ANA 12; ANA-12; N-(2-(((Hexahydro-2-oxo-1H-azepin-3-yl)amino)carbonyl)phenyl)benzo(b)thiophene-2-carboxamide; N-[2-[[(Hexahydro-2-oxo-1H-azepin-3-yl)amino]carbonyl]phenyl]benzo[b]thiophene-2-carboxamide; ANA-12; 219766-25-3; N-(2-((2-oxoazepan-3-yl)carbamoyl)phenyl)benzo[b]thiophene-2-carboxamide; N-[2-[(2-oxoazepan-3-yl)carbamoyl]phenyl]-1-benzothiophene-2-carboxamide; ANA12
| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
TrkB (Kd = 10 nM)
|
|---|---|
| 体外研究 (In Vitro) |
体外活性:ANA-12 直接且选择性地与 TrkB 结合,并抑制 TrkB 下游过程,而不改变 TrkA 和 TrkC 功能。在 nnr5 PC12-TrkB 细胞中,ANA-12 在浓度低至 10 nM 时即可阻止脑源性神经营养因子 (BDNF) 诱导的神经突生长。在 DRG 神经元中,ANA-12 消除了 BDNF 对增加内向电流的影响。激酶测定:Maxisorp ELISA 96 孔板用碳酸盐缓冲液 (pH 9.6) 中的不同浓度的 Trk BECD -Fc、20 mg/ml BSA 或 1 mg/mL IgG-Fc(多克隆抗 TrkB)包被过夜。 4°C。将板用 0.5% BSA 的 PBS 溶液在室温下饱和 2 小时,并在 PBS-Tween 0.05% 中彻底清洗。然后将 Bodipy–ANA-12 在 0.5% PBS-BSA 中室温孵育 1 小时,然后添加 0.5% PBS-BSA 中的 BDNF 再孵育 1 小时。在 PBS-Tween 0.05% 中进行大量洗涤后,通过 520 ± 10 nm 处的荧光对结合的 bodipy-ANA-12 量进行定量。外推分析的可检测范围通过用 bodipy-ANA-12 包被 ELISA 板并在 520 ± 10 nm 处读取荧光来评估。细胞测定:分别添加 BDNF (1 nM)、NGF (2 nM) 和 NT-3 (10 nM) 后,在 nnr5 PC12–TrkB、–TrkA 和 –TrkC 细胞中评估分子对神经突生长的调节。通过显微镜确定每个计数视野中具有直径超过 2 个细胞的神经突的细胞数量(每孔 2 个视野,每个条件 3 个孔)。连续 3 天,每 24 小时进行一次盲计数。
N-T19类似物的筛选揭示了一种有效的TrkB拮抗剂。[1] KIRA-ELISA和神经突生长评估显示,只有N-T19能够维持其在神经元和神经元样系统中的作用。然而,尽管N-T19在抑制TrkB活性方面表现出很高的功效,但其相对较低的效价促使我们寻找能保持N-T19原有的高效但效价更高的类似物。为此,使用Bioinfo-DB数据库进行第二轮计算机筛选,以鉴定共享相同分子支架的N-T19类似物(图4A)。14个新分子被鉴定为接近类似物,并在第一组使用KIRA-ELISA检测的功能筛选中进行了测试。四种化合物的活性最高,但只有1种(ANA-12)在重组细胞中表现出亚微摩尔效力。ANA-12的深入药理表征证实了该分子的高效(完全抑制)和效力(亚微摩尔)(图4B)。与母体化合物NT-19一样,ANA-12在神经元中显示出2位点的作用模式,但令人惊讶的是,它也在重组细胞中发挥作用。两种细胞体系(分别为50 μM和50 nM)在低亲和力位点和高亲和力位点上的电位具有可比性。 值得注意的是,在测试的14个分子中,ANA-12的结构与母体先导化合物的结构最接近,两个分子的区别只是在ANA-12中多了一个苯片段(图4A)。 ANA-12直接选择性结合TrkB。[1] 然后,我们通过将荧光标记的化合物(见方法)与嵌合的TrkBECD-Fc或BSA或IgG-Fc孵育作为非特异性结合的阴性对照,确定ANA-12是否直接与TrkB结合。如图5A所示,ANA-12以剂量依赖的方式特异性结合到TrkB的细胞外结构域,而不与BSA或IgG-Fc结合。与TrkBECD-Fc的饱和结合研究表明,ANA-12与TrkBECD-Fc结合的Kd值为12 μM(图5B),对应于之前在KIRA-ELISA中观察到的功能性低亲和力位点。检测到的高亲和位点位于荧光检测的非线性范围内,因此表现为小的压痕。