| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
BTK (IC50 = 0.5 nM); TEC (IC50 = 10 nM); ITK (IC50 = 15 nM); BLK (IC50 = 23 nM); TXK (IC50 = 28 nM); BMX (IC50 = 32 nM); LCK (IC50 = 71 nM); SRC (IC50 = 1100 nM)
BMS-986142 potently inhibits human recombinant BTK with an IC50 of 0.5 nM in enzymatic assays. BMS-986142 exhibits high selectivity against a panel of 384 kinases; only five other kinases (Tec, ITK, BLK, Txk, and BMX) are inhibited with selectivity for BTK that is less than 100 times. Out of these kinases, only Tec (IC50=10 nM) is inhibited with a selectivity that is less than thirty times when compared to BTK. These four kinases are members of the Tec family of kinases. BMS-986142 does not prevent peripheral blood B cells from expressing CD86 or CD69 when stimulated by CD40L (IC50>10,000 nM for both). With an IC50 of 9 nM, BMS-986142 inhibits BTK-dependent calcium flux in response to anti-IgM treatment of Ramos B cells, activating BCR[2]. |
|---|---|
| 体外研究 (In Vitro) |
BMS-986142 有效抑制人重组 BTK,在酶测定中 IC50 为 0.5 nM。 BMS-986142 对一组 384 种激酶表现出高选择性;只有五种其他激酶(Tec、ITK、BLK、Txk 和 BMX)受到抑制,且对 BTK 的选择性小于 100 倍。在这些激酶中,只有 Tec (IC50=10 nM) 受到抑制,其选择性低于 BTK 的 30 倍。这四种激酶是 Tec 激酶家族的成员。当受到 CD40L 刺激时,BMS-986142 不会阻止外周血 B 细胞表达 CD86 或 CD69(两者的 IC50>10,000 nM)。 BMS-986142 的 IC50 为 9 nM,可响应 Ramos B 细胞的抗 IgM 治疗而抑制 BTK 依赖性钙流,从而激活 BCR[2]。
BMS-986142 在酶学实验中有效抑制人重组BTK,IC50为0.5 nM,并且对包含384种激酶的面板显示出高选择性,主要抑制其他Tec家族激酶。[2] 在原代人B细胞中,通过B细胞受体(BCR)刺激后,BMS-986142 抑制了Ramos B细胞中BCR刺激的钙流(IC50 = 9 nM)、扁桃体B细胞的增殖(IC50 = 3 nM)、外周血B细胞(IC50 = 4 nM)和记忆B细胞(IC50 = 3 nM)上CD86的表达,以及扁桃体B细胞产生TNF-α(IC50 = 3 nM)和IL-6(IC50 = 5 nM)。它不抑制CD40L诱导的CD86表达(IC50 >10,000 nM)。[2] 在Ramos B细胞中,BMS-986142 抑制了抗IgM刺激的磷脂酶C-γ2磷酸化(IC50 ≈20 nM)和BTK在Y551位点的自身磷酸化。[2] 在人外周血单个核细胞(PBMCs)中,BMS-986142 抑制了免疫复合物(IC)刺激的、FcγR依赖的TNF-α(IC50 = 3 nM)和IL-6(IC50 = 4 nM)的产生,但不抑制LPS(TLR4)刺激的TNF-α产生(IC50 >30,000 nM)。[2] 在全血实验中,BMS-986142 抑制了BCR刺激的B细胞上CD69表达(IC50 = 90 nM)和FcεRI刺激的嗜碱性粒细胞上CD63表达(IC50 = 89 nM)。[2] BMS-986142 剂量依赖性地抑制了RANK配体(RANK-L)诱导的人单核细胞前体细胞的破骨细胞生成,在浓度低至15 nM时即可观察到抑制作用。[2] |
| 体内研究 (In Vivo) |
