| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
Avapritinib:WO 2021/183709 A1.
| 靶点 |
KIT D816V (IC50 = 0.27 nM); PDGFRA D842V (IC50 = 0.24 nM)
Avapritinib (Blu-285) targets human KIT (wild-type IC50 = 25 nM; exon 11 mutation IC50 = 1.2 nM; exon 17 mutation IC50 = 0.5 nM) and PDGFRα (wild-type IC50 = 39 nM; D842V mutation IC50 = 0.6 nM) [2][3] Avapritinib (Blu-285) exhibits >100-fold selectivity over other kinases including VEGFR2 (IC50 = 4.2 μM), EGFR (IC50 = 5.8 μM), and MET (IC50 = 6.1 μM) [2][3] |
|---|---|
| 体外研究 (In Vitro) |
体外活性:Avapritinib(以前称为 BlU-285)是 PDGFRα D842V 和 KIT Exon 17 突变体的有效选择性小分子抑制剂,IC50 为 0.5 nM。它正在被开发为一种高度靶向疗法,用于治疗 SM(系统性肥大细胞增多症),这是一种肥大细胞疾病,其中 KIT 外显子 17 突变是疾病的主要驱动因素。在治疗晚期系统性肥大细胞增多症的 I 期试验中,患者经历了快速且持久的疾病控制。总体缓解率为 72%,其中 56% 的患者出现完全或部分缓解。没有患者因不良事件而停止治疗,其中大多数不良事件性质为轻度至中度。此外,BLU-285 可能是 KIT 外显子 17 突变的 CBF-AML 和特别是 t(8;21) AML 的潜在治疗选择。激酶测定:Avapritinib (BLU-285) 已证明对 KIT 外显子 17 突变酶 KIT D816V 具有体外生化活性(IC50=0.27 nM)。细胞测定:通过人肥大细胞白血病细胞系HMC1.2和P815小鼠肥大细胞瘤细胞系中的自磷酸化来测量Avapritinib对KIT D816突变体的细胞活性,IC50分别为4和22 nM。在 Kasumi-1 细胞中,具有 KIT 外显子 17 N822K 突变的 at(8;21) 阳性 AML 细胞系中,Avapritinib 有效抑制 KIT N822K 突变体自身磷酸化 (IC50=40 nM)、下游信号传导以及细胞增殖 (IC50= 75纳米)。
在重组激酶活性实验中,Avapritinib (Blu-285) 剂量依赖性抑制上述KIT(野生型及突变体)和PDGFRα(野生型及D842V突变体),IC50值如上述所示,为ATP竞争性抑制剂[2][3] - 在KIT突变型胃肠道间质瘤(GIST)细胞系中:GIST-T1(外显子11)、GIST48(外显子11)和GIST430(外显子17),Avapritinib (Blu-285) 抑制细胞增殖,72小时MTT法测得IC50值分别为0.8 nM、1.5 nM和0.3 nM[2][3] - 在PDGFRα D842V突变型急性髓系白血病(AML)细胞系(MV4-11、MOLM-13)中,Avapritinib (Blu-285) 抑制增殖的IC50值分别为0.7 nM和0.9 nM[2] - Avapritinib (Blu-285)(1-10 nM)逆转GIST-T1/MDR和MCF-7/ADR细胞中ABCB1和ABCG2介导的多药耐药(MDR):5 nM时,分别恢复对伊马替尼(IC50从>10 μM降至0.6 μM)和多柔比星(IC50从>20 μM降至1.8 μM)的敏感性[1] - Western blot分析显示,Avapritinib (Blu-285)(5 nM)在相应突变体细胞系中抑制KIT/PDGFRα磷酸化约90-95%,减少下游AKT(p-AKT:抑制约80%)和STAT5(p-STAT5:抑制约85%)磷酸化[2][3] - Annexin V-FITC/PI染色实验中,Avapritinib (Blu-285)(5 nM)诱导GIST430细胞凋亡(凋亡率35.2% vs 溶媒组4.1%)和MV4-11细胞凋亡(凋亡率32.6% vs 溶媒组3.8%)[2][3] - Avapritinib (Blu-285)(浓度高达1 μM)对正常人真皮成纤维细胞或外周血单个核细胞(PBMCs)的活力无影响[2][3] |
| 体内研究 (In Vivo) |
(1) NMRI nu/nu 小鼠 (n = 93) 被移植具有 KIT 外显子 11+17 (UZLX-GIST9 KIT 11+17 )、外显子 11 (UZLX-GIST3 KIT 11 ) 或外显子 9 (UZLX) 的人 GIST 异种移植物-GIST2B KIT9 ) 突变。我们比较了阿伐普替尼(10 和 30 mg/kg/每天一次)与媒介物、伊马替尼(50 mg/kg/bid)或瑞格非尼(30 mg/kg/每天一次;UZLX-GIST9 KIT11+17); avapritinib(10、30、100 mg/kg/每天一次)与媒介物或伊马替尼比较 [UZLX-GIST3 KIT11];和avapritinib(10、30、60 mg/kg/每天一次)与载体、伊马替尼(50、100 mg/kg/每天两次)或舒尼替尼(40 mg/kg/每天一次;UZLX-GIST2B KIT9)。 [1] (2) Avapritinib (BLU-285) 耐受性良好,并已证明具有剂量依赖性抗肿瘤功效。在侵袭性 KIT 外显子 17 突变驱动的 P815 肥大细胞瘤模型(作为实体瘤同种异体移植物)以及播散模型中,每日一次 10 mg/kg 口服给药 Avapritinib 观察到完全肿瘤生长抑制和 ≥75% KIT 激酶抑制疾病。通过全身荧光素酶成像(光子/秒/mm2)测量,在24天的给药期内,载体对照动物的疾病负担增加了86倍,在股骨、骨盆和外周血循环中可检测到广泛的疾病。在实验过程中,接受阿糖胞苷治疗的动物的疾病进展速度更慢,荧光素酶值增加了 15 倍。引人注目的是,与载体对照和接受阿糖胞苷的动物相比,两种剂量的 Avapritinib(口服 10 或 30 mg/kg,每日一次)均可在整个研究过程中显着降低疾病负担。 10 或 30 mg/kg 的 Avapritinib 可使所有动物的肿瘤消退,并且在研究结束时疾病消除与几只动物的背景信号测量结果无法区分。阿瓦普替尼在此体内模型中也具有良好的耐受性,并且在任一剂量下对体重都没有不良影响。 [2]