线性化和外推分析表明,ANA-12也与高亲和力位点结合,Kd约为10 nM(数据未显示)。ANA-12与TrkB结合的2位点拟合模型显示,高亲和力位点占总结合位点的20%(数据未显示),这一数值与KIRA-ELISA检测中观察到的30%相似(图4B)。 为了进一步研究ANA-12的结合特性,我们将BDNF加入到ANA-12/TrkB复合物中(图5B)。这导致与TrkBECD-Fc结合的ANA-12的最大数量减少了60%,而曲线没有向右移动(Kd, 16 μM),表明这是一种非竞争机制。综上所述,这些数据表明高亲和力和低亲和力的结合位点共存于TrkB的细胞外结构域,BDNF和ANA-12并不竞争TrkB上的相同位点。 新化合物与TrkB-d5的计算对接表明,推测的结合模式与N-T19相似(图5C): ANA-12的内酰胺部分与TrkB的His299和His300主链原子相互作用,而化合物的无环酰胺部分与TrkB特异性Gln347和Asp298侧链之间形成氢键。该配体的形状适合TrkB-d5 ADEB β-片,特别是通过7元环与可接近的二硫桥Cys302-Cys347之间的疏水接触,以及通过与一束组氨酸残基(His299, His300, His335)的芳香相互作用。 ANA-12影响与TrkB相关的细胞功能,但不影响TrkA和TrkC。[1] 神经突生长被用来验证ANA-12对细胞过程的影响(图5、D和E)。我们观察到,在表达trkb的细胞中,浓度低至10 nM的ANA-12可以阻止bdnf诱导的神经突生长,证实了KIRA-ELISA检测中观察到的高效效(图5D)。在浓度高达10-100 μM时,ANA-12完全消除了BDNF的作用,因为即使在3天后也没有观察到单个神经突起或分支。为了评估该化合物对TrkB的选择性,我们使用了另外两种表达TrkA或TrkC的nnr5-PC12细胞系,它们的神经突生长分别依赖于NGF和NT-3(图5E)。在这些细胞系中,ANA-12对神经突的生长没有影响。浓度高达100 μM的ANA-12孵育3天后,对NGF-和nt -3依赖性的神经突长度和分支没有影响,证实了其对trkb相关信号的特异性。 |
| 体内研究 (In Vivo) |
在成年 C57BL6/129SveV F1s 小鼠中,ANA-12(0.5 mg/kg,腹腔注射)可降低大脑中的 TrKB 活性,减少焦虑和抑郁相关行为,而不影响神经元存活。在雄性 C57BL/6 小鼠中,ANA-12(0.5 mg/kg,腹腔注射)对脂多糖诱导的抑郁样行为显示出抗抑郁样作用。在雄性 Sprague-Dawley 大鼠中,ANA-12(3 μg/剂)可阻断内侧孤束核 (mNTS) BDNF 减少食物摄入的作用。在雄性野生型小鼠中,ANA-12 逆转乙醇摄入并诱导 D3 受体下调,但在 D3R-/- 小鼠中无效。在雄性 CocSired 大鼠中,ANA-12(0.5 mg/kg,腹腔注射)可逆转可卡因自我给药的减少。
初级传入末端的NMDA受体可以通过增加神经递质释放来促进痛觉过敏。在大鼠和小鼠中,我们发现鞘内NMDA诱导神经激肽1受体(NK1R)内化(测量P物质释放)的能力需要事先注射BDNF。在原发性传入神经中选择性敲除NMDA受体可减少NMDA诱导的NK1R内化,从而证实了这些受体在突触前的位置。BDNF的作用是由原肌球蛋白相关激酶B (trkB)受体介导,而不是由p75神经营养因子受体(p75(NTR))介导,因为它不是由proBDNF产生的,并且被trkB拮抗剂ANA-12抑制,而不被p75(NTR)抑制剂t1 - pep5抑制。这些作用可能是通过trkB受体的截断形式介导的,因为在背根神经节(DRG)神经元中几乎没有全长trkB的表达。Src家族激酶抑制剂阻断BDNF的作用,表明trkB受体通过Src家族激酶磷酸化促进这些NMDA受体的激活。培养DRG神经元的Western blot结果显示,BDNF增加了NMDA受体NR2B亚基的Tyr(1472)磷酸化,已知具有增强作用。膜片钳记录显示,BDNF而非proBDNF增加了培养的DRG神经元的NMDA受体电流。在神经性疼痛模型中,nmda诱导的NK1R内化也可以通过脂多糖激活背角小胶质细胞来实现。这些作用被BDNF清道夫、trkB受体拮抗剂和Src家族激酶抑制剂所减弱,这表明小胶质细胞释放的BDNF在神经性疼痛的初级传入事件中增强了NMDA受体。[1] LPS导致海马CA3和齿状回(DG)和前额叶皮质(PFC)的BDNF减少,而LPS增加伏隔核(NAc)的BDNF。地塞米松抑制实验显示lps处理小鼠下丘脑-垂体-肾上腺轴活动过度。