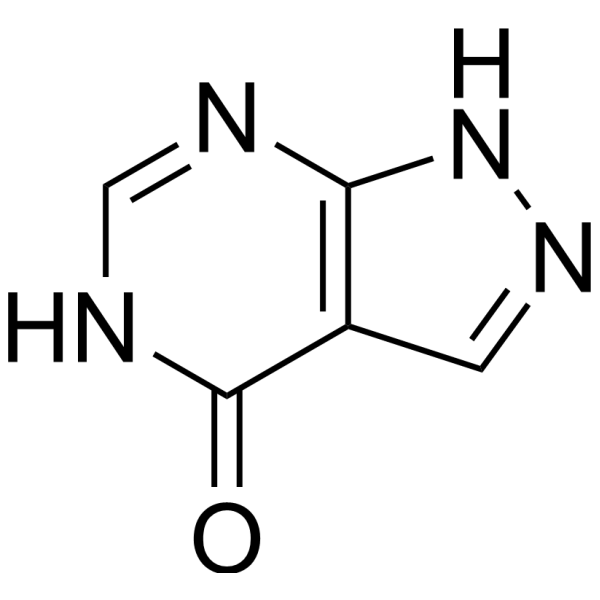
别嘌呤醇是一种有效的口服生物活性黄嘌呤氧化酶抑制剂(拮抗剂),IC50 为 0.2-50 μM。别嘌呤醇用于研究高尿酸血症和痛风。别嘌呤醇可降低 HIF-1α 和 HIF-2α 蛋白表达。别嘌呤醇具有抗抑郁和抗伤害活性。别嘌呤醇具有抗利什曼活性。
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
Time-limited Triplet Combination of Pirtobrutinib, Venetoclax, and Obinutuzumab for Patients With Treatment-naïve Chronic Lymphocytic Leukemia (CLL) or Richter Transformation (RT)
CTID: NCT05536349
Phase: Phase 2 Status: Recruiting
Date: 2024-11-25
Chemotherapy in Patients With Relapsed Small Cell Lung Cancer in Combination With Allopurinol and MycoPhenolate (CLAMP Trial)
CTID: NCT05049863
Phase: Phase 1 Status: Active, not recruiting
Date: 2024-11-18
Phase 2 to Assess Efficacy and Safety in AR882 Alone or in Combination with Allopurinol in Patients with Tophaceous Gout
CTID: NCT05253833
Phase: Phase 2 Status: Completed
Date: 2024-11-13
Inotuzumab Ozogamicin and Frontline Chemotherapy in Treating Young Adults With Newly Diagnosed B Acute Lymphoblastic Leukemia
CTID: NCT03150693
Phase: Phase 3 Status: Suspended
Date: 2024-10-26
Allopurinol Improves Heart Function in African Americans With Resistant Hypertension
CTID: NCT05888233
Phase: Phase 2 Status: Recruiting
Date: 2024-10-15
View More
Treat-to-Target Serum Urate Versus Treat-to-Avoid Symptoms in Gout
CTID: NCT04875702
Phase: Phase 4 Status: Recruiting
Date: 2024-10-01
Phase 2b/3 Study to Assess ABP-671 a Novel URAT1 Inhibitor in Participants With Gout
CTID: NCT05818085
Phase: Phase 2/Phase 3 Status: Active, not recruiting
Date: 2024-09-19
Extension Study of ABP-671 in Participants With Gout
CTID: NCT06276556
Phase: Phase 2/Phase 3 Status: Terminated
Date: 2024-09-19
Phase II Study to Evaluate Efficacy and Safety of D-0120 in Combination With Allopurinol in Subjects With Gout
CTID: NCT05665699
Phase: Phase 2 Status: Recruiting
Date: 2024-08-15
Cerebrum and Cardiac Protection With Allopurinol in Neonates With Critical Congenital Heart Disease Requiring Cardiac Surgery With Cardiopulmonary Bypass
CTID: NCT04217421
Phase: Phase 3 Status: Recruiting
Date: 2024-05-16
Uric Acid, Klotho and Salt Sensitivity in Young Adults Born Preterm
CTID: NCT04026776
PhaseEarly Phase 1 Status: Recruiting
Date: 2024-05-09
Allo HSCT Using RIC and PTCy for Hematological Diseases
CTID: NCT05805605
Phase: Phase 2 Status: Recruiting
Date: 2024-05-07
Effect of Allopurinol and Febuxostat on Urinary 2,8-Dihydroxyadenine Excretion
CTID: NCT02752633
Phase: Phase 4 Status: Completed
Date: 2024-04-09
Allopurinol and Quality of Life in Liver Cirrhosis
CTID: NCT05828836
Phase: Phase 2/Phase 3 Status: Completed
Date: 2023-10-31
Tigulixostat, Phase 3 Study, Allopurinol Controlled in Gout Patients
CTID: NCT05586971
Phase: Phase 3 Status: Recruiting
Date: 2023-10-18
The Effect of Allopurinol on the Risk of Cardiovascular Events in Patients With Cardiovascular Risk
CTID: NCT05943821
Phase: Phase 3 Status: Recruiting
Date: 2023-10-12