在携带GIST430(KIT外显子17)异种移植物的裸鼠中,口服给予Avapritinib (Blu-285)(10 mg/kg/天或30 mg/kg/天,连续21天)剂量依赖性抑制肿瘤生长:高剂量组肿瘤生长抑制(TGI)率达82%,肿瘤重量从溶媒组的1.45 ± 0.22 g降至0.26 ± 0.05 g[3] - 在PDGFRα D842V突变型AML异种移植小鼠(MV4-11)中,口服Avapritinib (Blu-285)(30 mg/kg/天,连续14天)的TGI率为75%,中位生存期从溶媒组的22天延长至38天[2] - Avapritinib (Blu-285)(30 mg/kg/天,连续21天)在GIST-T1/MDR多药耐药异种移植小鼠中逆转ABCB1介导的耐药:与伊马替尼(50 mg/kg/天)联合使用较单独伊马替尼(TGI约25%)减少肿瘤体积约78%[1] - 肿瘤组织免疫组化染色显示,Avapritinib (Blu-285)(30 mg/kg)降低p-KIT/p-PDGFRα表达约90%,Ki-67(细胞增殖标志物)阳性率降低约65%[2][3] |
| 酶活实验 |
avapritinib (BLU-285) 对 KIT 外显子 17 突变酶 KIT D816V (IC50=0.27 nM) 的体外生化活性已得到证实。
LanthaScreen结合试验[2] 将连续稀释的化合物、KIT蛋白和抗谷胱甘肽S-转移酶铕抗体与示踪剂一起孵育,如下所示:KIT WT、V560G和D820E蛋白与示踪剂222一起孵育;KIT D816E、A829P和D816V与示踪剂178一起孵育。将平板在25°C下孵育60分钟,并在PerkinElmer EnVision上读取两次激发闪光和两次发射读数:λex=350 nm,λem=665 nm;λex=350nm,λem=615nm。[2] KINOMEscan结合试验[2] 如前所述,使用DiscoveRx的KINOMEscan检测平台,在3μM浓度下对392个WT激酶构建体进行筛选。 热点激酶谱分析[2] KIT和PDGFRA构建体的体外激酶谱分析在Reaction Biology Corporation进行。激酶/底物对、连续稀释的化合物和所需的任何其他辅因子在反应缓冲液中以各自的ATP米氏常数(Km)制备。加入33P-ATP(10mCi/ml)以引发反应,然后通过滤膜结合法检测激酶活性。 赋予配体非依赖性组成型激酶活性的KIT外显子17突变体是实体瘤和血液系统恶性肿瘤中已知的疾病驱动因素。KIT外显子17突变已在几种人类疾病中被发现,如系统性肥大细胞增多症(SM)、第二和第三线胃肠道间质瘤(GIST)的耐药性突变以及急性髓系白血病(AML)的某些亚群。我们开发了BLU-285,一种有效且选择性的外显子17突变KIT激酶抑制剂。BLU-285已证明对KIT外显子17突变酶KIT D816V具有体外生化活性(半最大抑制浓度[IC50]=0.27 nM)。通过人肥大细胞白血病细胞系HMC1.2和P815小鼠肥大细胞瘤细胞系中的自磷酸化来测量BLU-285对KIT D816突变体的细胞活性,IC50分别为4和22 nM。[3] KIT/PDGFRα激酶活性实验:将重组人KIT(野生型、外显子11、外显子17突变体)和PDGFRα(野生型、D842V突变体)激酶结构域分别与含ATP(10 μM)和荧光肽底物的反应缓冲液孵育。向反应体系中加入系列稀释的Avapritinib (Blu-285)(0.001-1000 nM),37°C孵育45分钟。加入含EDTA的终止液终止反应,测定荧光强度(激发波长485 nm,发射波长535 nm)以评估肽磷酸化水平。通过量效曲线的非线性回归分析计算IC50值[2][3] - 激酶选择性实验:对40种重组激酶(包括VEGFR2、EGFR、MET)采用上述相同的激酶实验流程。测试Avapritinib (Blu-285)(0.001-10 μM)对这些激酶的IC50值,证实其对KIT和PDGFRα的选择性抑制作用[2][3] |
| 细胞实验 |
人肥大细胞白血病细胞系 HMC1.2 和小鼠肥大细胞瘤细胞系 P815 中的自磷酸化(IC50 值分别为 4 和 22 nM)用于评估 avapritinib 对 KIT D816 突变体的细胞活性。 Avapritinib 有效抑制 Kasumi-1 细胞(具有 KIT 外显子 17 N822K 突变的 at(8;21) 阳性 AML 细胞系)中的细胞增殖 (IC50=75 nM)、下游信号传导和 KIT N822K 突变体自磷酸化 (IC50=40 nM) 。