腹腔注射7,8- dhf对lps诱导的抑郁样行为有抗抑郁作用,腹腔注射ANA-12可阻断其抗抑郁作用。令人惊讶的是,单独的ANA-12对lps诱导的抑郁样行为表现出抗抑郁样作用。此外,双侧向NAc输注ANA-12具有抗抑郁作用。此外,LPS导致CA3、DG和PFC的脊柱密度降低,而LPS增加了NAc的脊柱密度。有趣的是,7,8- dhf显著减弱了lps诱导的CA3、DG和PFC中p-TrkB和脊柱密度的降低,而ANA-12显著减弱了lps诱导的NAc中p-TrkB和脊柱密度的增加。 结论:lps诱导的炎症可能通过改变CA3、DG、PFC和NAc的BDNF和脊柱密度导致抑郁样行为,这可能参与了7,8- dhf和ANA-12的抗抑郁作用。[2] 为了定位介导后脑室传递BDNF能量平衡效应的神经元,将脑室阈下剂量直接递送至内侧孤束核(mNTS)。mNTS BDNF可显著减少食物摄入,而这一作用被预先给予高选择性TrkB受体拮抗剂{[n2 -2-2-氧氮平-3-基氨基]羰基苯基苯并(b)噻吩-2-carboxamide (ANA-12)}阻断,表明TrkB受体激活介导了后脑BDNF对食物摄入的影响。由于BDNF和瘦素都与黑素皮质素信号相互作用以减少食物摄入,我们还研究了后脑瘦素的摄入抑制作用是否涉及后脑特异性BDNF/TrkB激活。后脑瘦素递送可显著增加后脑背迷走神经复合体内BDNF蛋白含量。为了评估BDNF/TrkB受体信号是否在瘦素信号的下游作用以控制能量平衡,瘦素和ANA-12被共同给予mNTS。TrkB受体拮抗剂可减弱瘦素的摄入抑制作用,提示mNTS TrkB受体激活有助于调解后脑瘦素的厌食作用。总的来说,这些结果表明,trkb介导的mNTS信号负调控食物摄入,部分地,瘦素给药到NTS的摄入抑制作用。[3] 事实上,TrkB选择性拮抗剂ANA-12阻断BDNF通路逆转了慢性稳定的乙醇摄入,并强烈降低了D3R的纹状体表达。最后,我们对丁螺环酮进行了评估,丁螺环酮是一种被批准用于治疗焦虑症的药物,具有D3R拮抗剂活性(经分子模型分析证实),可有效抑制乙醇摄入。因此,通过D3R的DA信号对于乙醇相关的奖励和消耗是必不可少的,并且可能代表了断奶的新治疗靶点。[4] 给药BDNF受体拮抗剂(TrkB受体拮抗剂ANA-12)逆转了雄性可卡因遗传大鼠的可卡因自我给药减少。此外,在自我服用可卡因的男性精子中,乙酰化组蛋白H3与Bdnf启动子的关联增加。综上所述,这些发现表明,父亲自愿摄入可卡因会导致种系的表观遗传重编程,对雄性后代的mPFC基因表达和对可卡因强化的抵抗力产生深远影响。[5] |
| 酶活实验 |
将不同浓度的 Trk BECD -Fc、20 mg/ml BSA 或 1 mg/mL IgG-Fc(多克隆抗 TrkB)涂在 Maxisorp ELISA 96 孔板上,并在 4° 下放置过夜C,pH 为 9.6 的碳酸盐缓冲液。室温下在 0.5% BSA 的 PBS 溶液中溶解两小时后,将板在 0.05% PBS-Tween 中彻底清洗。在 0.5% PBS-BSA 中室温孵育一小时后,将 BDNF 添加到溶液中并再孵育一小时。使用 Bodipy-ANA-12 重复此过程。在 PBS-Tween 0.05% 中彻底洗涤后,通过 520 ± 10 nm 处的荧光测量结合的 bodipy-ANA-12 的量。为了确定外推分析的检测范围,将 ELISA 板涂有 bodipy-ANA-12,并测量 520 ± 10 nm 处的荧光。
ANA-12 binding assay [1] 在4°C的碳酸盐缓冲液(pH 9.6)中,用不同浓度的TrkBECD-Fc(如图所示)、20 mg/ml BSA或1 mg/ml IgG-Fc(多克隆抗trkb)包被Maxisorp ELISA 96孔板,过夜。在室温下,用0.5%的BSA在PBS中饱和2小时,并用0.05%的PBS- tween广泛洗涤。将Bodipy-ANA-12在0.5% PBS-BSA中室温孵育1小时,再加入BDNF在0.5% PBS-BSA中孵育1小时,如图所示。在0.05% PBS-Tween中广泛洗涤后,在520±10 nm处荧光定量体脂- ana -12结合量。外推分析的检测范围通过在ELISA板上涂覆bodipy-ANA-12并在520±10 nm处读取荧光来评估。 |
| 细胞实验 |
在 nnr5 PC12-TrkB、-TrkA 和 -TrkC 细胞中,分别添加 BDNF (1 nM)、NGF (2 nM) 和 NT-3 (10 nM) 后评估分子对神经突生长的影响。显微镜对每个计数区域(每个条件三个孔,每个孔两个区域)中的细胞数量进行计数,其中神经突的直径超过两个细胞。三天内,每 24 小时在黑暗中进行一次计数。
