| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5g |
|
||
| 10g |
|
||
| 25g |
|
||
| 50g |
|
||
| Other Sizes |
|
| 体外研究 (In Vitro) |
在过表达 ENT1 和 ENT2 的 HeLa 细胞中,三乙酸尿苷抑制 [3H] 尿苷吸收,IC50 值分别为 228.4 μM 和 28.4 μM [4]。
|
|---|---|
| 体内研究 (In Vivo) |
每 8 小时口服一次尿苷三乙酸酯 (2 g/kg),总共 15 剂,已被证明可以提高 5-FU 过量小鼠的存活率并降低毒性 [3]。每 8 小时口服一次尿苷三乙酸酯 (2 g/kg),共 15 剂,已显示可降低 5-FU 毒性并提高 DPD 缺陷小鼠的存活率 [3]。
|
| 动物实验 |
动物/疾病模型: 5-FU 过量(腹腔注射,300 mg/kg)BALB/c 小鼠模型 [3]
剂量: 2 g/kg 给药途径: 灌胃(po),每 8 小时一次,共 15 次。 实验结果: 分别在 24、48、72、96、120 和 144 小时开始给药的各组存活率分别提高至 90%、60%、30%、20%、0% 和 0%。 动物/疾病模型: 5-乙炔基尿嘧啶诱导(腹腔注射,2 mg/kg)DPD(二氢嘧啶脱氢酶)缺陷小鼠模型[3] 剂量: 2 g/kg 给药途径: 灌胃(po),每8小时一次,共15次。 实验结果: 5-FU给药后4小时,存活率提高至100%;24小时内,存活率提高至80%。在 48、72、96、120 和 144 小时内开始治疗的各组患者的生存率分别提高到 40%、50%、20%、30% 和 0%。 |
| 药代性质 (ADME/PK) |
吸收、分布和排泄
口服给药后,血浆中尿苷的最大浓度通常在 2 至 3 小时内达到。 尿苷可通过肾脏排泄,也可在大多数组织中通过正常的嘧啶分解代谢途径进行代谢。 循环中的尿苷通过特定的核苷转运蛋白被哺乳动物细胞摄取,并可穿过血脑屏障。 代谢/代谢物 口服给药后,尿苷三乙酸酯在体内非特异性酯酶的作用下脱乙酰化,生成尿苷进入血液循环。 生物半衰期 2 至 2.5 小时 |
| 参考文献 |
[1]. Ma WW, et al. Emergency use of uridine triacetate for the prevention and treatment of life-threatening 5-fluorouracil and capecitabine toxicity. Cancer. 2017 Jan 1;123(2):345-356.
[2]. Cada DJ, et al. Uridine Triacetate. Hosp Pharm. 2016 Jun;51(6):484-8. [3]. Rolando A G Garcia, et al. Prompt treatment with uridine triacetate improves survival and reduces toxicity due to fluorouracil and capecitabine overdose or dihydropyrimidine dehydrogenase deficiency. Toxicol Appl Pharmacol. 2018 Aug 15;353:67-73. [4]. Siennah R Miller, et al. Predicting Drug Interactions with Human Equilibrative Nucleoside Transporters 1 and 2 Using Functional Knockout Cell Lines and Bayesian Modeling. Mol Pharmacol. 2021 Feb;99(2):147-162. |
| 其他信息 |
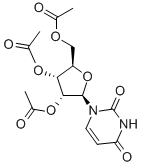
尿苷三乙酸酯是一种乙酸酯,是尿嘧啶的一种衍生物,其中三个羟基氢被乙酸基团取代。作为尿苷的前药,它用于治疗遗传性乳清酸尿症和控制氟尿嘧啶中毒。它具有多种功能,包括作为前药、神经保护剂和孤儿药。它属于尿苷类化合物,是一种乙酸酯。
尿苷三乙酸酯,曾用名维斯托尿苷,是一种口服有效的天然核苷尿苷的前药。它用于治疗遗传性乳清酸尿症(商品名:Xuriden),或用于紧急治疗氟尿嘧啶或卡培他滨过量或中毒(商品名:Vistogard)。它以尿苷三乙酸酯的形式给药,因为与等摩尔剂量的尿苷相比,这种形式能使进入体循环的尿苷量增加4至6倍。当用于治疗或预防氟尿嘧啶和其他抗代谢药物相关毒性时,尿苷三乙酸酯的作用机制是与5-氟尿嘧啶(5-FU)代谢物竞争掺入非癌细胞的遗传物质。它能降低两种细胞毒性中间体——5-氟-2'-脱氧尿苷-5'-单磷酸(FdUMP)和5-氟尿苷三磷酸(FUTP)——相关的毒性和细胞死亡。通常情况下,FdUMP抑制胸苷酸合成酶,而胸苷酸合成酶是胸苷合成以及DNA复制和修复所必需的;FUTP则掺入RNA,导致RNA链缺陷。因此,这些代谢物会引起多种不良反应,例如中性粒细胞减少症、黏膜炎、腹泻和手足综合征。与其他许多抗肿瘤药物一样,这些副作用限制了5-FU的给药剂量,进而影响治疗效果。通过预先给予尿苷(以前药尿苷三乙酸酯的形式),可以给予更高剂量的5-氟尿嘧啶(5-FU),从而提高疗效并减少毒副作用。如果在开始治疗后96小时内出现严重副作用,尿苷三乙酸酯也可作为抢救治疗。尿苷三乙酸酯还用于治疗遗传性乳清酸尿症,也称为尿苷单磷酸合成酶缺乏症。这种罕见的先天性常染色体隐性遗传性嘧啶代谢障碍是由尿苷单磷酸合成酶(UMPS)缺陷引起的。UMPS是一种双功能酶,催化嘧啶从头合成途径的最后两个步骤。由于UMPS缺乏,患者会出现全身性嘧啶核苷酸缺乏,这解释了该疾病的大部分症状。