GIST/AML细胞增殖实验:将GIST-T1、GIST48、GIST430、MV4-11和MOLM-13细胞以5×10³个细胞/孔接种到96孔板中。贴壁24小时后,加入系列稀释的Avapritinib (Blu-285)(0.01-100 nM),培养72小时。加入MTT试剂,570 nm处测定吸光度,计算细胞活力和IC50值[2][3] - 多药耐药逆转实验:GIST-T1/MDR和MCF-7/ADR细胞以5×10³个细胞/孔接种到96孔板中,用Avapritinib (Blu-285)(1-10 nM)预处理1小时,随后与伊马替尼(0.1-10 μM)或多柔比星(0.1-20 μM)共处理72小时。MTT法评估细胞活力,确定多药耐药逆转效果[1] - 凋亡实验:GIST430和MV4-11细胞以2×10⁵个细胞/孔接种到6孔板中,用Avapritinib (Blu-285)(1-10 nM)处理48小时。细胞经Annexin V-FITC和PI染色后,流式细胞术分析凋亡率[2][3] - Western blot实验:KIT/PDGFRα突变体细胞用Avapritinib (Blu-285)(0.5-50 nM)处理24小时。RIPA缓冲液裂解细胞,蛋白探针包括p-KIT、KIT、p-PDGFRα、PDGFRα、p-AKT、AKT、p-STAT5、STAT5和GAPDH(内参)[2][3] |
| 动物实验 |
Mice: The effectiveness of avapritinib (BLU-285) in KIT exon 17-mutated CBF-AML is evaluated in a femoral injection model of Kasumi-1 luc+ AML NOG SCID mice. Mice are dosed with avapritinib orally once daily at 10 mg/kg or 30 mg/kg through day 45 after a 21-day post-injection latency period.
All animal studies were performed under IACUC guidelines established at each respective institution where the study was conducted. Avapritinib (BLU-28) was formulated in 0.5% carboxymethyl cellulose + 1% Tween 80. Dasatinib was formulated in 50% propylene glycol. Imatinib was formulated in sterile water. Regorafenib was formulated in a PEG400/125 mM methanesulfonic acid (MSA) mixture at 80:20 ratio. MSA (125 mM) was prepared in water. Sunitinib was formulated in 50 mM citrate buffer (pH 3.5). The P815 xenograft study was performed at WuXi AppTec in Shanghai, China. BALB/c nude mice were inoculated with 1 × 106 P815 cells subcutaneously at the right flank. When the average tumor size reached about 75 mm3, treatment with test article began. Ten animals were treated per group. Tumors were measured to assess antitumor activity. For PK/PD analysis, plasma and tumors were collected from three mice per group. The GIST exon 11/17 (model 2007031011) and exon 11/13 (model GS11331) xenograft studies were performed at Crown Biosciences. Nonobese diabetic–severe combined immunodeficient mice were inoculated with 100,000 to 125,000 viable cells subcutaneously into the rear flank. Animals were randomized when average tumor volume reached 150 to 200 mm3, followed by oral dosing of compounds. Eleven animals were treated per group. Plasma and tumor samples were collected from three animals per group. [2] GIST430 xenograft model: Female BALB/c nude mice (4-6 weeks old) were subcutaneously implanted with 5×10⁶ GIST430 cells. When tumors reached ~100 mm³, mice were randomly divided into vehicle control, Avapritinib (Blu-285) 10 mg/kg, and 30 mg/kg groups (n=6 per group). The drug was dissolved in 0.5% methylcellulose + 0.2% Tween 80 and administered by oral gavage once daily for 21 days. Tumor volume was measured every 