KIRA-ELISA [1] 如前所述,使用改良版KIRA-ELISA定量TrkB受体自磷酸化。将TetOn-rhTrkB细胞接种于96孔平板(每孔4 × 104个细胞),用1000 ng/ml强力霉素孵育过夜,诱导TrkB受体的表达。将皮质神经元接种于多聚氮薄包被的96孔平底培养板(每孔12 × 104个细胞),在37℃、5% CO2中培养7 - 8天。每次检测前验证TetOn-rhTrkB细胞的荧光水平。细胞用DMEM仔细清洗4次,然后用化合物处理20分钟,用BDNF刺激20分钟(重组细胞,4 nM;神经元,0.4 nM),在含有0.5% BSA和25 mM HEPES(对照培养基)的DMEM中,在37°C, 5% CO2中。在冰上去除培养基停止实验,在室温下,通过添加增溶缓冲液(150 mM NaCl, 50 mM Hepes, 0.5% Triton X-100, 0.01%硫柳汞,2mm原钒酸钠,添加蛋白酶抑制剂混合物)使膜溶解1小时。将裂解液转移到预先包被抗gfp(1:5000)用于rhTrkB或抗TrkB (1 μg/ml)用于神经元TrkB的ELISA微滴板上。生物素化抗磷酸酪氨酸(0.5 μg/ml)和酶标链霉亲和素(1:4000)孵育后发现磷酸化。加入TMB后,用1 N盐酸酸化,在450 nm处读取吸光度。总TrkB也使用KIRA-ELISA定量,使用rhTrkB的单克隆抗gfp(1:3000)或神经元TrkB的单克隆抗TrkB(1:1000)沉淀受体,并使用rhTrkB的多克隆抗gfp(1:5000)或神经元TrkB的多克隆抗TrkB (1 μg/ml)检测受体。在细胞培养中,总TrkB信号在所有处理条件下都没有变化。在脑组织中,总TrkB的数量是可变的,用于对磷酸化TrkB获得的信号进行归一化。 |
| 动物实验 |
在17%二甲基亚砜(DMSO)磷酸盐缓冲液中,于注射当日配制氯胺酮(盐酸氯胺酮,10 mg/kg)、7,8-二羟基黄酮(7,8-DHF;10 mg/kg)和ANA-12,N2-(2-{[(2-氧代氮杂环庚烷-3-基)氨基]羰基}苯基)苯并[b]噻吩-2-甲酰胺(0.5 mg/kg)。所选剂量为氯胺酮(10 mg/kg)、7,8-DHF(10 mg/kg)和ANA-12(0.5 mg/kg)。小鼠接受所有化合物的腹腔注射 (ip)。\n
\n\nANA-12 的给药及体内 KIRA-ELISA 分析 [1] \n根据 Banbury 会议关于小鼠行为学遗传背景的建议,使用 C57BL/6 和 129SveV 小鼠杂交获得的 F1 代杂交小鼠。将 3 月龄的成年 C57BL/6/129SveV F1 小鼠随机分为生理盐水组(1% DMSO 溶于 0.9% NaCl 溶液)和 ANA-12 组(溶于生理盐水)。生理盐水组和 ANA-12 组(0.5 mg/kg 体重)均以 10 μl/g 体重的剂量腹腔注射。2 或 4 小时后,将小鼠断头处死,并在冰上迅速取出脑组织。必要时,随后解剖纹状体、皮层和海马。组织样本迅速用冰冷的PBS缓冲液洗涤,转移至冰冷的KIRA-ELISA溶解缓冲液中,并在4℃下过夜。测定蛋白质浓度,上样等量蛋白质,并按上述方法进行KIRA-ELISA检测。\n \n\nANA-12在小鼠脑组织中的稳定性和生物利用度分析[1] \n小鼠血清中的稳定性分析和小鼠脑组织中的定量分析由TechMedILL实验室(法国斯特拉斯堡高等生物技术学院,伊尔基尔希)完成。稳定性评估方法为:将ANA-12在小鼠血清中于37℃孵育15、30、45和60分钟。将混合物均质化,并用乙腈沉淀蛋白质,然后通过液相色谱-质谱联用仪(Agilent LC-MS ESI qTOF,连接 C18-1 × 10 × 1.9 色谱柱)进行分析。如上所述,通过腹腔注射小鼠 ANA-12 (0.5 mg/kg) 来评估其在小鼠脑内的生物利用度。分别在注射后 30 分钟、1 小时、2 小时、4 小时和 6 小时(每个时间点 3 只小鼠)取出脑组织,并在 0.9% NaCl 溶液中研磨,然后用乙腈处理。混合物经超速离心澄清后,使用一系列脱水和乙腈/水 (1/1 v/v) 重悬步骤提取化合物,最后进行 LC-MS 检测和分析。通过向空白混合物(生理盐水处理小鼠的脑组织)中添加已知量的化合物来制备参考样品。 ANA-12 的浓度(ng/g 脑组织)是通过计算检测峰下的面积从参考样品中得出的。\n \n\n体内细胞死亡分析[1] \n使用 TUNEL 检测法评估亚慢性 ANA-12 处理对小鼠脑组织细胞死亡的影响。将小鼠随机分为 4 组,每天腹腔注射一次生理盐水(含 1% DMSO)或 0.5、1.0 或 2.0 mg/kg 的 ANA-12,持续 1 周。