在研究结束时,4、10 和 30 mg/kg 的 BMS-986142 导致临床明显疾病分别减少 26%、43% 和 79%,呈剂量依赖性。有趣的是,当与 MTX 一起给药时,4 mg/kg BMS-986142 在临床评分方面提供了额外的益处(54% 抑制),而单独使用 MTX 的抑制率为 19%。与单独使用任一药物分别为 24% 和 10% 相比,BMS-986142 4 mg/kg 与 MTX 联合给药可使炎症和骨吸收减少 53%。此外,10 和 30 mg/kg BMS-986142 显着抑制血清抗 II 型胶原蛋白 IgG 滴度。如果直到第 21 天才施用 BMS-986142(胶原蛋白加强剂),临床评分也会出现剂量依赖性下降。试验结束时,BMS-986142 2、4 和 25 mg/kg 剂量分别使临床评分降低 17%、37% 和 67%[2]。
在小鼠初次抗钥孔戚血蓝蛋白(KLH)抗体反应模型中,预防性口服给予 BMS-986142 (3, 10, 30 mg/kg,每日一次)剂量依赖性地抑制了抗KLH IgM(第7天)和IgG(第14天)抗体滴度,在10和30 mg/kg剂量下观察到显著降低。[2] 在胶原抗体诱导性关节炎(CAIA)小鼠模型中,预防性口服给予 BMS-986142 (5, 20 mg/kg,每日一次)降低了临床关节炎评分(分别抑制72%和>90%)以及炎症和骨吸收的组织学评分。[2] 在胶原诱导性关节炎(CIA)小鼠模型中,预防性口服给予 BMS-986142 (4, 10, 30 mg/kg,每日一次)剂量依赖性地降低了临床评分(抑制率分别为26%、43%、79%)、关节组织学损伤、血清抗II型胶原IgG滴度以及疾病相关的脾浆细胞增加。[2] 在CIA模型中,次优剂量的 BMS-986142 (4 mg/kg,每日一次)与甲氨蝶呤(MTX, 0.25 mg/kg,每日一次)联合给药,比单药治疗产生了对临床评分和组织学更大的抑制。[2] 在CIA模型中,从疾病发作开始进行治疗性口服给药 BMS-986142 (2, 4, 25 mg/kg,每日一次)剂量依赖性地降低了临床评分(抑制率分别为17%、37%、67%)并防止了骨丢失。[2] 在治疗性CIA模型中,低剂量 BMS-986142 (2 或 4 mg/kg,每日一次)与依那西普(15 mg/kg,每周两次,腹腔注射)联合给药,比任一单药治疗产生了显著更强的疗效,改善了临床评分、骨矿物密度和骨表面积。[2] 在预防性CIA模型中,BMS-986142 (10 mg/kg,每日一次)与鼠源CTLA-4-Ig(0.05 或 0.2 mg/kg,每周两次,腹腔注射)联合给药,在降低临床评分和组织学损伤方面比单药治疗产生了叠加益处。[2] |
| 酶活实验 |
测试化合物、荧光素肽 (1.5 μM)、ATP (20 μM)、人重组 BTK (1 nM) 和测定缓冲液(20 mM HEPES,pH 7.4、10 mM MgCl2、0.015% Brij 35 表面活性剂和 4 mM DTT,溶于将1.6%DMSO加入到V形底384孔板中,最终体积为30μL。室温孵育 60 分钟后,向每个样品中添加 45 μL 35 mM EDTA 以终止反应。通过电泳分离荧光底物和磷酸化产物以分析反应混合物。通过将抑制数据与无抑制剂的对照(0%抑制)和无酶的对照(100%抑制)进行比较,计算抑制水平。通过创建剂量反应曲线找到抑制 50% BTK 活性所需的浓度 (IC50)。将化合物溶解在 DMSO 中,浓度为 10 mM,并在 11 个浓度下进行评估。
对于主要激酶实验,将人重组BTK与荧光肽底物、ATP和 BMS-986142 在测定缓冲液中于室温下孵育。使用微流控迁移率变动分析系统对产物转化进行定量。对其他激酶(Tec, ITK, BLK, Txk, BMX)使用了类似的实验,ATP浓度设定为每种激酶的表观Km。[2] 使用竞争结合实验检测了高浓度(1000 nM)的 BMS-986142 与包含384种蛋白和脂质激酶的广泛面板的相互作用。此外,使用酶学实验评估了其在200 nM浓度下与包含337种激酶的面板的相互作用,其中测量了³²P-ATP掺入肽或蛋白底物的量。[2] |
| 细胞实验 |
Ramos B 细胞响应 B 细胞受体 (BCR) 的刺激而磷酸化磷脂酶 C (PLC)-γ2:将 Ramos B 细胞在含有 10% 胎牛血清 (FBS) 的培养基中于 37°C 预孵育一小时后使用不同浓度的 BMS-986142,使用 50 μg/mL 的 AffiniPure F(ab')2 片段山羊抗人免疫球蛋白 (Ig)M 在 37°C 下精确刺激细胞两分钟。然后加入冰冷的磷酸盐缓冲盐水来淬灭细胞。细胞裂解沉淀后,用兔抗人磷酸化Y759-PLCγ2进行免疫印迹测定PLCγ2的水平,并使用Odyssey红外成像系统分析结果。为了保证均匀加载,肌动蛋白控制被标准化。
