| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1mg |
|
||
| 5mg |
|
||
| Other Sizes |
|
| 靶点 |
In invertebrates, the primary targets are glutamate-gated and gamma-aminobutyric acid (GABA)-gated chloride channels, leading to increased chloride ion conductance and subsequent paralysis in parasites. [2]
For its broad-spectrum antiviral activity, key targets include the inhibition of importin α/β-mediated nuclear transport, which affects the nuclear trafficking of viral proteins. Specific viral targets mentioned include the non-structural 3 helicase (NS3 helicase) of flaviviruses, the nuclear import of HIV-1 integrase and dengue virus NS5 polymerase, the nuclear import of UL42 protein of pseudorabies virus, and the nuclear localization signal-mediated import of the capsid protein (Cap) of porcine circovirus 2. [2] |
|---|---|
| 体外研究 (In Vitro) |
伊维菌素由伊维菌素B1a(不低于80%)和伊维菌素B1b(不超过20%)两种同源分子的混合物组成,是大环内酯类阿维菌素的成员。在剂量为 0.3 μg/ml 时,伊维菌素的主要成分伊维菌素 B1a 是惰性的,而在相同剂量下,次要成分伊维菌素 B1b 会导致蜗牛 100% 死亡[1]。
在0.3 μg/ml的浓度下,伊维菌素B1a对成年光滑双脐螺(Biomphalaria glabrata)无效,死亡率为0%。这与次要成分伊维菌素B1b形成对比,后者在相同浓度下导致100%的螺死亡。[1] 伊维菌素(该药物,其中伊维菌素B1a是主要成分)在体外对多种RNA和DNA病毒表现出广谱抗病毒活性。[2] 对于SARS-CoV-2,本文献引用的一项研究报告称,用伊维菌素处理受感染的Vero-hSLAM细胞后,48小时后病毒RNA减少了约5000倍。[2] 针对新城疫病毒,伊维菌素在100 μg/ml浓度下显示出强抗病毒活性,但在原代鸡成纤维细胞中表现出细胞毒性。[2] 体外抗病毒作用也有报道针对寨卡病毒、甲型流感病毒、西尼罗河病毒、委内瑞拉马脑炎病毒、猪繁殖与呼吸综合征病毒、基孔肯雅病毒、HIV-1、黄热病病毒、登革热病毒、日本脑炎病毒、蜱传脑炎病毒、伪狂犬病病毒、猪圆环病毒2型、细小病毒和牛疱疹病毒1型等。[2] |
| 体内研究 (In Vivo) |
伊维菌素的体内抗病毒潜力已有报道针对西尼罗河病毒和新城疫病毒。[2]
然而,在Ifnar1基因敲除小鼠模型中,伊维菌素未能有效预防致命的寨卡病毒感染。[2] 体内活性也有报道针对伪狂犬病病毒和细小病毒。[2] |
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
Absorbed well. High-fat meals can improve absorption. Ivermectin is metabolized in the liver, and almost all of it and its metabolites are excreted in the feces, expected to be completed within 12 days. Less than 1% of the administered dose is excreted in the urine. The volume of distribution is 3–3.5 L/kg, and it does not cross the blood-brain barrier. Metabolism/Metabolites Primarily metabolized in the liver. Ivermectin and its metabolites are almost entirely excreted in the feces, expected to be completed within 12 days. Less than 1% of the administered dose is excreted in the urine. Biological Half-Life The half-life of ivermectin after oral administration is approximately 18 hours. |
| 毒性/毒理 (Toxicokinetics/TK) |
Hepatotoxicity
A single dose of ivermectin was associated with a low incidence of elevated serum transaminases. One case of clinically significant liver injury following ivermectin use (Case 1) has been reported. This patient developed liver injury one month after a single dose, characterized by hepatocellular elevations of serum enzymes, but without jaundice. The patient recovered rapidly and completely. Elevated serum transaminases are not uncommon in trials of ivermectin for the prevention of SARS-CoV-2 infection and for improving the course of early and severe COVID-19, but their incidence was not significantly different between patients treated with ivermectin and those treated with placebo or control drugs. Probability Score: D (Possibly a rare cause of mild, clinically significant liver injury). Protein binding 93% In the molluscicidal activity test against Biomphalaria glabrata, ivermectin B1a at a concentration of 0.3 μg/ml showed no toxicity, and the mortality rate during the observation period was 0%. [1] |
| 参考文献 | |
| 其他信息 |
Pharmacodynamics