末次注射后 24 小时处死小鼠,经心脏灌注 PBS,随后灌注 4% 多聚甲醛。将脑组织在 4% 多聚甲醛中固定过夜,并使用振动切片机获得 50 μm 厚的冠状切片。将游离切片用PBS充分洗涤,并在室温下用含0.5% Triton X-100的PBS进行90分钟的透化处理。之后,将切片冲洗并贴于载玻片上。根据制造商的说明,使用DeadEnd荧光TUNEL系统对3′ OH-DNA链断裂进行标记。反应通过用2×SSC溶液冲洗载玻片并用PBS充分洗涤来终止。切片用VECTASHIELD plus DAPI封片剂封片,并在520 nm波长下使用荧光显微镜观察荧光素染色。阳性对照的制备方法为:将生理盐水处理动物的切片在含0.5% Triton X-100的PBS中,于37℃下用DNA酶(10 U/ml)预处理90分钟。阴性对照(无荧光斑点)的制备方法与上述相同,只是不使用末端转移酶处理切片。\n \n\nANA-12 的行为学测试 [1] \n所有行为学测试均在同一时间(下午 1:00)于安静且独立的房间内进行,环境光照条件明亮(800–900 lux,旷场实验除外,300–400 lux)。注射 0.5 mg/kg ANA-12 4 小时后,采用与上述相同的程序进行行为学测试。为了消除气味线索,每次动物实验后,所有测试设备均使用消毒剂 Roccal 进行彻底清洁。\n \n\nANA-12 对焦虑相关行为的影响 [1] \n使用旷场实验、高架十字迷宫和新奇抑制进食范式测试焦虑相关行为。\n \n\n给药 [2] \nANA-12,N2-(2-{[(2-氧代氮杂环庚烷-3-基)氨基]羰基}苯基)苯并[b]噻吩-2-甲酰胺(0.5mg/kg,腹腔注射),溶于 1% 二甲基亚砜生理盐水中。 7,8-DHF 和 ANA-12 的剂量也按照先前报道的方法选择(Ren 等,2013、2014;Cazorla 等,2011)。\n \n\n手术及双侧 ANA-12 注射入伏隔核 [2] \n小鼠用戊巴比妥钠(5mg/kg)麻醉,并固定于立体定位仪上。将微量注射针双侧插入伏隔核壳部(+1.7 AP,±0.75 ML,-3.6 DV)(Paxinos 和 Watson,1998)。术后24小时,腹腔注射LPS(0.5mg/kg)或生理盐水(10ml/kg)。注射LPS(或生理盐水)23小时后,双侧注射ANA-12(0.1 nmol/L,0.1 μL/min,持续5分钟)或载体。在最后一次注射后4小时和6小时进行行为学评估(图3B)。实验4:mNTS内的BDNF/TrkB受体信号传导。[3]所有大鼠(n = 9)均接受两次单侧NTS实质内注射,两次注射间隔约20分钟,并在第二次注射后1、3、6和24小时测定食物摄入量。实验条件采用平衡设计,具体如下:对照组(先注射100 nl DMSO,再注射100 nl aCSF)、ANA-12组(先注射1或3 μg ANA-12,再注射aCSF)、BDNF组(先注射DMSO,再注射0.2 μg BDNF)以及联合组(先注射1或3 μg ANA-12,再注射0.2 μg BDNF)。分别在注射药物前和注射后24小时测量体重。\n \n\n实验6:后脑TrkB受体信号传导和瘦素。[3] \n所有大鼠(n = 9)均接受两次单侧孤束核(NTS)实质注射,两次注射间隔约20分钟,并在第二次注射后1、3、6和24小时测量食物摄入量。四个平衡条件如下:对照条件(100 nl DMSO,随后 100 nl 碳酸氢钠)、ANA-12条件(3 μg ANA-12,随后碳酸氢钠)、瘦素条件(DMSO,随后 0.2 μg 瘦素)和组合条件(3 μg ANA-12,随后 0.2 μg 瘦素)。在注射药物之前和注射后 24 小时立即测量体重。\n \n\n\n药物和治疗[4] \n乙醇、马来酸 U99194A、盐酸 SB277011A、盐酸丁螺环酮、8-OH-DPAT 和ANA-12溶于生理盐水中,并进行腹腔注射(ip)(剂量为 10 ml/kg),但 ANA-12 溶于 10% 二甲基亚砜中。 U99194A 的使用剂量为 10 mg/kg(Harrison 和 Nobrega,2009),SB277011A 的使用剂量为 10 mg/kg(Song 等,2012),丁螺环酮的使用剂量范围为 0.1–10 mg/kg(Martin 等,1992),8-OH-DPAT 的使用剂量为 1 mg/kg(Martin 等,1992),ANA-12 的使用剂量为 0.5 mg/kg(Cazorla 等,2011)。