxib to 5-hydroxyrofecoxib in S9 fraction follows two pathways: one NADPH-dependent and the other NAD+ (non-cytochrome P450), with the latter accounting for approximately 40% of the total activity. The generated 5-hydroxyrofecoxib undergoes NADPH-dependent reduction (“reverse reduction”) upon incubation with hepatic cytoplasmic fractions, regenerating rofecoxib. Upon incubation with dialysis hepatic cytoplasm, rofecoxib was observed to undergo net hydration to 3,4-dihydro-5-hydroxyrofecoxib; however, in the presence of NADPH, a 3,4-dihydrohydroxy acid derivative was generated. Although 3,4-dihydro-5-hydroxyrofecoxib can be reduced to 3,4-dihydrohydroxy acid in the presence of NADPH via the cytosol, the former does not appear to be an intermediate in the overall reductive metabolic pathway of rofecoxib. Net reductive metabolism was superior to oxidative metabolism after incubation with the S9 component for more than 2 hours. These in vitro results are consistent with previous studies on the metabolism of rofecoxib in humans and provide valuable insights into the mechanisms of this drug's complex metabolism. The metabolism of rofecoxib is primarily mediated by the reductive action of cytosol enzymes. Its major metabolites are the cis- and trans-dihydro derivatives of rofecoxib, accounting for approximately 56% of the radioactive recovery in urine. Another 8.8% of the dose is recovered as glucuronides of the hydroxy derivatives, which are products of oxidative metabolism. The biotransformation of rofecoxib and its metabolites in humans is reversible to a limited extent (<5%). These metabolites do not possess COX-1 or COX-2 inhibitory activity. Known metabolites of rofecoxib include 5-hydroxyrofecoxib. Biological half-life 17 hours Approximately 17 hours. Oral absorption: In rats, oral administration of rofecoxib (10 mg/kg) showed rapid absorption, with a peak plasma concentration (Cmax) of approximately 2.5 μg/mL, reached at 1 hour (Tmax). Oral bioavailability was approximately 90%, compared to intravenous administration [1] -Plasma protein binding: In human plasma, rofecoxib exhibits high protein binding (approximately 97%), primarily binding to albumin; the binding rate is concentration-independent in the concentration range of 0.1–10 μg/mL [1] -Elimination: In dogs, the plasma half-life (t1/2) of rofecoxib is approximately 11 hours; it is primarily eliminated through metabolism (oxidation and glucuronidation), with less than 5% of the dose excreted unchanged in the urine [1] |
| 毒性/毒理 (Toxicokinetics/TK) |
Hepatotoxicity
In thousands of clinical studies involving at least 3 months of treatment, the incidence of serum ALT elevations exceeding three times the upper limit of normal was 1.8% in the rofecoxib treatment group, compared to 0.3% in the placebo group and 0.1-0.4% in other commonly used nonsteroidal anti-inflammatory drugs (NSAIDs). Therefore, rofecoxib-induced ALT elevations are uncommon and usually clinically insignificant, resolving spontaneously with continued use. In rare cases, rofecoxib can cause clinically significant, symptomatic drug-induced liver injury, accompanied by jaundice. The pattern of elevated liver enzymes is typically cholestatic or mixed (Case 1), but hepatocellular injury has also been reported. The latency period for liver injury varies greatly, ranging from weeks to years, but usually appears within 1 to 12 weeks of starting treatment. Autoimmune and immune hypersensitivity features are uncommon. Probability score: C (likely a rare cause of clinically significant liver injury). Protein Binding 87% Drug Interactions Rifampin co-administration with rofecoxib may reduce rofecoxib plasma concentrations by 50%;…. In patients receiving methotrexate 7.5 to 15 mg/week, daily administration of rofecoxib 75 mg for 10 days increased plasma