Open Label PK, PD and DDI of Dotinurad and Allopurinol in Gout Patients With Hyperuricemia
CTID: NCT06056570
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2023-09-28
Allo HSCT Using RIC for Hematological Diseases
CTID: NCT02661035
Phase: Phase 2 Status: Completed
Date: 2023-09-21
A Multicentre, Randomized, Double-blind, Allopurinol Controlled Study to Evaluate the Efficacy and Safety of SHR4640 in Subjects With Gout
CTID: NCT04956432
Phase: Phase 3 Status: Active, not recruiting
Date: 2023-08-29
Effect of Allopurinol on Markers of Mineral and Bone Metabolism
CTID: NCT05601271
Phase: N/A Status: Active, not recruiting
Date: 2023-08-29
Rituximab, Chemotherapy, and Filgrastim in Treating Patients With Burkitt's Lymphoma or Burkitt's Leukemia
CTID: NCT00039130
Phase: Phase 2 Status: Completed
Date: 2023-08-21
Data Analysis for Drug Repurposing for Effective Alzheimer's Medicines - (DREAM) Probenecid vs Allopurinol
CTID: NCT04746989
Phase: Status: Active, not recruiting
Date: 2023-07-18
Effect of Allopurinol for Hypoxic-ischemic Brain Injury on Neurocognitive Outcome
CTID: NCT03162653
Phase: Phase 3 Status: Recruiting
Date: 2023-07-11
Sex-related Differences in Arterial Stiffness in Type 2 Diabetics: Role of Uric Acid
CTID: NCT03648996
Phase: Phase 2 Status: Completed
Date: 2023-07-03
Study of Verinurad in Heart Failure With Preserved Ejection Fraction
CTID: NCT04327024
Phase: Phase 2 Status: Completed
Date: 2023-06-29
A Study in Healthy Subjects to Assess Drug Availability of 4 Different Formulations of Verinurad and Allopurinol
CTID: NCT04550234
Phase: Phase 1 Status: Completed
Date: 2023-05-10
Allopurinol to Prevent Cirrhosis Related Morbidities
CTID: NCT05545670
Phase: Phase 2/Phase 3 Status: Completed
Date: 2023-05-09
Allopurinol Versus Atorvastatin to Prevent Complications of Liver Cirrhosis
CTID: NCT05511766
Phase: Phase 2/Phase 3 Status: Recruiting
Date: 2023-04-13
Pharmacokinetics of Verinurad and Allopurinol in Combination With Cyclosporine and Rifampicin in Healthy Volunteers
CTID: NCT04532918
Phase: Phase 1 Status: Completed
Date: 2023-03-27
Treatment of Hyperuricemia With Rasburicase in Patients With Acute Lymphoblastic Leukemia or High Grade Lymphoma
CTID: NCT00199043
Phase: Phase 3 Status: Completed
Date: 2023-03-17
A Study of Verinurad and Allopurinol in Patients With Chronic Kidney Disease and Hyperuricaemia
CTID: NCT03990363
Phase: Phase 2 Status: Completed
Date: 2023-03-02
Allopurinol and Trimetazidine as a Preventive of Acute Kidney Injury in PCI Patients
CTID: NCT05540184
Phase: Phase 4 Status: Recruiting
Date: 2023-01-19
Evaluation of a Treatment With Allopurinol in Adenylosuccinate Lyase Deficiency
CTID: NCT03776656
Phase: Phase 2 Status: Completed
Date: 2022-10-12
A Trial Comparing Single Agent Melphalan to Carmustine, Etoposide, Cytarabine, and Melphalan (BEAM) as a Preparative Regimen for Patients With Multiple Myeloma Undergoing High Dose Therapy Followed by Autologous Stem Cell Reinfusion