\n \n在双瓶选择范式中,经过 30 天的自愿饮酒程序后,D3R−/− 和 WT 小鼠被随机分配到八个实验组(每组 n=6/10):WT/载体组、WT/U99194A 组、WT/SB277011A 组、 WT/buspirone、D3R−/−/vehicle、D3R−/−/U99194A、D3R−/−/SB277011A 和 D3R−/−/buspirone。动物每天腹腔注射一次,连续14天。第14天,在最后一次给药后1小时处死动物并取脑组织。在另一组实验中,小鼠经过30天的自愿饮酒程序后,随机分为五组(每组n=5/7):WT naïve、WT/vehicle、WT/ANA-12、D3R−/−/vehicle 和 D3R−/−/ANA-12。动物连续4天每天腹腔注射一次选择性Trkb拮抗剂ANA-12,剂量为0.5 mg/kg(Cazorla等人,2011;Vassoler等人,2013)。第4天,在最后一次给药后1小时处死动物并取脑组织。 |
| 药代性质 (ADME/PK) |
全身给药ANA-12可抑制脑内TrkB活性。[1]
本研究旨在开发一种全身给药后可抑制成年哺乳动物脑内TrkB活性的小分子。我们首先检测了ANA-12在到达脑组织前是否在小鼠血清中稳定且未降解为代谢产物。为此,我们将ANA-12在37℃下于小鼠血清中孵育15、30、45和60分钟。液相色谱-质谱联用(LC-MS)分析结果表明,混合物中未检测到代谢产物,且未观察到ANA-12随时间的降解(图6A)。 随后,我们检测了全身给药后ANA-12是否能穿过血脑屏障并到达脑组织。为此,我们向成年小鼠腹腔注射了ANA-12(0.5 mg/kg)。动物分别于注射后0.5、1、2、4或6小时处死,并提取脑组织,采用液相色谱-质谱联用(LC-MS)法定量分析ANA-12。图6B显示,腹腔注射ANA-12后,最早可在30分钟(~400 nM)至6小时内(~10 nM)在脑组织中检测到活性浓度的ANA-12。 随后,我们检测了ANA-12是否抑制成年动物脑组织中的TrkB活性,并在注射0.5 mg/kg ANA-12后2小时和4小时测定了脑组织中TrkB的抑制程度。选择此时间点是基于我们之前对环磷酰胺B(cyclotraxin-B)的观察结果,环磷酰胺B至少需要3小时才能使脑组织中的TrkB失活。 KIRA-ELISA定量分析磷酸化TrkB的结果显示,0.5 mg/kg的ANA-12可部分抑制全脑内源性TrkB的总活性(2小时时抑制8%,4小时时抑制25%;图6C)。 尽管TrkB广泛分布于脑内,但ANA-12对不同脑区受体的抑制作用可能并不均匀。这可能导致全脑TrkB活性呈现表观上的部分抑制,某些脑区TrkB活性被完全抑制,而其他脑区TrkB活性则未受影响或仅受影响很小。因此,我们测定了不同脑区之间的抑制幅度。我们再次向成年小鼠注射0.5 mg/kg的ANA-12,并在2小时或4小时后收集不同脑区(纹状体、皮层和海马)的样本(图6D)。KIRA-ELISA分析表明,注射2小时后,纹状体中TrkB的抑制效果优于海马和皮层。 4小时后,所有分析结构的抑制程度相当(25%–30%),但纹状体中的TrkB抑制程度似乎略高于海马和皮层。综上所述,这些观察结果表明,极低剂量的ANA-12足以在4小时后均匀地部分抑制整个大脑中的TrkB活性。 |
| 毒性/毒理 (Toxicokinetics/TK) |
ANA-12不影响神经元存活。[1]
由于抑制BDNF/TrkB信号通路可诱导中枢神经系统神经元死亡,我们验证了ANA-12能否长期给药而不对大脑产生毒性作用。为此,我们给成年小鼠每日注射不同剂量的ANA-12(0.5、1.0和2.0 mg/kg)或生理盐水,持续一周。末次注射后,处死小鼠,取脑组织进行TUNEL荧光染色检测细胞凋亡(图9)。在注射生理盐水或0.5或1.0 mg/kg ANA-12的小鼠脑组织中,几乎检测不到TUNEL阳性细胞。值得注意的是,全脑检查显示,凋亡细胞仅存在于海马齿状回。然而,在接受 2.0 mg/kg ANA-12 的小鼠的齿状回中观察到 TUNEL 阳性细胞数量略有增加。在其他任何研究区域(皮质、纹状体、海马 CA1-3 区、下丘脑、丘脑、黑质、腹侧被盖区、苍白球和中缝核)均未检测到染色。 |
| 参考文献 | |
| 其他信息 |
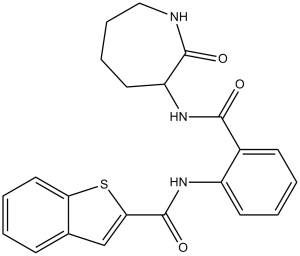
ANA-12 是一种仲酰胺,其结构为邻氨基苯甲酸,其中羧基与α-氨基-ε-己内酰胺的伯氨基发生缩合反应,而芳基氨基与1-苯并噻吩-2-羧酸的羧基发生缩合反应。它是原肌球蛋白受体激酶B(TrkB,又称酪氨酸受体激酶B)的选择性非竞争性拮抗剂。它可作为原肌球蛋白相关激酶B受体拮抗剂、抗抑郁药和抗焦虑药发挥作用。它是一种仲酰胺,属于己内酰胺类化合物和1-苯并噻吩类化合物。它在功能上与 2-氨基己基-6-内酰胺和邻氨基苯甲酸相关。