methotrexate concentrations by 23%; the effect of the recommended dose of rofecoxib is unclear; therefore, monitoring for methotrexate toxicity is recommended. Aspirin co-administration with rofecoxib may increase the risk of gastrointestinal ulcers or gastrointestinal complications. In elderly patients, co-administration of rofecoxib with antacids containing calcium carbonate, aluminum, or magnesium reduced the area under the plasma concentration-time curve (AUC) by 13% and 8%, respectively; both antacids reduced peak plasma concentrations of rofecoxib by approximately 20%. For more complete data on rofecoxib interactions (14 in total), please visit the HSDB record page. Gastrointestinal toxicity: No significant gastric mucosal damage was observed in rats treated with rofecoxib (10, 30 mg/kg/day, orally) for 28 days; in contrast, indomethacin (a non-selective COX inhibitor) caused severe gastric ulcers at a dose of 5 mg/kg/day[1] - Hepatotoxicity: In mice, no significant changes in serum alanine aminotransferase (ALT) or aspartate aminotransferase (AST) levels were observed after 14 days of treatment with rofecoxib (orally, at doses up to 30 mg/kg/day)[2] - Combination therapy toxicity: In a mesothelioma xenograft model, rofecoxib (10 mg/kg/day) in combination with gefitinib (25 mg/kg/day) did not result in a significant increase in mortality or organ toxicity (assessed by histopathological evaluation of the liver, kidneys and spleen) compared to the monotherapy group[3] |
| 参考文献 |
|
| 其他信息 |
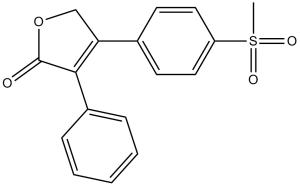
Rofecoxib is a butenolactone compound with the structure furan-2(5H)-one, substituted with a phenyl group at position 3 and a p-(methanesulfonyl)phenyl group at position 4. It is a selective cyclooxygenase-2 inhibitor, used to treat osteoarthritis from 1999 to 2004, but was withdrawn from the market due to concerns about its potential to increase the risk of heart attack and stroke. Rofecoxib has a dual role as a cyclooxygenase-2 inhibitor, a nonsteroidal anti-inflammatory drug (NSAID), and an analgesic. It is a sulfone butenolactone compound. Rofecoxib is used to treat osteoarthritis, rheumatoid arthritis, acute pain in adults, primary dysmenorrhea, and acute migraine attacks with or without aura. Rofecoxib is a solid. This compound belongs to the stilbene class. These are organic compounds containing a 1,2-stilbene moiety. Stilbene compounds (C6-C2-C6) are derived from the common styrene (C6-C3) skeletal unit. Introducing one or more hydroxyl groups onto the benzene ring yields stilbene compounds. Rofecoxib has a half-life of 17 hours and an average oral bioavailability of approximately 93% at recommended therapeutic doses of 125, 25, and 50 mg. Rofecoxib targets proteins including elastin and prostaglandin G/H synthase 2. Cytochrome P450 1A2, cytochrome P450 3A4, cytochrome P450 2C9, cytochrome P450 2C8, and prostaglandin G/H synthase 1 are known to metabolize rofecoxib. On September 30, 2004, Merck withdrew rofecoxib from the market due to the potential increase in the risk of heart attack and stroke with long-term high-dose use. Rofecoxib is a nonsteroidal anti-inflammatory drug (NSAID) that selectively inhibits cyclooxygenase-2 (Cox-2) and was previously used to treat chronic arthritis and mild to moderate musculoskeletal pain. In 2004, it was withdrawn from the market due to the association of long-term use with an increased risk of cardiovascular events. In addition, rofecoxib has been associated with transient elevations in serum transaminases during treatment and rare drug-induced liver disease. Rofecoxib is a synthetic nonsteroidal derivative of phenylfuranone with anti-inflammatory, antipyretic, and analgesic effects, and may have antitumor activity. Rofecoxib binds to and inhibits cyclooxygenase-2 (COX-2), thereby inhibiting the conversion of arachidonic acid to prostaglandins. The COX-related metabolic pathway may be a key regulator of cell proliferation and angiogenesis. Certain epithelial tumor cell types overexpress pro-angiogenic COX-2. (NCI04)