CTID: NCT03570983
Phase: Phase 2 Status: Unknown status
Date: 2022-06-22
A Pilot Study of Allopurinol As A Modifier of 6-MP Metabolism in Pediatric ALL
CTID: NCT02046694
PhaseEarly Phase 1 Status: Completed
Date: 2022-06-21
A Study of Evaluating the Efficacy and Safety of SHR4640 in Subjects With Gout
CTID: NCT04052932
Phase: Phase 3 Status: Completed
Date: 2022-06-13
Allopurinol Effect on MDA,NO,KIM-1 Urine Levels, RI and Renal Elastography in Kidney Stone Patients Post ESWL
CTID: NCT05414669
Phase: Phase 4 Status: Completed
Date: 2022-06-10
Alemtuzumab and Combination Chemotherapy in Treating Patients With Untreated Acute Lymphoblastic Leukemia
CTID: NCT00061945
Phase: Phase 1/Phase 2 Status: Completed
Date: 2022-05-03
PERL Continuous Glucose Monitoring (CGM) Study
CTID: NCT03334318
Phase: Status: Completed
Date: 2022-03-29
A Study to Assess the Effect of Verinurad on the Electric Activity of the Heart
CTID: NCT04256629
Phase: Phase 1 Status: Completed
Date: 2022-01-31
Effect of Allopurinol on Left Ventricular Function in Children With Dilated Cardiomyopathy
CTID: NCT05193838
Phase: Status: Unknown status
Date: 2022-01-18
XILO-FIST, the Effect of Allopurinol on the Brain Heart and Blood Pressure After Stroke
CTID: NCT02122718
Phase: Phase 4 Status: Completed
Date: 2021-11-12
Chemotherapy and Azidothymidine, With or Without Radiotherapy, for High Grade Lymphoma in AIDS-Risk Group Members
CTID: NCT00000703
Phase: N/A Status: Completed
Date: 2021-11-03
A Phase III Randomized Trial of Low-Dose Versus Standard-Dose mBACOD Chemotherapy With rGM-CSF for Treatment of AIDS-Associated Non-Hodgkin's Lymphoma
CTID: NCT00000658
Phase: Phase 3 Status: Completed
Date: 2021-11-03
Crossover Clinical Trial, Randomized, Double Blind, Placebo Controlled Trial
CTID: NCT04983160
Phase: Phase 2 Status: Completed
Date: 2021-07-30
Uric Acid Reduction as a Novel Treatment for Pediatric Chronic Kidney Disease
CTID: NCT03865407
Phase: Phase 2 Status: Terminated
Date: 2021-05-27
Busulfan and Cyclophosphamide Followed By ALLO BMT
CTID: NCT01685411
Phase: N/A Status: Terminated
Date: 2021-04-13
Lowering Uric Acid in Live Kidney Donors
CTID: NCT03353298
Phase: Phase 2 Status: Completed
Date: 2021-02-24
Allopurinol in Patients With Refractory Angina to Improve Ischemic Symptoms
CTID: NCT04368819
Phase: Phase 2/Phase 3 Status: Unknown status
Date: 2021-02-24
Center of Research Translation (CORT) Project 2
CTID: NCT02038179
Phase: Phase 2/Phase 3 Status: Completed
Date: 2021-01-11
A Multicenter Clinical Trial of Allopurinol to Prevent Kidney Function Loss in Type 1 Diabetes
CTID: NCT02017171
Phase: Phase 3 Status: Completed
Date: 2020-12-04
The Effect of Local Antioxidant Therapy on Racial Differences in Vasoconstriction
CTID: NCT03680404
Phase: Phase 1 Status: Completed
Date: 2020-11-05
