| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|
| 靶点 |
Bruton’s tyrosine kinase (BTK) [1, 2].
IC₅₀: 0.69 ± 0.091 nM (for BTK kinase activity inhibition) [2]. Cereblon (CRBN) [2]. IC₅₀: 316 ± 82 nM (for competitive binding to CRBN-DDB1 complex against biotin-labelled thalidomide) [2]. DC50 unknown |
|---|---|
| 体外研究 (In Vitro) |
BTK 降解:BGB-16673 在体外强效降解 BTK。在人全血中,亚纳摩尔未结合浓度下 2-3 小时内可实现接近完全的 BTK 降解。在加入人血清的 TMD-8 细胞中也观察到同样强效的降解 [2]。
BTK 激酶抑制:BGB-16673 对 BTK 激酶活性有强效抑制作用,IC₅₀ 为 0.69 ± 0.091 nM (n=3) [2]。 CRBN 结合:BGB-16673 与 CRBN-DDB1 复合物竞争性结合,拮抗生物素标记的沙利度胺,IC₅₀ 为 316 ± 82 nM (n=3) [2]。 细胞活力:BGB-16673 在野生型 TMD-8 淋巴瘤细胞以及表达临床相关 BTK 突变(包括 C481S、T474I 和 L528W)的 TMD-8 细胞系中均表现出强效的抗增殖活性,其活性优于 ibrutinib 和 pirtobrutinib [1]。 选择性:BGB-16673 对 BTK 的结合亲和力(IC₅₀ 0.69 nM)约是对 E3 连接酶 CRBN 结合亲和力(IC₅₀ 316 nM)的 500 倍,这一关键设计特征使得模型简化成为可能 [2]。 |
| 体内研究 (In Vivo) |
小鼠 TMD-8 异种移植模型:在皮下接种 TMD-8 肿瘤的 NCG 小鼠中,口服给予 BGB-16673(6 和 20 mg/kg,临床可达剂量)以剂量依赖性方式有效抑制肿瘤生长。在两个剂量下,BGB-16673 的疗效均优于临床相关剂量的 BTK 抑制剂(ibrutinib、acalabrutinib、zanubrutinib 和 pirtobrutinib)[1]。
小鼠 PK/PD 研究:在小鼠口服给药后(单次剂量 2、6 和 20 mg/kg;以及 3 mg/kg 每日一次连续 5 天),BGB-16673 在血液和 TMD-8 肿瘤中诱导快速且剂量依赖性的 BTK 降解。单次 20 mg/kg 剂量后(未结合 Cmax ~1.5 nM),血液和肿瘤中均达到接近完全的 BTK 降解。连续 5 天每日给予 3 mg/kg 后,血液中实现 >90% 的 BTK 降解,肿瘤细胞中实现 >70% 的降解 [2]。 基因表达分析:对药物处理的肿瘤进行 RNA-seq 分析显示,BGB-16673 诱导了与 BTK 抑制剂不同的生物学效应。GO 富集分析表明,BGB-16673 对细胞周期相关基因、NFκB 靶点以及某些 Bcl-2 家族成员的抑制更强,与其优越的疗效相关 [1]。 |
| 酶活实验 |
BTK 激酶活性实验:采用时间分辨荧光共振能量转移法进行。BTK 蛋白(aa 393-659,His 标签)与 BGB-16673 在室温孵育 60 分钟。通过加入 ATP(47 μM)和 AQ15 肽(0.4 μM)启动反应,缓冲液含 50 mM Tris pH 7.5、10 mM MgCl₂、2 mM MnCl₂、0.1 mM EDTA、0.01% BSA、1 mM DTT、0.005% Tween-20 和 20 mM SEB。反应 60 分钟后,加入含 MAbPT66-K 和 Streptavidin-XL665 的终止/检测液。记录 TR-FRET 信号(激发 337 nm,发射 665 nm/620 nm)。通过将数据拟合至四参数 logistic 方程计算 IC₅₀ [2]。