此外,从头合成嘧啶途径产生的乳清酸无法转化为尿苷单磷酸(UMP),会随尿液排出,这也是该疾病俗称“乳清酸尿症”的原因。而且,尿液中的乳清酸结晶可引起梗阻性尿路疾病发作。当以尿苷三乙酸酯前药的形式给药时,几乎所有细胞都能利用尿苷合成尿苷核苷酸,从而补偿遗传性乳清酸尿症患者合成尿苷核苷酸的遗传缺陷。当细胞内尿苷核苷酸水平恢复正常时,反馈抑制作用会减少乳清酸的过度生成,从而降低尿液中乳清酸的排泄。 尿苷三乙酸酯是一种合成的尿苷前药,可在体内转化为尿苷。尿苷是一种嘧啶核苷酸,已被用于治疗多种疾病,包括抑郁症和遗传性肌病。 (NCI04) 药物适应症 尿苷三乙酸酯以商品名 Xuriden(FDA 批准)上市,用于治疗遗传性乳清酸尿症。尿苷三乙酸酯以商品名 Vistogard(FDA 批准)上市,用于以下情况下成人和儿童患者的紧急治疗:氟尿嘧啶或卡培他滨过量用药后,无论是否存在症状;或在氟尿嘧啶或卡培他滨给药结束后 96 小时内出现影响心脏或中枢神经系统的早期发作、严重或危及生命的毒性反应,和/或早期发作、异常严重的不良反应(例如,胃肠道毒性和/或中性粒细胞减少症)。 FDA 标签 作用机制 尿苷三乙酸酯是一种合成的尿苷前药,可在体内转化为尿苷。尿苷三乙酸酯用于治疗或预防氟尿嘧啶和其他抗代谢药物相关的毒性时,其作用机制在于能够与5-氟尿嘧啶(5-FU)代谢物竞争性地掺入非癌细胞的遗传物质中。它能降低两种细胞毒性中间体——5-氟-2'-脱氧尿苷-5'-单磷酸(FdUMP)和5-氟尿苷三磷酸(FUTP)——相关的毒性和细胞死亡。通过预先给予尿苷(以前药尿苷三乙酸酯的形式),可以给予更高剂量的5-FU,从而提高疗效并减少中性粒细胞减少症、黏膜炎、腹泻和手足综合征等毒副作用。尿苷三乙酸酯还用于替代疗法,治疗遗传性乳清酸尿症,也称为尿苷单磷酸合成酶(UMPS)缺乏症。由于尿嘧啶核苷酸合成酶(UMPS)缺陷,患者会出现全身性嘧啶核苷酸缺乏,这解释了该疾病的大部分症状。此外,从头合成嘧啶途径产生的乳清酸无法转化为尿嘧啶核苷酸(UMP),会随尿液排出,这也是该疾病俗称“乳清酸尿症”的原因。而且,尿液中的乳清酸结晶可引起梗阻性尿路疾病发作。当以尿苷三乙酸酯(一种前药)的形式给药时,几乎所有细胞都可以利用尿苷合成尿嘧啶核苷酸,从而补偿遗传性乳清酸尿症患者合成方面的遗传缺陷。 |
| 分子式 |
C15H18N2O9
|
|---|---|
| 分子量 |
370.3114
|
| 精确质量 |
370.101
|
| CAS号 |
4105-38-8
|
| PubChem CID |
20058
|
| 外观&性状 |
White to off-white solid powder
|
| 密度 |
1.4±0.1 g/cm3
|
| 熔点 |
124-134ºC
|
| 折射率 |
1.552
|
| LogP |
-0.11
|
| tPSA |
142.99
|
| 氢键供体(HBD)数目 |
1
|
| 氢键受体(HBA)数目 |
9
|
| 可旋转键数目(RBC) |
8
|
| 重原子数目 |
26
|
| 分子复杂度/Complexity |
660
|
| 定义原子立体中心数目 |
4
|
| SMILES |
O1[C@]([H])(C([H])([C@]([H])([C@@]1([H])C([H])([H])OC(C([H])([H])[H])=O)OC(C([H])([H])[H])=O)OC(C([H])([H])[H])=O)N1C([H])=C([H])C(N([H])C1=O)=O
|
| InChi Key |
AUFUWRKPQLGTGF-FMKGYKFTSA-N
|
| InChi Code |
InChI=1S/C15H18N2O9/c1-7(18)23-6-10-12(24-8(2)19)13(25-9(3)20)14(26-10)17-5-4-11(21)16-15(17)22/h4-5,10,12-14H,6H2,1-3H3,(H,16,21,22)/t10-,12-,13-,14-/m1/s1
|
| 化学名 |
[(2R,3R,4R,5R)-3,4-diacetyloxy-5-(2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methyl acetate
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ≥ 100 mg/mL (~270.04 mM)
H2O : ~10 mg/mL (~27.00 mM) |
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (6.75 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (6.75 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.5 mg/mL (6.75 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 配方 4 中的溶解度: 24 mg/mL (64.81 mM) in PBS (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶. 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7004 mL | 13.5022 mL | 27.0044 mL | |
| 5 mM | 0.5401 mL | 2.7004 mL | 5.4009 mL | |
| 10 mM | 0.2700 mL | 1.3502 mL | 2.7004 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。