Study of Cardiovascular Disease and Obstructive Sleep Apnea
CTID: NCT01637623
Phase: Phase 2 Status: Completed
Date: 2020-08-14
The Effects of BCRP Q141K on Allopurinol Pharmacokinetics and Dynamics
CTID: NCT02956278
Phase: Phase 4 Status: Completed
Date: 2020-06-23
Rasburicase in Patients at High Risk for Tumor Lysis Syndrome (TLS) During Cycle-2
CTID: NCT01200485
Phase: Phase 2 Status: Completed
Date: 2020-01-31
Nonmyeloablative Stem Cell Transplantation for Chronic Lymphocytic Leukemia (CLL)
CTID: NCT00899431
Phase: Phase 2 Status: Terminated
Date: 2020-01-27
Xanthine Oxidase Inhibition in Renal Transplant Recipients
CTID: NCT01332799
Phase: Phase 4 Status: Terminated
Date: 2019-10-15
Allopurinol Maintenance Study for Bipolar Disorder
CTID: NCT00732251
Phase: Phase 4 Status: Terminated
Date: 2019-10-02
Phase II Dose Finding Study of RDEA3170 Versus Placebo in Japanese Patients With Gout or Asymptomatic Hyperuricemia
CTID: NCT02078219
Phase: Phase 2 Status: Completed
Date: 2019-09-24
Does Allopurinol Reduce Thickening of the Left Ventricle of the Heart in Patient With Treated Hypertension?
CTID: NCT02237339
Phase: Phase 4 Status: Completed
Date: 2019-09-17
Zurig (Febuxostat) 40mg Efficacy and Safety Trial
CTID: NCT02600780
Phase: Phase 4 Status: Completed
Date: 2019-08-01
Genetics of Hyperuricemia Therapy in Hmong
CTID: NCT02371421
Phase: Status: Completed
Date: 2019-07-15
A Study to Assess the Safety and Pharmacokinetics of Verinurad and Allopurinol in Asian and Chinese Subjects
CTID: NCT03836599
Phase: Phase 1 Status: Completed
Date:
A Phase 2b, Multicentre, Randomised, Double-blind, Placebo-controlled Study of Verinurad and Allopurinol in Patients with Chronic Kidney Disease and Hyperuricaemia
CTID: null
Phase: Phase 2 Status: Ongoing, Completed
Date: 2019-06-28
Evaluation of allopurinol treatment for autistic disorders and epilepsy in adenylosuccinate lyase deficiency (ADSL)
CTID: null
Phase: Phase 2 Status: Completed
Date: 2018-08-30
The Role of Uric Acid Metabolism in Pathogenesis of Anaphylaxis: the Effect of Allopurinol on Experimentally-induced Allergic Reaction to Peanut in Peanut Allergic Adults- a randomised, double-blind placebo-controlled, cross-over, single centre study.
CTID: null
Phase: Phase 2 Status: GB - no longer in EU/EEA
Date: 2018-03-09
Optimizing 6-mercaptopurine therapy in pediatric acute lymphoblastic leukemia by using allopurinol. Clinical study in children 1-19 years on maintenance therapy for acute lymphoblastic leukemia.
CTID: null
Phase: Phase 2 Status: Completed
Date: 2016-12-09
Low-dose azathioprine and allopurinol versus azathioprine monotherapy for patients with ulcerative colitis: protocol for an investigator initiated, open, multicentre, parallel arm, randomised controlled trial
CTID: null
Phase: Phase 3 Status: Completed
Date: 2016-08-25
JAB02 Repurposing allopurinol as a novel anti-inflammatory treatment for persistent allergic asthma.