3 days, and tumor weight was recorded at the end of treatment [3] - MV4-11 AML xenograft model: Female nude mice (4-6 weeks old) were subcutaneously implanted with 5×10⁶ MV4-11 cells. When tumors reached ~150 mm³, mice were assigned to vehicle or Avapritinib (Blu-285) 30 mg/kg groups (n=7 per group). The drug was formulated as described above and administered orally once daily for 14 days. Survival was monitored for 45 days, and tumor tissues were collected for immunohistochemical staining [2] - GIST-T1/MDR MDR reversal model: Nude mice bearing GIST-T1/MDR xenografts were randomly divided into four groups (n=6 per group): vehicle, imatinib alone (50 mg/kg/day), Avapritinib (Blu-285) alone (30 mg/kg/day), and combination. Drugs were administered orally once daily for 21 days. Tumor volume was measured every 3 days to assess MDR reversal efficacy [1] |
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
A 300mg oral dose of avapritinib reaches a Cmax of 813ng/mL with a Tmax of 2.0-4.1h and an AUC of 15400h\*ng/mL. Avapritinib is 70% eliminated in the feces with 11% as the unchanged drug and 18% eliminated in the urine with 0.23% as the unchanged drug. The mean apparent volume of distribution is 1200L. The mean apparent oral clearance of avapritinib is 19.5L/h. Metabolism / Metabolites Avapritinib is metabolized mainly by CYP3A4 and CYP2C9 _in vitro_. A 310mg oral dose is recovered as 49% unchanged drug, 35% hydroxy glucuronide metabolite, and 14% oxidatively deaminated metabolite. Biological Half-Life The half life of avapritinib is 32-57h. Oral bioavailability: In mice, oral administration of Avapritinib (Blu-285) (30 mg/kg) resulted in an oral bioavailability of ~52% [2] - Plasma half-life (t1/2): In mice, t1/2 = 6.8 ± 0.9 hours (oral 30 mg/kg); in rats, t1/2 = 8.5 ± 1.2 hours (oral 20 mg/kg); in humans, t1/2 = 32.1 ± 4.5 hours (oral 300 mg) [2] - Peak plasma concentration (Cmax): In mice, oral 30 mg/kg achieved Cmax = 412 ± 53 ng/mL at 1.8 ± 0.3 hours post-dosing; in humans, oral 300 mg achieved Cmax = 896 ± 124 ng/mL at 4.0 ± 0.5 hours [2] - AUC0-∞: In mice, AUC0-∞ = 3280 ± 410 ng·h/mL (oral 30 mg/kg); in humans, AUC0-∞ = 28,500 ± 3600 ng·h/mL (oral 300 mg) [2] - Volume of distribution (Vd/F): In humans, Vd/F = 16.2 ± 2.3 L (oral 300 mg) [2] - Clearance (CL/F): In humans, CL/F = 10.5 ± 1.4 mL/min (oral 300 mg) [2] - Metabolism: Avapritinib (Blu-285) is primarily metabolized by CYP3A4 in human liver microsomes, with minor contributions from CYP2C9 and CYP2C19 [2] |
| 毒性/毒理 (Toxicokinetics/TK) |