对于刺激人外周血B细胞表达CD86和CD69,将PBMCs的E阴性组分在 BMS-986142 存在下,用抗人IgG+IgM F(ab')2片段刺激18小时。用抗CD20和抗CD86或CD69的抗体对细胞进行染色,在圈选CD20+ B细胞后,使用流式细胞术通过中位荧光强度(MFI)定量表达水平。对CD40或TLR4刺激使用了类似的实验设置。[2] 对于全血实验中BCR刺激的B细胞CD69表达,将人全血与 BMS-986142 孵育,并用抗人IgM F(ab')2片段和IL-4刺激18小时。对细胞进行CD20和CD69染色,裂解,固定,并通过流式细胞术分析。[2] 对于BCR刺激的人扁桃体B细胞产生IL-6和TNF-α,分离的细胞先用 BMS-986142 预孵育,然后用抗人IgM F(ab')2片段和IL-4刺激。4.5小时后收集上清液,通过酶免疫分析法测量细胞因子水平。[2] 对于BCR刺激的人扁桃体B细胞增殖,将细胞与 BMS-986142 一起培养,并用抗人IgG+IgM F(ab')2片段刺激72小时,随后通过液体闪烁计数法测量[³H]-胸腺嘧啶核苷掺入量。[2] 对于Ramos B细胞中BCR刺激的钙流,将负载染料的细胞铺板,用 BMS-986142 处理,并用抗人IgM刺激。使用荧光成像板读数仪测量钙流。[2] 对于Ramos B细胞中BCR刺激的PLCγ2磷酸化,先用 BMS-986142 预孵育的细胞用抗人IgM F(ab')2片段精确刺激2分钟,然后裂解。通过免疫印迹法测量磷酸化PLCγ2水平。[2] 对于Ramos B细胞中BCR刺激的BTK磷酸化,类似处理的细胞被刺激、固定,用Alexa647标记的抗磷酸化BTK(Y551)抗体染色,并通过流式细胞术分析。[2] 对于PBMCs中IC刺激的TNF-α和IL-6产生,用 BMS-986142 处理的细胞被预先形成的IgG免疫复合物刺激7小时。通过ELISA测量上清液中的细胞因子水平。[2] 对于RANK-L诱导的破骨细胞生成,将人单核细胞前体细胞与巨噬细胞集落刺激因子、RANK-L和 BMS-986142 一起培养9天。然后固定细胞,对抗酒石酸酸性磷酸酶(TRAP)进行染色,并计数TRAP阳性多核细胞的数量。[2] |
| 动物实验 |
小鼠:在雄性DBA/1小鼠中,于尾根部皮下注射200 μg混合的牛II型胶原蛋白。21天后对小鼠进行类似刺激。BMS-986142溶于乙醇:TPGS:PEG300 (5:5:90) 混合溶液中,用于立即开始每日一次口服给药,以进行预防性给药;然而,对于治疗性给药,给药时间推迟至加强免疫后的第21天。在用于BMS-986142联合MTX预防性研究的小鼠中,剂量如下:赋形剂;4、10或30 mg/kg的BMS-986142;4 mg/kg的BMS-986142联合0.25 mg/kg的MTX;或每日0.25 mg/kg的MTX。小鼠每天接受赋形剂、每天接受 2、4 或 25 mg/kg 的 BMS-986142、每天接受 2 或 4 mg/kg 的 BMS-986142 加每周两次腹腔注射依那西普 15 mg/kg,或每周两次腹腔注射依那西普 15 mg/kg,用于涉及 BMS-986142 加依那西普的治疗研究。小鼠每天接受载体、每天接受 10 或 30 mg/kg 的 BMS-986142、每周两次腹腔注射 0.05 或 0.2 mg/kg 的鼠 CTLA-4-Ig,或者每天接受 10 mg/kg 的 BMS-986142 加每周两次腹腔注射 0.05 或 0.2 mg/kg 的鼠 CTLA-4-Ig,用于涉及 BMS-986142 加鼠细胞毒性 T 淋巴细胞相关蛋白 4 免疫球蛋白 (CTLA-4-Ig) 的预防性研究。从第0天开始给药,持续36天的研究[2]。
在抗KLH抗体反应研究中,雌性BALB/c小鼠于第0天腹腔注射(IP)KLH进行免疫。小鼠每日一次(QD)通过灌胃(PO)给予BMS-986142(3、10、30 mg/kg),该药物配制于PEG 400:水(80:20 v/v)溶液中。分别于第7天和第14天采集血液,通过ELISA法分析抗KLH抗体滴度。[2] 在胶原抗体诱导的关节炎(CAIA)模型中,将抗胶原蛋白II单克隆抗体的混合物腹腔注射给雌性BALB/c小鼠。立即开始每日口服给予BMS-986142(3、10、30 mg/kg),其配制于乙醇:生育酚PEG 1000琥珀酸酯:PEG300(5:5:90)混合溶液中。三天后,腹腔注射LPS以增强关节炎。每日监测小鼠的爪部炎症情况。[2] 对于胶原诱导性关节炎(CIA)模型,雄性DBA/1小鼠于第0天皮下注射牛II型胶原蛋白佐剂乳化物进行免疫,并于第21天进行加强免疫。在预防性研究中,于第0天开始每日口服给予BMS-986142,其配制于乙醇:TPGS:PEG300(5:5:90)混合溶液中。在治疗性研究中,于第21天(加强免疫)开始给药。根据实验不同,剂量有所差异(例如,单药治疗剂量为 4、10 和 30 mg/kg;与依那西普联合用药剂量为 2、4 和 25 mg/kg)。加强免疫后,每周数次监测小鼠爪部炎症情况。在实验结束时采集血液样本,用于药物浓度和抗胶原蛋白 II 抗体的测定。脾脏用于流式细胞术分析。后爪采用微型 CT 和组织学方法进行分析。[2] 在 CIA 模型联合用药研究中,BMS-986142每日口服一次。联合用药包括:甲氨蝶呤(MTX,0.25 mg/kg,每日口服)、依那西普(15 mg/kg,每周两次腹腔注射)或鼠源 CTLA-4-Ig(0.05 或 0.2 mg/kg,每周两次腹腔注射)。[2] |