CRBN-DDB1 结合实验:采用 TR-FRET 方法。CRBN(aa 40-442)和 DDB1(1-1140)蛋白复合物与 BGB-16673 在室温孵育 60 分钟。加入生物素标记的沙利度胺(20 nM),随后加入检测试剂(Mab Anti-6His Eu cryptate Gold 和 Streptavidin-XL665)。记录 TR-FRET 信号(激发 337 nm,发射 665 nm/620 nm)。通过将数据拟合至四参数 logistic 方程计算 IC₅₀ [2]。 |
| 细胞实验 |
体外全血 BTK 降解实验:将人或小鼠全血与 BGB-16673(0.05-12,000 nM)孵育 0、1、3、6 或 24 小时(每个时间点 n=2)。处理后裂解细胞,通过 ELISA 检测 BTK 蛋白 [2]。
体外 TMD-8 细胞 BTK 降解实验:TMD-8 细胞在含 10% FBS 的 RPMI1640 中培养。药物处理前,将培养基替换为人或小鼠血清。细胞与 BGB-16673(0.32-5000 nM)孵育 0、1、3 或 6 小时(每个时间点 n=3)。处理后裂解细胞,通过 HTRF 检测 BTK 蛋白 [2]。 细胞活力/增殖实验:在野生型和 BTK 突变型 TMD-8 细胞系中评估抗增殖活性。详细方案未提供 [1]。 |
| 动物实验 |
小鼠肿瘤异种移植模型:将2 × 10⁵个TMD-8肿瘤细胞皮下植入6-8周龄的雌性NCG小鼠体内。当平均肿瘤体积达到约200 mm³时,将小鼠随机分为治疗组(每个时间点每组n=4)。BGB-16673溶于0.5%甲基纤维素溶液中,并以10 mL/kg体重进行口服给药。单次给药研究中,分别给予2、6和20 mg/kg的剂量。重复给药研究中,每日给予3 mg/kg,连续5天。给药后不同时间点采集血液和肿瘤样本(2 和 6 mg/kg 剂量组为 4、8、24 和 30 小时;20 mg/kg 剂量组为 4、8、24、30、48、72 和 120 小时;重复给药组为末次给药后 0、4、8、24、30、48、72 和 120 小时)[2]。
小鼠药代动力学/药效学研究:口服给药后,用 K₂EDTA 抗凝管采集血液样本。离心(1000 g,10 分钟)获得血浆。采用蛋白沉淀法,然后进行 LC-MS/MS 分析,测定血浆中 BGB-16673 的浓度。血液和肿瘤中 BTK 蛋白水平的测定方法如上所述[2]。 |
| 参考文献 |
|
| 分子式 |
C47H54N12O4
|
|---|---|
| 分子量 |
851.01
|
| 精确质量 |
850.439098
|
| 元素分析 |
C, 66.33; H, 6.40; N, 19.75; O, 7.52
|
| CAS号 |
2736508-60-2
|
| PubChem CID |
166521972
|
| 外观&性状 |
Light yellow to green yellow solid powder
|
| 密度 |
1.284±0.06 g/cm3(Temp: 25 °C; Press: 760 Torr)(predicted)
|
| LogP |
6.2
|
| tPSA |
182 Ų
|
| 氢键供体(HBD)数目 |
3
|
| 氢键受体(HBA)数目 |
12
|
| 可旋转键数目(RBC) |
11
|
| 重原子数目 |
63
|
| 分子复杂度/Complexity |
1560
|
| 定义原子立体中心数目 |
1
|
| SMILES |
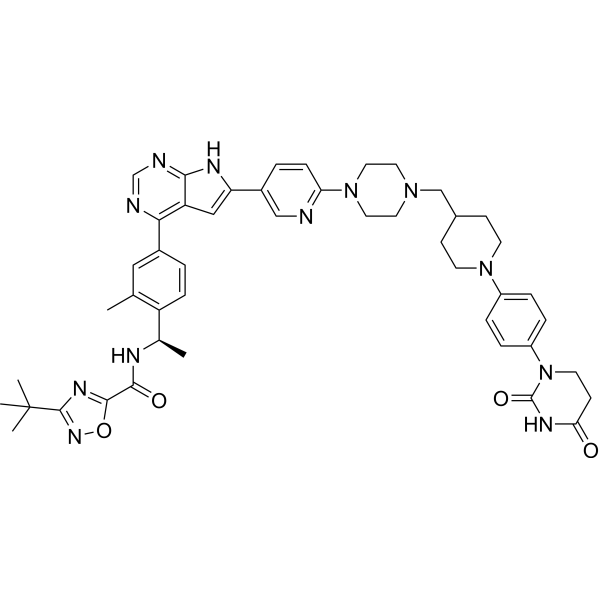
C12C=C(C3=CC=C(N4CCN(CC5CCN(C6C=CC(N7CCC(=O)NC7=O)=CC=6)CC5)CC4)N=C3)NC=1N=CN=C2C1C=CC([C@@H](C)NC(=O)C2ON=C(C(C)(C)C)N=2)=C(C)C=1
|
| InChi Key |
ZSOLMVZWDSGPDD-SSEXGKCCSA-N
|
| InChi Code |
InChI=1S/C47H54N12O4/c1-29-24-32(6-12-36(29)30(2)51-43(61)44-54-45(55-63-44)47(3,4)5)41-37-25-38(52-42(37)50-28-49-41)33-7-13-39(48-26-33)58-22-20-56(21-23-58)27-31-14-17-57(18-15-31)34-8-10-35(11-9-34)59-19-16-40(60)53-46(59)62/h6-13,24-26,28,30-31H,14-23,27H2,1-5H3,(H,51,61)(H,49,50,52)(H,53,60,62)/t30-/m1/s1