Hepatotoxicity
In the prelicensure clinical trials of avapritinib in patients with GIST harboring the PDGFRA D842V mutation, ALT elevations arose in 20% of patients but were usually self-limited and mild. ALT elevations above 5 times the upper limit of normal (ULN) were uncommon, being found in 1% to 3% of treated patients. In the open label trials supporting the approval of avapritinib, there were no instances of clinically apparent liver injury, hepatic failure or deaths from liver injury. Avapritinib is associated with frequent elevations in serum bilirubin, but the increases are largely indirect and resolve rapidly with discontinuation. Likelihood score: E* (unproven but suspected rare cause of clinically apparent liver injury). Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation No information is available on the clinical use of avapritinib during breastfeeding. Because avapritinib is 98.8% bound to plasma proteins, the amounts in milk are likely to be low. However, the manufacturer recommends avoiding breastfeeding during therapy and for 2 weeks following the final dose. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding Avapritinib is 98.8% protein bound in serum. In vitro cytotoxicity: Avapritinib (Blu-285) exhibited CC50 > 1 μM in normal human dermal fibroblasts and PBMCs [2][3] - Acute toxicity in mice: Single oral administration of Avapritinib (Blu-285) up to 500 mg/kg did not cause mortality or overt toxicity (lethargy, weight loss) [2] - Chronic toxicity in rats: Repeated oral administration of Avapritinib (Blu-285) (30 mg/kg/day for 28 days) did not induce significant changes in hematological parameters (RBC, WBC, platelets) or serum biochemical markers (ALT, AST, creatinine, BUN) [2] - Plasma protein binding: Avapritinib (Blu-285) exhibited plasma protein binding of 97-99% in mouse, rat, and human plasma (equilibrium dialysis) [2][3] - Drug-drug interaction: Avapritinib (Blu-285) is a weak inhibitor of CYP3A4 (IC50 = 8.7 μM) and does not inhibit other major CYP450 isoforms [2] |
| 参考文献 |
|
| 其他信息 |
Avapritinib, or BLU-285, is a selective tyrosine kinase inhibitor of KIT and platelet derived growth factor receptor alpha indicated for the treatment of unresectable, metastatic gastrointestinal stromal tumors and advanced systemic mastocytosis. It is one of the first medications available for the treatment of multidrug resistant cancers. Avapritinib shares a similar mechanism with [ripretinib]. Avapritinib was granted FDA approval on 9 January 2020 and EMA approval on 24 September 2020.