| 药代性质 (ADME/PK) |
在 KLH 研究中,于第 14 天测量了 BMS-986142 的血浆药物浓度。在 3、10 和 30 mg/kg PO QD 的剂量下,药物浓度分别在每天不到 11、14 和 17 小时内维持在小鼠全血 BCR 刺激的 CD69 IC50 (140 nM) 以上。 3 mg/kg 剂量组 24 小时后的血药浓度低于检测限。[2]
在 CAIA 模型中,每日一次口服 5 mg/kg 的剂量可提供约 16 小时的每日血药浓度高于小鼠全血 BCR CD69 IC50 水平,而 20 mg/kg 的剂量可提供接近完全(24 小时)的血药浓度覆盖。[2] 在预防性 CIA 模型中,每日一次口服 4、10 和 30 mg/kg 的剂量分别可提供 9、14 和 21 小时的每日血药浓度高于小鼠全血 BCR CD69 IC50 水平。[2] 在治疗性 CIA 模型中,每日一次口服 2、4 和 25 mg/kg 的剂量分别可提供不足 10、11 和 22 小时的每日血药浓度高于小鼠全血 BCR CD69 IC50 水平。[2] |
| 参考文献 |
|
| 其他信息 |
BMS-986142 正在临床试验 NCT02880670(放射性标记 BMS-986142 在健康男性受试者中的药代动力学和代谢研究)中进行研究。
BMS-986142 是一种新型的口服可逆小分子布鲁顿酪氨酸激酶 (BTK) 抑制剂。 BTK 是一种关键的细胞内酶,在 B 细胞和髓系细胞中表达,调控类风湿性关节炎 (RA) 病理生物学中的关键通路,包括 B 细胞受体 (BCR) 信号传导、Fcγ 受体 (FcγR) 依赖性细胞因子生成以及 RANK 配体 (RANK-L) 诱导的破骨细胞生成。[2] 该药物在 RA 小鼠模型(CIA 和 CAIA)中显示出疗效,无论是作为单药治疗还是与标准治疗药物(甲氨蝶呤、依那西普、CTLA-4-Ig)联合治疗,均显示出疗效,提示其在 RA 临床研究中具有潜力。[2] 在这些模型中观察到的疗效无需持续、完全抑制 BTK,因为仅需达到每日部分覆盖目标 IC50 的药物浓度即可有效。[2] BMS-986142 的可逆性使其能够进行精确的剂量滴定,并可能降低长期用药带来的风险。免疫抑制,尤其是在联合治疗中。[2] 在本文发表时,BMS-986142 的安全性和有效性正在类风湿性关节炎和原发性干燥综合征的临床试验中进行评估。[2] |
| 分子式 |
C32H30F2N4O4
|
|
|---|---|---|
| 分子量 |
572.61
|
|
| 精确质量 |
572.223
|
|
| 元素分析 |
C, 67.12; H, 5.28; F, 6.64; N, 9.78; O, 11.18
|
|
| CAS号 |
1643368-58-4
|
|
| 相关CAS号 |
|
|
| PubChem CID |
86582336
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| 密度 |
1.4±0.1 g/cm3
|
|
| 沸点 |
746.5±70.0 °C at 760 mmHg
|
|
| 闪点 |
405.3±35.7 °C
|
|
| 蒸汽压 |
0.0±2.6 mmHg at 25°C
|
|
| 折射率 |
1.667
|
|
| LogP |
4.51
|
|
| tPSA |
120
|
|
| 氢键供体(HBD)数目 |
3
|
|
| 氢键受体(HBA)数目 |
6
|
|
| 可旋转键数目(RBC) |
4
|
|
| 重原子数目 |
42
|
|
| 分子复杂度/Complexity |
1090
|
|
| 定义原子立体中心数目 |
1
|
|
| SMILES |
FC1C=C(C(N)=O)C2=C(C=1C1C=CC=C(C=1C)N1C(N(C)C3C(=CC=CC=3C1=O)F)=O)C1=C(C[C@@H](C(C)(C)O)CC1)N2
|
|
| InChi Key |
ZRYMMWAJAFUANM-INIZCTEOSA-N
|
|
| InChi Code |
InChI=1S/C32H30F2N4O4/c1-15-17(7-6-10-24(15)38-30(40)19-8-5-9-21(33)28(19)37(4)31(38)41)25-22(34)14-20(29(35)39)27-26(25)18-12-11-16(32(2,3)42)13-23(18)36-27/h5-10,14,16,36,42H,11-13H2,1-4H3,(H2,35,39)/t16-/m0/s1
|
|
| 化学名 |