Drug Indications For the treatment of osteoarthritis, rheumatoid arthritis, acute pain and primary dysmenorrhea in adults, and acute attacks of migraine with or without aura. FDA Label Mechanism of Action The anti-inflammatory, analgesic, and antipyretic effects of nonsteroidal anti-inflammatory drugs (NSAIDs) appear to be achieved by inhibiting prostaglandin synthesis. Although its exact mechanism of action is not fully understood, these effects appear to be achieved by inhibiting COX-2 isoenzymes at sites of inflammation, thereby reducing the synthesis of certain prostaglandins from their arachidonic acid precursors. Rofecoxib selectively inhibits cyclooxygenase-2 (COX-2), an enzyme that plays a crucial role in mediating inflammation and pain. Unlike non-selective nonsteroidal anti-inflammatory drugs (NSAIDs), rofecoxib does not inhibit platelet aggregation and has very low or almost no affinity for COX-1. Rofecoxib is an NSAID with anti-inflammatory, analgesic, and antipyretic therapeutic effects. Studies have suggested that rofecoxib reduces the production of prostaglandin precursors by inhibiting the activity of cyclooxygenase-2 (COX-2). At therapeutic concentrations, rofecoxib does not inhibit cyclooxygenase-1 (COX-1) isoenzymes in the human body. Therapeutic Use /September 20, 2004/ Merck announced that, due to an increased risk of cardiovascular events (including heart attack and stroke) in patients taking rofecoxib (Vioxx), the company has voluntarily withdrawn rofecoxib (Vioxx) from the U.S. and global markets for safety reasons. Vioxx is a prescription COX-2 selective nonsteroidal anti-inflammatory drug (NSAID) approved by the FDA in May 1999 for the relief of signs and symptoms of osteoarthritis, the treatment of acute pain in adults, and the relief of dysmenorrhea symptoms. It was later approved for the relief of signs and symptoms of rheumatoid arthritis in adults and children. Anti-inflammatory drug. Rofecoxib is indicated for the relief of signs and symptoms of osteoarthritis. /U.S. product label includes/ Rofecoxib is indicated for short-term (5 days) relief of acute pain, especially in cases requiring anti-inflammatory effects, such as after dental or orthopedic surgery. /US product label contains/ Rofecoxib is indicated for short-term (5 days) relief of pain and other symptoms of primary dysmenorrhea. /US product label contains Drug Warning / Merck announced a voluntary withdrawal of Vioxx (rofecoxib) from the US and global markets due to an increased risk of cardiovascular events (including heart attack and stroke) in patients taking Vioxx, posing a safety concern. Vioxx is a prescription COX-2 selective nonsteroidal anti-inflammatory drug (NSAID) approved by the FDA in May 1999 for the relief of signs and symptoms of osteoarthritis, the treatment of acute pain in adults, and the treatment of menstrual symptoms. It was later approved for the relief of signs and symptoms of rheumatoid arthritis in adults and children. Currently, whether cyclooxygenase-2 (COX-2) specific inhibitors cause elevated blood pressure, thus requiring treatment in clinical practice, remains controversial. We conducted a retrospective case-control study of 17,844 subjects aged ≥65 years from two US states to assess the risk of new-onset hypertension. We used a multivariate logistic regression model to assess the relative risk of developing new-onset hypertension requiring treatment in patients taking celecoxib or rofecoxib compared to patients taking other COX-2 specific inhibitors, nonspecific nonsteroidal anti-inflammatory drugs (NSAIDs), or not