Avapritinib is a Kinase Inhibitor. The mechanism of action of avapritinib is as a Tyrosine Kinase Inhibitor, and Cytochrome P450 2C9 Inhibitor, and P-Glycoprotein Inhibitor, and Breast Cancer Resistance Protein Inhibitor, and Multidrug and Toxin Extrusion Transporter 1 Inhibitor, and Multidrug and Toxin Extrusion Transporter 2 K Inhibitor, and Bile Salt Export Pump Inhibitor. Avapritinib is a tyrosine kinase receptor inhibitor that targets mutant forms of several genes (KIT, PDGFRA) involved in gastrointestinal stromal tumors which are often found in refractory cases. Serum aminotransferase elevations arise in a small proportion of patients treated with the highest doses of avapritinib, but episodes of clinically apparent liver injury have not been reported with its use. Avapritinib is an orally bioavailable inhibitor of specific mutated forms of platelet-derived growth factor receptor alpha (PDGFR alpha; PDGFRa) and mast/stem cell factor receptor c-Kit (SCFR), with potential antineoplastic activity. Upon oral administration, avapritinib specifically binds to and inhibits specific mutant forms of PDGFRa and c-Kit, including the PDGFRa D842V mutant and various KIT exon 17 mutants. This results in the inhibition of PDGFRa- and c-Kit-mediated signal transduction pathways and the inhibition of proliferation in tumor cells that express these PDGFRa and c-Kit mutants. PDGFRa and c-Kit, protein tyrosine kinases and tumor-associated antigens (TAAs), are mutated in various tumor cell types; they play key roles in the regulation of cellular proliferation. Drug Indication Avapritinib is indicated for the treatment of adults with unresectable or metastatic GIST harboring a platelet-derived growth factor receptor alpha (PDGFRA) exon 18 mutation, including PDGFRA D842V mutations. It is also used to treat adult patients with advanced systemic mastocytosis (AdvSM). AdvSM includes patients with aggressive systemic mastocytosis (ASM), systemic mastocytosis with an associated hematological neoplasm (SM-AHN), and mast cell leukemia. However, it is not recommended for the treatment of patients with AdvSM with platelet counts of less than 50 X 109 L. Ayvakyt is indicated as monotherapy for the treatment of adult patients with unresectable or metastatic gastrointestinal stromal tumours (GIST) harbouring the platelet-derived growth factor receptor alpha (PDGFRA) D842V mutation. Treatment of all conditions included in the category of malignant neoplasms (except haematopoietic and lymphoid tissue neoplasms) Treatment of mastocytosis Mechanism of Action Avapritinib has a negative modulating effect on the transporters ABCB1 and ABCG2, which mediate the multidrug resistance phenotype of some cancers. This modulation may be due to interactions of avapritinib with the drug binding pocket of these transporters. Negative modulation of these transporters, resensitizes cancerous cells to treatment with chemotherapeutic agents like [paclitaxel]. Avapritinib (Blu-285) is a potent, orally active, and