最后,其他与 N-T19 和 ANA-12 具有相同 3-(酰氨基)-ε-己内酰胺骨架的化合物已被描述为小鼠的促认知剂,尽管其确切的作用机制尚未阐明。鉴于 BDNF/TrkB 偶联在认知和记忆中的核心作用,评估这些化合物对 TrkB 受体的影响并将其与 ANA-12 和 N-T19 进行比较将很有意义。据我们所知,ANA-12 是第一个在体内产生强效且特异性作用的非肽类 TrkB 受体拮抗剂。这种蛋白水解稳定的小分子是一种有价值的药理学工具,能够帮助我们更好地研究BDNF/TrkB信号通路在病理生理条件下的作用,并将作为设计高效口服生物利用度高的TrkB调节剂的先导化合物。[1] 总之,我们的研究表明,LPS诱导的炎症会导致抑郁样行为,并引起海马、前额叶皮层(PFC)和伏隔核(NAc)内BDNF蛋白和树突棘密度的改变。此外,TrkB激动剂7,8-DHF和拮抗剂ANA-12分别使海马和PFC中异常的树突棘以及NAc中异常的树突棘恢复正常,从而对LPS诱导的抑郁行为表现出抗抑郁作用。因此,海马、PFC和NAc中异常的BDNF-TrkB信号通路可能在炎症诱导的抑郁症中发挥作用。最后,在重度抑郁症(MDD)中,TrkB激动剂和TrkB拮抗剂可能作为潜在的治疗药物,用于治疗海马和前额叶皮层(PFC)中脑源性神经营养因子(BDNF)水平较低,以及伏隔核(NAc)中BDNF水平较高的患者。[2] 中脑边缘多巴胺(DA)控制药物和酒精渴求行为,但特定DA受体亚型的作用尚不清楚。我们检验了D3R基因缺失或D3R药理学阻断抑制小鼠乙醇偏好的假设。我们用D3R拮抗剂SB277011A和U99194A处理或不处理D3R缺陷小鼠(D3R-/-)及其野生型(WT)同窝小鼠,并在长期自由选择乙醇饮用实验(双瓶选择)和类似暴饮的乙醇饮用范式(黑暗饮酒,DID)中进行了测试。通过分子建模进一步评估了D3R拮抗剂的选择性。在双瓶选择和DID范式中,D3R(-/-)小鼠的乙醇摄入量可忽略不计,而野生型(WT)小鼠的乙醇摄入量则很高。D3R拮抗剂治疗可抑制WT小鼠的乙醇摄入,但对D3R(-/-)小鼠无效。乙醇摄入增加了WT和D3R(-/-)小鼠中RACK1和脑源性神经营养因子(BDNF)的表达;WT小鼠中D3R也显著过表达。因此,与RACK1/BDNF激活相关的D3R表达增加似乎在自愿摄入乙醇的过程中发挥强化机制的作用。事实上,TrkB选择性拮抗剂ANA-12阻断BDNF通路可逆转慢性稳定乙醇摄入,并显著降低纹状体中D3R的表达。最后,我们评估了丁螺环酮,这是一种已获批准用于治疗焦虑症的药物,具有D3受体拮抗活性(经分子建模分析证实),结果显示其能有效抑制乙醇摄入。因此,通过D3受体的多巴胺信号传导对于乙醇相关的奖赏和摄入至关重要,并可能代表一种新的戒断治疗靶点。[4] 我们描述了一种由大鼠可卡因自我给药引起的可遗传表型。我们观察到,在雄性后代中,可卡因自我给药行为的习得延迟且维持能力降低,而雌性后代则未观察到此现象。脑源性神经营养因子(BDNF)mRNA和BDNF蛋白在内侧前额叶皮层(mPFC)中增加,并且仅在有可卡因经验的雄性后代中,乙酰化组蛋白H3与BDNF启动子的结合增加。给予 BDNF 受体拮抗剂(TrkB 受体拮抗剂 ANA-12)可逆转雄性可卡因后代大鼠可卡因自我给药行为的减少。此外,在自我给药可卡因的雄性大鼠的精子中,乙酰化组蛋白 H3 与 Bdnf 启动子的结合增加。综上所述,这些发现表明,父亲自愿摄入可卡因会导致生殖细胞的表观遗传重编程,从而对雄性后代的 mPFC 基因表达和对可卡因强化的抵抗力产生深远影响。[5] |
| 分子式 |
C22H21N3O3S
|
|
|---|---|---|
| 分子量 |
407.49
|
|
| 精确质量 |
407.13
|
|
| 元素分析 |
C, 64.85; H, 5.19; N, 10.31; O, 11.78; S, 7.87
|
|
| CAS号 |
219766-25-3
|
|
| 相关CAS号 |
|
|
| PubChem CID |
2799722
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| LogP |
4.786
|
|
| tPSA |
126.01
|
|
| 氢键供体(HBD)数目 |
3
|
|
| 氢键受体(HBA)数目 |
4
|
|
| 可旋转键数目(RBC) |
4
|
|
| 重原子数目 |
29
|
|
| 分子复杂度/Complexity |