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2016-08-17
Thiopurines with Low Dose Allopurinol: a prospective one way cross-over study
CTID: null
Phase: Phase 4 Status: Completed
Date: 2016-07-08
The Effect of Intensive Urate Lowering Therapy (ULT) with Febuxostat in Comparison with Allopurinol on Cardiovascular Risk in Patients with Gout Using Surrogate Markers: a Randomized, Controlled Trial (Acronym: the FORWARD Trial)
CTID: null
Phase: Phase 4 Status: Completed
Date: 2015-09-11
Does allopurinol reduce right ventricular mass in lung disease associated pulmonary hypertension?
CTID: null
Phase: Phase 4 Status: Completed
Date: 2015-01-21
Xanthine oxidase inhibition for improvement of long-term outcomes following ischaemic stroke and transient ischaemic attack (XILO-FIST).
CTID: null
Phase: Phase 3 Status: GB - no longer in EU/EEA
Date: 2015-01-02
A Prospective Study to Evaluate the Effect of Allopurinol on Muscle Energetics in Older People with Impaired Physical Function.
CTID: null
Phase: Phase 4 Status: Completed
Date: 2014-12-12
Does Allopurinol regress Left Ventricular Hypertrophy in Patients with Treated Essential Hypertension?
CTID: null
Phase: Phase 4 Status: Completed
Date: 2014-07-22
A randomised, doublet blinded, placebo controlled cross-over study of Allopurinols effect to prevent loss of kidney function in type 1 diabetes
CTID: null
Phase: Phase 3 Status: Completed
Date: 2014-07-02
Study of allopurinol to prevent GFR loss in type 1 diabetes
CTID: null
Phase: Phase 3 Status: Completed
Date: 2014-03-07
Crossover clinical trial, randomized, double blind, placebo controlled trial. Modulation of cellular mediators and repair endothelial damage in patients with chronic renal disease through inhibition of xanthine oxidase.
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2014-01-31
The effect of sodium nitrite infusion on renal variables, brachial and central blood pressure during enzyme inhibition by allopurinol, enalapril or acetazolamid in healthy subjects. A randomized, double-blinded, placebo controlled, cross-over study
CTID: null
Phase: Phase 4 Status: Completed
Date: 2014-01-03
Allopurinol and cardiovascular outcomes in patients with ischaemic heart disease (ALL-HEART)
CTID: null
Phase: Phase 4 Status: GB - no longer in EU/EEA
Date: 2013-10-01
Does ALlopurinol regress lefT ventricular hypertrophy in End stage REnal Disease: The ALTERED study
CTID: null
Phase: Phase 4 Status: Completed
Date: 2013-08-06
Effect of low-dose Azathioprine and Allopurinol compared to Azathioprine on clinical outcomes in Inflammatory Bowel Disease
CTID: null
Phase: Phase 3 Status: Completed
Date: 2013-05-31
A multicentre, randomised, double-blind, parallel group study on the therapeutic efficacy and safety of Febuxostat (taken once daily) and the therapeutic efficacy and safety of Allopurinol on serum urate concentration in subjects suffering from hyperuricemia and gout.