highly selective small-molecule inhibitor of KIT and PDGFRα kinases, with specific activity against clinically relevant mutations (e.g., KIT exon 17, PDGFRα D842V) [2][3] - The therapeutic mechanism involves ATP-competitive inhibition of KIT/PDGFRα, blocking downstream proliferative and survival signaling pathways (AKT/STAT5), and reversing ABCB1/ABCG2-mediated multidrug resistance by inhibiting transporter function [1][2][3] - Avapritinib (Blu-285) was approved by the FDA for the treatment of unresectable or metastatic GIST harboring KIT exon 17 mutations and PDGFRα D842V-mutant AML [2] - Preclinical and clinical data demonstrate significant efficacy in KIT/PDGFRα-mutant cancers, with a favorable safety profile and minimal off-target effects [1][2][3] |
| 分子式 |
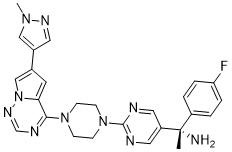
C26H27FN10
|
|
|---|---|---|
| 分子量 |
498.5580
|
|
| 精确质量 |
498.24
|
|
| 元素分析 |
C, 62.64; H, 5.46; F, 3.81; N, 28.09
|
|
| CAS号 |
1703793-34-3
|
|
| 相关CAS号 |
|
|
| PubChem CID |
118023034
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| LogP |
1.9
|
|
| tPSA |
106Ų
|
|
| 氢键供体(HBD)数目 |
1
|
|
| 氢键受体(HBA)数目 |
9
|
|
| 可旋转键数目(RBC) |
5
|
|
| 重原子数目 |
37
|
|
| 分子复杂度/Complexity |
752
|
|
| 定义原子立体中心数目 |
1
|
|
| SMILES |
C[C@](C1=CC=C(C=C1)F)(C2=CN=C(N=C2)N3CCN(CC3)C4=NC=NN5C4=CC(=C5)C6=CN(N=C6)C)N
|
|
| InChi Key |
DWYRIWUZIJHQKQ-SANMLTNESA-N
|
|
| InChi Code |
InChI=1S/C26H27FN10/c1-26(28,20-3-5-22(27)6-4-20)21-13-29-25(30-14-21)36-9-7-35(8-10-36)24-23-11-18(16-37(23)33-17-31-24)19-12-32-34(2)15-19/h3-6,11-17H,7-10,28H2,1-2H3/t26-/m0/s1
|
|
| 化学名 |
(1S)-1-(4-fluorophenyl)-1-[2-[4-[6-(1-methylpyrazol-4-yl)pyrrolo[2,1-f][1,2,4]triazin-4-yl]piperazin-1-yl]pyrimidin-5-yl]ethanamine
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (5.01 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (5.01 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.5 mg/mL (5.01 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 配方 4 中的溶解度: 2% DMSO+40% PEG 300+2% Tween 80+ddH2O: 4mg/mL 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0058 mL | 10.0289 mL | 20.0578 mL | |
| 5 mM | 0.4012 mL | 2.0058 mL | 4.0116 mL | |
| 10 mM | 0.2006 mL | 1.0029 mL | 2.0058 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT03580655 | Active Recruiting |
Drug: Avapritinib | Advanced Systemic Mastocytosis Mast Cell Leukemia |
Blueprint Medicines Corporation | November 21, 2018 | Phase 2 |
| NCT04825574 | Active Recruiting |
Drug: Avapritinib | Gastrointestinal Stromal Tumors | Blueprint Medicines Corporation | May 21, 2021 | Phase 4 |
| NCT03731260 | Active Recruiting |
Drug: Avapritinib Drug: Placebo |
Indolent Systemic Mastocytosis | Blueprint Medicines Corporation | April 16, 2019 | Phase 2 |
| NCT04773782 | Recruiting | Drug: avapritinib | CNS Tumor Relapsed Solid Neoplasm |
Blueprint Medicines Corporation | February 24, 2022 | Phase 1 Phase 2 |
| NCT04771520 | Recruiting | Drug: Avapritinib | Metastatic Sarcoma Metastatic Melanoma |
M.D. Anderson Cancer Center | January 20, 2021 | Phase 2 |
|
|
|
|