628
|
|
| 定义原子立体中心数目 |
0
|
|
| SMILES |
S1C2=C([H])C([H])=C([H])C([H])=C2C([H])=C1C(N([H])C1=C([H])C([H])=C([H])C([H])=C1C(N([H])C1([H])C(N([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H])=O)=O)=O
|
|
| InChi Key |
TUSCYCAIGRVBMD-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C22H21N3O3S/c26-20(25-17-10-5-6-12-23-21(17)27)15-8-2-3-9-16(15)24-22(28)19-13-14-7-1-4-11-18(14)29-19/h1-4,7-9,11,13,17H,5-6,10,12H2,(H,23,27)(H,24,28)(H,25,26)
|
|
| 化学名 |
N-[2-[(2-oxoazepan-3-yl)carbamoyl]phenyl]-1-benzothiophene-2-carboxamide
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 1.43 mg/mL (3.51 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 悬浮液;超声助溶。
例如,若需制备1 mL的工作液,可将100 μL 14.3 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 1.43 mg/mL (3.51 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 14.3 mg/mL 澄清 DMSO 储备液加入到 900 μL 玉米油中并混合均匀。 View More
配方 3 中的溶解度: 1 mg/mL (2.45 mM) in 5% DMSO + 40% PEG300 + 5% Tween80 + 50% Saline (这些助溶剂从左到右依次添加,逐一添加), 悬浊液; 超声助溶。 配方 4 中的溶解度: ≥ 0.45 mg/mL (1.10 mM) (饱和度未知) in 5% DMSO + 95% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 配方 5 中的溶解度: ≥ 0.45 mg/mL (1.10 mM) (饱和度未知) in 5% DMSO + 95% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 6 中的溶解度: 2% DMSO+30% PEG 300+2% Tween 80+ddH2O: 2mg/mL . 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4540 mL | 12.2702 mL | 24.5405 mL | |
| 5 mM | 0.4908 mL | 2.4540 mL | 4.9081 mL | |
| 10 mM | 0.2454 mL | 1.2270 mL | 2.4540 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
Effects of 7,8-DHF and ANA-12 on LPS-induced changes in phosphorylation of TrkB in the mouse brain. Int J Neuropsychopharmacol. 2015 Feb; 18(4): pyu077. |
Role of TrkB and mTORC1 in the antidepressant action of 7,8-DHF and ANA-12 on LPS-induced depression-like behavior. Int J Neuropsychopharmacol. 2014 Oct 31;18(4). |
Role of TrkB and mTORC1 in the antidepressant action of 7,8-DHF and ANA-12 on LPS-induced depression-like behavior. Int J Neuropsychopharmacol. 2014 Oct 31;18(4). |