CTID: null
Phase: Phase 4 Status: Completed
Date: 2013-01-28
Febuxostat for Tumor Lysis Syndrome Prevention in Hematologic Malignancies: a Randomized, Double Blind, Phase III Study versus Allopurinol
CTID: null
Phase: Phase 3 Status: Completed
Date: 2012-09-17
A pilot study of allopurinol to prevent GFR loss in type 1 diabetes
CTID: null
Phase: Phase 3 Status: Prematurely Ended
Date: 2012-09-10
A phase 3, multicenter, randomized, double-blind, placebo-controlled, parallel-group study of the efficacy and safety of lenalidomide (Revlimid®) as maintenance therapy for high-risk patients with chronic lymphocytic leukemia following first-line therapy
CTID: null
Phase: Phase 3 Status: Prematurely Ended, Completed
Date: 2012-05-18
Long-term Allopurinol Safety Study Evaluating Outcomes in Gout Patients (LASSO)
CTID: null
Phase: Phase 4 Status: Completed
Date: 2012-01-18
Allopurinol as a possible new therapy for acute coronary syndromes: The Next Steps
CTID: null
Phase: Phase 4 Status: Completed
Date: 2011-12-23
EFFECT OF THE ADMINISTRATION OF ALLOPURINOL ON THE PREVENTION OF MUSCLE MASS LOSS IN IMMOBILIZED SUBJECTS.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2011-11-22
FAST- Febuxostat versus Allopurinol Streamlined Trial A prospective, randomised, open-label, blinded endpoint (PROBE) clinical trial evaluating long term cardiovascular safety of febuxostat in comparison with allopurinol in patients with chronic symptomatic hyperuricaemia
CTID: null
Phase: Phase 4 Status: Prematurely Ended, Completed
Date: 2011-08-03
Identification of the effect of allopurinol treatment on platelets from diabetic patients with stable coronary ischemic disease and aspirin resistance. XUE Study.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2011-06-08
Implication des radicaux libres produits par la xanthine oxydase dans les altérations mitochondriales du muscle squelettique de patients diabétiques
CTID: null
Phase: Phase 3 Status: Completed
Date: 2011-06-01
Estudio doble ciego de tratatmiento preventivo con alopurinol 3% crema vs placebo de la eritrodisestesia palmoplantar secundaria al tratamiento con capecitabina
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2011-02-28
A Phase II Placebo Controlled, Multicenter Study to Investigate the Safety and Efficacy of ATH008 cream in Patients with Palmar-Plantar Erythrodysesthesia Syndrome (PPES) secondary to capecitabine therapy.
CTID: null
Phase: Phase 2 Status: Ongoing, Completed
Date: 2011-02-03
Allopurinol as a possible oxygen sparing agent during exercise in peripheral arterial disease
CTID: null
Phase: Phase 4 Status: Completed
Date: 2010-09-08
Randomized, Double-Blind, Multicenter, Placebo-Controlled, Combination Study to Evaluate the Safety, Efficacy and Potential Pharmacokinetic Interaction of RDEA594 and Allopurinol in Gout Patients with an Inadequate Hypouricemic Response with Standard Doses of Allopurinol
CTID: null
Phase: Phase 2 Status: Completed
Date: 2009-12-09
Effect of xantina oxidasi inhibition on left ventricular function and on energetic stores of the haert in patients affected by chronic cardiac insufficiency.
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2009-09-30
Does antenatal allopurinol during asphyxia reduce post-hypoxic-ischemic reperfusion damage in the newborn?
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2009-07-21
Do Xanthine Oxidase Inhibitors Regress Left ventricular Hypertrophy in Diabetes?
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2009-07-01
Do xanthine oxidase inhibitors reduce left ventricular hypertrophy and endothelial dysfunction in normotensive patients with chronic stable angina?
CTID: null
Phase: Phase 4 Status: Completed
Date: 2009-06-09
The effect of allopurinol on carotid ultrasound intima-media thickness and markers of endothelial function in patients with recent stroke - a pilot study.
CTID: null
Phase: Phase 4 Status: Completed
Date: 2009-03-13
Evaluación de la utilidad del alopurinol en la prevención de la morbimortalidad cardiovascular de los pacientes con enfermedad renal crónica (ERC) e hiperuricemia
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2009-02-04
Does allopurinol (a xanthine oxidase inhibitor) have clinically useful anti-ischaemic effects in angina pectoris? - A randomised, double-blind, placebo-controlled trial
CTID: null
Phase: Phase 4 Status: Completed
Date: 2008-12-29
Do Xanthine Oxidase Inhibitors reduce both Left Ventricular Hypertrophy and Vascular Dysfunction in Cardiovascular patients with Renal Dysfunction?