taking any NSAIDs. During the study period from 1999 to 2000, a total of 3915 patients were diagnosed with hypertension and started treatment; each case was matched with 4 controls. In all models, celecoxib was not significantly associated with the occurrence of hypertension. Patients taking rofecoxib had a significantly increased relative risk of developing new-onset hypertension compared to patients taking celecoxib (odds ratio (OR) 1.6; 95% confidence interval (CI) 1.2 to 2.1), patients taking nonspecific nonsteroidal anti-inflammatory drugs (NSAIDs) (OR 1.4; 95% CI 1.1 to 1.9), or patients not taking NSAIDs (OR 1.6; 95% CI 1.3 to 2.0). No significant effect was found in dosage or duration of treatment. In patients with a history of chronic kidney disease, liver disease, or congestive heart failure, the relative risk of developing new-onset hypertension was twice that of patients taking rofecoxib compared to those taking celecoxib (OR 2.1; 95% CI 1.0 to 4.3). In this retrospective case-control study of patients ≥ 65 years of age, rofecoxib use was associated with an increased relative risk of new-onset hypertension. This was not observed in patients taking celecoxib. In a double-blind study, 35 stable subjects (taking low-dose aspirin, with ≥ 2 previous acute coronary events and two screening CRP values > 2.0 mg/L) were randomized to either the COX-2 inhibitor rofecoxib (25 mg) or placebo, taken daily for 6 months. Serum CRP, interleukin-6 (IL-6), P-selectin, matrix metalloproteinase-9 (MMP-9), and brachial artery endothelial function were assessed. In the placebo group, the median baseline CRP was 3.16 mg/L (25% and 75% quartiles were 1.90 and 5.78 mg/L, respectively), and at 6 months it was 4.22 mg/L (25% and 75% quartiles were 2.04 and 6.25 mg/L, respectively). In the rofecoxib group, the baseline CRP level was 3.45 mg/L (25% and 75% quartiles were 2.08 and 5.78 mg/L, respectively), and at 6 months it was 1.41 mg/L (25% and 75% quartiles were 1.17 and 4.81 mg/L, respectively) (P=0.03). Compared with placebo, rofecoxib also reduced IL-6 levels at 6 months (P=0.0002). After 3 months of treatment, significant discontinuation effects were observed in CRP and IL-6 levels in the rofecoxib group (P=0.005 and P=0.009, respectively). Rofecoxib had no significant effect on P-selectin, MMP-9, or brachial artery vascular reactivity. Long-term COX-2 inhibition reduced CRP and IL-6 levels without affecting P-selectin and MMP-9, and had no adverse effect on endothelial function in stable patients with a history of recurrent acute coronary events and elevated CRP. These results further support the rationale for evaluating the clinical benefit of COX-2 inhibitors in patients with ischemic heart disease. A 73-year-old woman with rheumatoid arthritis was given rofecoxib 25 mg/day in addition to other medications (her condition was stable). Six months after starting rofecoxib, linear plaques appeared in the infraorbital and temporal regions of both eyes. Several pruritic purplish-red papules also appeared on the right wrist and left dorsum of the foot. Pigmentation also appeared on the right buccal mucosa. Because the rash was localized and the patient was initially unwilling to undergo a skin biopsy, rofecoxib was continued and topical steroids were started. One month later, the patient returned to the dermatology clinic with significantly improved skin response. The skin biopsy performed during this visit was consistent with a diagnosis of LDE. The following day, her rheumatologist decided to discontinue the causative drug, rofecoxib. Two months later, all skin lesions had completely resolved. Rofecoxib was not used again. LDE is a rare skin reaction that can be associated with multiple medications. Rofecoxib is a cyclooxygenase-2 inhibitor, and there have been no previous reports