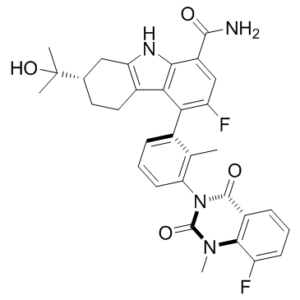
(7S)-3-fluoro-4-[3-(8-fluoro-1-methyl-2,4-dioxoquinazolin-3-yl)-2-methylphenyl]-7-(2-hydroxypropan-2-yl)-6,7,8,9-tetrahydro-5H-carbazole-1-carboxamide
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.08 mg/mL (3.63 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 20.8 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: 2.08 mg/mL (3.63 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 悬浊液; 超声助溶。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.08 mg/mL (3.63 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7464 mL | 8.7319 mL | 17.4639 mL | |
| 5 mM | 0.3493 mL | 1.7464 mL | 3.4928 mL | |
| 10 mM | 0.1746 mL | 0.8732 mL | 1.7464 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT02257151 | Completed | Drug: BMS-986142 Drug: Placebo |
Healthy Adult | Bristol-Myers Squibb | September 2014 | Phase 1 |
| NCT02880670 | Completed | Drug: BMS-986142 | Arthritis | Bristol-Myers Squibb | August 2016 | Phase 1 |
| NCT02832180 | Completed | Drug: OC containing EE and NET Drug: BMS-986142 |
Arthritis | Bristol-Myers Squibb | May 2016 | Phase 1 |
| NCT02762123 | Completed | Drug: BMS-986142 200mg Drug: BMS-986142 350mg |
Rheumatoid Arthritis | Bristol-Myers Squibb | May 2016 | Phase 1 |
| NCT02638948 | Completed | Drug: BMS-986142 Drug: Placebo |
Rheumatoid Arthritis | Bristol-Myers Squibb | February 16, 2016 | Phase 2 |
BMS-986142 inhibits RANK-L-induced osteoclastogenesis in human monocytic precursors.PLoS One.2017 Jul 24;12(7):e0181782. |
BMS-986142 blocks neoantigen-induced antibody responses.PLoS One.2017 Jul 24;12(7):e0181782. |
BMS-986142 is efficacious against CIA in mice.PLoS One.2017 Jul 24;12(7):e0181782. |
Therapeutic treatment with BMS-986142 co-administered with etanercept protected from CIA in mice.PLoS One.2017 Jul 24;12(7):e0181782. |
BMS-986142 co-administered with CTLA-4-Ig shows an enhanced effect against CIA in mice.
Inhibition of anti-IgM-stimulated phosphorylation of phospholipase C-γ2 in Ramos B cells by BMS-986142.PLoS One.2017 Jul 24;12(7):e0181782. |
BMS-986142 is efficacious in the murine CAIA model.PLoS One.2017 Jul 24;12(7):e0181782. |