CTID: null
Phase: Phase 4 Status: Completed
Date: 2008-01-02
The APEX Trial: Effects of Allopurinol on Coronary and Peripheral Endothelial Function in Patients with Cardiac Syndrome X
CTID: null
Phase: Phase 4 Status: Completed
Date: 2007-11-29
A PHASE 1/2, MULTI-CENTER,OPEN LABEL STUDY OF THE SAFETY AND EFFICACY OF A STEPWISE DOSE-ESCALATION SCHEDULE OF LENALIDOMIDE MONOTHERAPY IN SUBJECTS WITH RELAPSED OR REFRACTORY B-CELL CHRONIC LYMPHOCYTIC LEUKEMIA
CTID: null
Phase: Phase 1, Phase 2 Status: Completed
Date: 2007-02-23
Phase I/II study of prevention of Colorectal Cancer CRC with Allopurinol in High-Risk Subjects
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2006-06-16
Allopurinol in the prevention of superficial bladder tumour recurrence
CTID: null
Phase: Phase 2 Status: Prematurely Ended
Date: 2006-04-28
New therapeutic targets in stroke prevention: the effect of allopurinol on the cerebral vasculature of patients with subcortical stroke
CTID: null
Phase: Phase 4 Status: Completed
Date: 2005-12-09
Reversibility of cerebrovascular endothelial dysfunction in diabetes: the role of allopurinol.
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2005-11-14
EXPLORING THE THERAPEUTIC POTENTIAL OF XANTHINE OXIDASE INHIBITORS IN CORONARY ARTERY DISEASE
CTID: null
Phase: Phase 4 Status: Completed
Date: 2005-10-06
Effect of folic acid supplementation and allopurinol on endothelial function in patients with rheumatoid arthritis treated with methotrexate
CTID: null
Phase: Phase 4 Status: Completed
Date: 2005-09-05
A multicenter, randomized, comparative trial on the effect of febuxostat in preventing cerebral and cardiorenovascular events in patients with hyperuricemia
CTID: UMIN000012134
PhaseNot applicable Status: Complete: follow-up complete
Date: 2013-10-28
Effects of allopurinol mouthwash and rebamipide gargle for stomatitis occurred during radiation therapy for cancer and cancer chemotherapy
CTID: UMIN000011759
PhaseNot applicable Status: Complete: follow-up complete
Date: 2013-09-13
Prospective, randomized, open-label,clinical trial comparing the effects of febuxostat and allopurinol on blood urate levels, endothelial function and makers for obesity/oxidative stress/chronic kidney diseases
CTID: UMIN000011741
Phase: Status: Complete: follow-up continuing
Date: 2013-09-13
Effects of allopurinol mouthwash and rebamipide gargle for stomatitis occurred during radiation therapy for cancer and cancer chemotherapy
CTID: UMIN000011759
PhaseNot applicable Status: Complete: follow-up complete
Date: 2013-09-13
Prospective, randomized, open-label,clinical trial comparing the effects of febuxostat and allopurinol on blood urate levels, endothelial function and makers for obesity/oxidative stress/chronic kidney diseases
CTID: UMIN000011741
Phase: Status: Complete: follow-up continuing
Date: 2013-09-13
A Study to Observe the Efficacy and Safety of Febuxostat in Subjects with Gout and Hyperuricemia
CTID: jRCT2080222060
Phase: Status:
Date: 2013-04-12
Effects of febuxostat on serum uric acid and arteriosclerosis in allopurinol-treated hyperuricemia patients with chronic kidney disease
CTID: UMIN000008909
PhaseNot applicable Status: Complete: follow-up complete
Date: 2012-10-01
Effect of febuxisostat on serum uric acid levels and endothelial function in patients with hyperuricemia
CTID: UMIN000008753
Phase: Phase IV Status: Complete: follow-up complete
Date: 2012-09-01
Influence of febuxostat and allopurinol on renal function
CTID: UMIN000008168
Phase: Status: Complete: follow-up complete
Date: 2012-06-14
Influence of febuxostat and allopurinol on renal function for hyperuricemia
CTID: UMIN000005964
Phase: Status: Complete: follow-up complete
Date: 2011-07-11
The urate-lowering efficacy and safety of febuxostat in the treatment of the hyperuricemia in chronic kidney disease.
CTID: UMIN000005573
PhaseNot applicable Status: Complete: follow-up complete
Date: 2011-05-16
None
CTID: jRCT2080221074
Phase: Status:
Date: 2010-04-19