Ivermectin is a semi-synthetic anthelmintic. It belongs to the avermectin class, a class of pentacyclic sixteen-membered lactones (i.e., macrocyclic lactone disaccharides) derived from the soil bacterium Streptomyces avermitilis. Avermectin drugs are potent and broad-spectrum antiparasitic drugs. Ivermectin B1a is the main component (approximately 80%) of commercially available avermectin, an antiparasitic drug. [1] It is a 22,23-dihydro derivative of avermectin B1a. [1] In this study evaluating the snail-killing activity of avermectin against snails, intermediate hosts of schistosomiasis, avermectin B1a was found to be inactive, while the minor component avermectin B1b was identified as the bioactive component causing snail death. [1] Ivermectin B1a is a component of ivermectin, a member of the avermectin family of compounds produced by Streptomyces avermitilis. Ivermectin is a 22,23-dihydro derivative of avermectin B1. [2] It is a drug approved by the U.S. Food and Drug Administration (FDA) and widely used for anthelmintic and antiparasitic treatment in humans and animals. [2] Its broad-spectrum antiviral activity is primarily attributed to its ability to inhibit nuclear transport mediated by nuclear import proteins α/β, thereby blocking the nuclear import of viral proteins essential for viral replication. [2] It has been proposed for the treatment of COVID-19, possibly in combination with hydroxychloroquine, but its clinical efficacy and safety for this indication need to be validated through randomized controlled trials. [2] Due to challenges in formulation, such as high cytotoxicity and low solubility, research has begun on delivery systems such as liposomes to improve their properties. [2] |
| 分子式 |
C48H74O14
|
|---|---|
| 分子量 |
875.09
|
| 精确质量 |
874.508
|
| CAS号 |
71827-03-7
|
| PubChem CID |
6321424
|
| 外观&性状 |
White to light yellow solid powder
|
| LogP |
5.601
|
| tPSA |
170.06
|
| 氢键供体(HBD)数目 |
3
|
| 氢键受体(HBA)数目 |
14
|
| 可旋转键数目(RBC) |
8
|
| 重原子数目 |
62
|
| 分子复杂度/Complexity |
1680
|
| 定义原子立体中心数目 |
20
|
| SMILES |
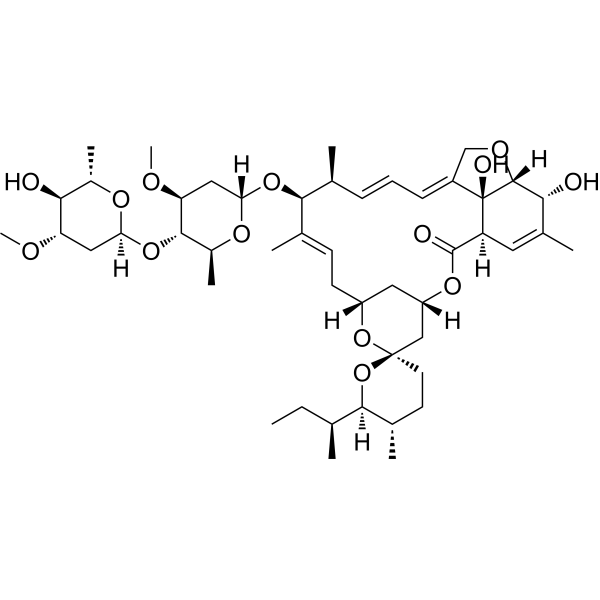
CC[C@H](C)[C@@H]1[C@H](CC[C@@]2(O1)C[C@@H]3C[C@H](O2)C/C=C(/[C@H]([C@H](/C=C/C=C/4\CO[C@H]5[C@@]4([C@@H](C=C([C@H]5O)C)C(=O)O3)O)C)O[C@H]6C[C@@H]([C@H]([C@@H](O6)C)O[C@H]7C[C@@H]([C@H]([C@@H](O7)C)O)OC)OC)\C)C
|
| InChi Key |
AZSNMRSAGSSBNP-XPNPUAGNSA-N
|
| InChi Code |
InChI=1S/C48H74O14/c1-11-25(2)43-28(5)17-18-47(62-43)23-34-20-33(61-47)16-15-27(4)42(26(3)13-12-14-32-24-55-45-40(49)29(6)19-35(46(51)58-34)48(32,45)52)59-39-22-37(54-10)44(31(8)57-39)60-38-21-36(53-9)41(50)30(7)56-38/h12-15,19,25-26,28,30-31,33-45,49-50,52H,11,16-18,20-24H2,1-10H3/b13-12+,27-15+,32-14+/t25-,26-,28-,30-,31-,33+,34-,35-,36-,37-,38-,39-,40+,41-,42-,43+,44-,45+,47+,48+/m0/s1
|
| 化学名 |
(1R,4S,5'S,6R,6'R,8R,10E,12S,13S,14E,16E,20R,21R,24S)-6'-[(2S)-butan-2-yl]-21,24-dihydroxy-12-[(2R,4S,5S,6S)-5-[(2S,4S,5S,6S)-5-hydroxy-4-methoxy-6-methyloxan-2-yl]oxy-4-methoxy-6-methyloxan-2-yl]oxy-5',11,13,22-tetramethylspiro[3,7,19-trioxatetracyclo[15.6.1.14,8.020,24]pentacosa-10,14,16,22-tetraene-6,2'-oxane]-2-one
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : 100 mg/mL (114.27 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (2.86 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (2.86 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL 澄清 DMSO 储备液加入到 900 μL 玉米油中并混合均匀。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.1427 mL | 5.7137 mL | 11.4274 mL | |
| 5 mM | 0.2285 mL | 1.1427 mL | 2.2855 mL | |
| 10 mM | 0.1143 mL | 0.5714 mL | 1.1427 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
Insecticide Resistance Management in Burkina Faso and Côte D'Ivoire
CTID: NCT03074435
Phase: Phase 3 Status: Completed
Date: 2024-10-01