|
| 化学名 |
3-tert-butyl-N-[(1R)-1-[4-[6-[6-[4-[[1-[4-(2,4-dioxo-1,3-diazinan-1-yl)phenyl]piperidin-4-yl]methyl]piperazin-1-yl]-3-pyridinyl]-7H-pyrrolo[2,3-d]pyrimidin-4-yl]-2-methylphenyl]ethyl]-1,2,4-oxadiazole-5-carboxamide
|
| 别名 |
BTK-IN-29; BGB16673; BGB-16673; BGB 16673; Catadegbrutinib; 2736508-60-2; PF6GPZ4DYT; RefChem:1081283;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
Soluble in DMSO: ~125 mg/mL (146.9 mM)
|
|---|---|
| 溶解度 (体内实验) |
注意: 如下所列的是一些常用的体内动物实验溶解配方,主要用于溶解难溶或不溶于水的产品(水溶度<1 mg/mL)。 建议您先取少量样品进行尝试,如该配方可行,再根据实验需求增加样品量。
注射用配方
注射用配方1: DMSO : Tween 80: Saline = 10 : 5 : 85 (如: 100 μL DMSO → 50 μL Tween 80 → 850 μL Saline)(IP/IV/IM/SC等) *生理盐水/Saline的制备:将0.9g氯化钠/NaCl溶解在100 mL ddH ₂ O中,得到澄清溶液。 注射用配方 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (如: 100 μL DMSO → 400 μL PEG300 → 50 μL Tween 80 → 450 μL Saline) 注射用配方 3: DMSO : Corn oil = 10 : 90 (如: 100 μL DMSO → 900 μL Corn oil) 示例: 以注射用配方 3 (DMSO : Corn oil = 10 : 90) 为例说明, 如果要配制 1 mL 2.5 mg/mL的工作液, 您可以取 100 μL 25 mg/mL 澄清的 DMSO 储备液,加到 900 μL Corn oil/玉米油中, 混合均匀。 View More
注射用配方 4: DMSO : 20% SBE-β-CD in Saline = 10 : 90 [如:100 μL DMSO → 900 μL (20% SBE-β-CD in Saline)] 口服配方
口服配方 1: 悬浮于0.5% CMC Na (羧甲基纤维素钠) 口服配方 2: 悬浮于0.5% Carboxymethyl cellulose (羧甲基纤维素) 示例: 以口服配方 1 (悬浮于 0.5% CMC Na)为例说明, 如果要配制 100 mL 2.5 mg/mL 的工作液, 您可以先取0.5g CMC Na并将其溶解于100mL ddH2O中,得到0.5%CMC-Na澄清溶液;然后将250 mg待测化合物加到100 mL前述 0.5%CMC Na溶液中,得到悬浮液。 View More
口服配方 3: 溶解于 PEG400 (聚乙二醇400) 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.1751 mL | 5.8754 mL | 11.7507 mL | |
| 5 mM | 0.2350 mL | 1.1751 mL | 2.3501 mL | |
| 10 mM | 0.1175 mL | 0.5875 mL | 1.1751 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。