of LDE. An objective causality assessment suggests that rofecoxib may have been the cause of the skin reaction. For more complete data on rofecoxib (31 total), please visit the HSDB records page. Pharmacodynamics Rofecoxib is a selective cyclooxygenase-2 (COX-2) inhibitor, belonging to the class of nonsteroidal anti-inflammatory drugs (NSAIDs). Unlike celecoxib, rofecoxib does not contain a sulfonamide chain and its metabolism does not require the CYP450 enzyme. Like other NSAIDs, rofecoxib has anti-inflammatory, analgesic, and antipyretic effects. NSAIDs appear to inhibit prostaglandin synthesis by inhibiting cyclooxygenase (COX), which is responsible for catalyzing the formation of prostaglandins in the arachidonic acid pathway. At least two isoenzymes, COX-1 and COX-2, have been identified. Although the exact mechanism is not fully elucidated, nonsteroidal anti-inflammatory drugs (NSAIDs) exert their anti-inflammatory, analgesic, and antipyretic effects primarily by inhibiting COX-2. Inhibition of COX-1 is the main reason for their adverse effects on the gastrointestinal mucosa. Because rofecoxib is selective for COX-2, it may be associated with a reduced risk of certain adverse events, but more data are needed to fully evaluate the drug. Rofecoxib is a selective COX-2 inhibitor used to treat pain and inflammation caused by diseases such as osteoarthritis and rheumatoid arthritis, with fewer gastrointestinal side effects compared to non-selective NSAIDs [1]. - The inhibitory effect of rofecoxib on retinal neovascularization suggests that it has potential therapeutic value in neovascular eye diseases such as diabetic retinopathy and age-related macular degeneration[2]. - The synergistic effect of rofecoxib and gefitinib in mesothelioma cells supports the exploration of combining COX-2 inhibitors with EGFR inhibitors for the treatment of mesothelioma, especially for patients with high COX-2 expression[3]. |
| 分子式 |
C17H14O4S
|
|---|---|
| 分子量 |
314.36
|
| 精确质量 |
314.061
|
| CAS号 |
162011-90-7
|
| 相关CAS号 |
Rofecoxib-d5;544684-93-7
|
| PubChem CID |
5090
|
| 外观&性状 |
Light yellow to green solid powder
|
| 密度 |
1.3±0.1 g/cm3
|
| 沸点 |
577.6±50.0 °C at 760 mmHg
|
| 熔点 |
207°C
|
| 闪点 |
303.1±30.1 °C
|
| 蒸汽压 |
0.0±1.6 mmHg at 25°C
|
| 折射率 |
1.619
|
| LogP |
1.34
|
| tPSA |
68.82
|
| 氢键供体(HBD)数目 |
0
|
| 氢键受体(HBA)数目 |
4
|
| 可旋转键数目(RBC) |
3
|
| 重原子数目 |
22
|
| 分子复杂度/Complexity |
556
|
| 定义原子立体中心数目 |
0
|
| InChi Key |
RZJQGNCSTQAWON-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C17H14O4S/c1-22(19,20)14-9-7-12(8-10-14)15-11-21-17(18)16(15)13-5-3-2-4-6-13/h2-10H,11H2,1H3
|
| 化学名 |
3-(4-methylsulfonylphenyl)-4-phenyl-2H-furan-5-one
|
| 别名 |
MK-0966; MK0966; MK0966; MK966; MK966; MK 966; MK 0966; Trade name: Vioxx; Ceoxx; Ceeoxx.
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (7.95 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL 澄清 DMSO 储备液加入900 μL 玉米油中,混合均匀。 配方 2 中的溶解度: 30% PEG400+0.5% Tween80+5% propylene glycol: 30mg/mL 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1811 mL | 15.9053 mL | 31.8107 mL | |
| 5 mM | 0.6362 mL | 3.1811 mL | 6.3621 mL | |
| 10 mM | 0.3181 mL | 1.5905 mL | 3.1811 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT04684511 | Terminated | Drug: TRM-201 (Rofecoxib) Drug: Placebo |
Hemophilic Arthropathy | Tremeau Pharmceuticals, Inc. | June 2, 2021 | Phase 3 |
| NCT00038389 | Terminated | Drug: Vioxx | Glioma Brain Neoplasms |
M.D. Anderson Cancer Center | October 2001 | Phase 1 |
| NCT00568295 | Completed | Drug: acetaminophen Drug: Rofecoxib |
Osteoarthritis of the Knee | Johnson & Johnson Consumer and Personal Products Worldwide |
October 1999 | Phase 3 |
| NCT00060476 | Completed | Drug: rofecoxib Drug: Comparator: placebo (unspecified) |
Prostate Cancer | Merck Sharp & Dohme LLC | January 8, 2003 | Phase 3 |
|
|---|
|
|