| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 2mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| Other Sizes |
|
| 靶点 |
YAP/TEAD interaction
The target of CA3 (CIL-56) is Yes-associated protein 1 (YAP1)/Tead transcriptional complex; [1] |
|---|---|
| 体外研究 (In Vitro) |
在体外,CA3显着减少食管腺癌细胞的生长。诱导细胞凋亡、减少肿瘤球形成和减少 ALDH1+ 细胞的数量都是 CA3 可能的作用。在 293T 细胞中共转染每个转录因子的单独启动子荧光素酶后,CA3 特异性抑制 Tead/YAP1 转录活性,但对 Super-TOP/Wnt、CBF1/Notch 或 AP-1 没有影响。抗辐射食管腺癌细胞中富含的 CSC 特性优先被 CA3 抑制[1]。
1. 抑制YAP1/Tead转录活性:在共转染Gal4-Tead、5×UAS-荧光素酶和YAP1 cDNA的293T细胞中,与其他化合物(A414、A432、A413、A433、VP)相比,CA3 (CIL-56)处理可显著降低YAP1/Tead荧光素酶报告基因活性。此外,在SKGT-4和JHESO食管腺癌(esophageal adenocarcinoma, EAC)细胞中,通过相同的荧光素酶报告基因实验发现,CA3 (CIL-56)可呈剂量依赖性降低YAP1/Tead转录活性[1] 2. 对EAC细胞的抗增殖活性:4种EAC细胞系(SKGT-4、Flo-1、JHESO、OACP)经CA3 (CIL-56)(0.1µM、0.5µM、1µM)处理后,MTS实验结果显示,与0.1% DMSO对照组相比,细胞增殖和活力呈剂量依赖性抑制。对于经多西环素诱导YAP1过表达的SKGT-4细胞(DOX+),CA3 (CIL-56)的抗增殖效应比未诱导YAP1的细胞(DOX−)更显著[1] 3. 诱导细胞周期阻滞和凋亡:SKGT-4和JHESO细胞经CA3 (CIL-56)(0.5µM、1µM)处理24h和48h后,经碘化丙啶染色并通过流式细胞术分析。结果显示,与对照组相比,细胞周期时相分布发生改变(细胞周期阻滞),且肿瘤细胞死亡指数显著升高[1] 4. 下调YAP1和SOX9表达:免疫印迹分析表明,CA3 (CIL-56)可呈剂量依赖性降低SKGT-4和JHESO细胞中YAP1及其下游靶蛋白SOX9的表达水平。实时定量聚合酶链反应(quantitative real-time PCR, qPCR)进一步证实,CA3 (CIL-56)可降低这些细胞中YAP1的mRNA水平。免疫荧光染色也显示,经CA3 (CIL-56)处理的JHESO细胞中YAP1和SOX9的表达降低[1] 5. 抑制辐射抵抗性EAC细胞的癌症干细胞(cancer stem cell, CSC)特性:Flo-1 XTR(辐射抵抗性EAC细胞)比亲本Flo-1-P细胞具有更强的CSC特性(肿瘤球形成能力增强、ALDH1+细胞比例升高)。CA3 (CIL-56)呈剂量依赖性抑制Flo-1-P和Flo-1 XTR细胞的肿瘤球形成,且对Flo-1 XTR细胞的抑制效应更显著。此外,CA3 (CIL-56)(0.5µM处理48h)可降低Flo-1 XTR细胞中ALDH1+细胞的比例,并下调这些辐射抵抗性细胞中YAP1、磷酸化EGFR(phospho-EGFR)和磷酸化S6(phospho-S6)的蛋白水平[1] 6. 与5-氟尿嘧啶(5-FU)的协同作用:4种EAC细胞系(SKGT-4、JHESO、OACP、Yes-6)及YAP1过表达的SKGT-4(DOX+)细胞经CA3 (CIL-56)与5-FU单独或联合处理后,MTS实验显示,与单独用药或对照组相比,联合处理可显著增强细胞生长抑制作用,且在YAP1高表达的EAC细胞中协同效应更强[1] 7. 对YAP1/Tead通路的特异性:在转染YAP1/Tead、Super-TOP/Wnt、CBF1/Notch或AP-1通路报告质粒的293T细胞中,CA3 (CIL-56)(0.5µM、1µM)仅特异性抑制YAP1/Tead的荧光素酶活性,而不影响Wnt、Notch或AP-1通路的活性[1] |
| 体内研究 (In Vivo) |
在异种移植模型中,CA3 表现出有效的抗肿瘤活性,且没有明显的毒性[1]。
CA3在可诱导的高YAP异种移植小鼠体内模型中具有较强的抗肿瘤作用[1] 为了进一步证实CA3靶向YAP1在体内的抗肿瘤作用,我们利用可诱导的YAP1高SKGT-4 (Dox+)细胞异种移植模型。以每只小鼠1 × 106个细胞的剂量给裸鼠植入Dox -和Dox+ SKGT-4细胞。在这个浓度下,我们观察到在注射10-14天后,只有Dox+ SKGT-4细胞能够形成肿瘤(图5B),这表明YAP1是体内驱动肿瘤生长所必需的。为了确定CA3对yap诱导的肿瘤生长的抑制作用,我们将携带Dox+ SKGT-4 EAC异种移植物的小鼠随机分为两组,然后分别给予对照磷酸盐缓冲盐水或1mg /kg的CA3治疗。在我们3周的给药计划结束时,测量SKGT-4异种移植肿瘤的重量和体积以及小鼠的体重。体内SKGT-4 Dox+异种移植物模型的结果表明,CA3处理的Dox+ SKGT-4异种移植物小鼠体内肿瘤大小和重量大大减少(图5b&5c),而在整个实验期间,植入Dox - SKGT-4细胞的小鼠未形成肿瘤(图5B)。CA3处理组与对照组小鼠体重无显著差异(图5D)。此外,免疫组织化学在小鼠肿瘤组织中进一步证实,CA3处理sktt -4 Dox+后,YAP1、SOX9和KI67的表达显著降低(图5E)。因此,CA3在体内有效抑制EAC肿瘤生长,这些作用至少部分归因于抑制干性基因YAP1和SOX9。 CA3与5-FU协同抑制EAC细胞体外和体内生长[1] 为了确定CA3单独或与5-FU联合处理对EAC细胞系生长的抑制作用,我们首先在96孔板中播种4个YAP1组成高表达的EC细胞系(sktt -4、JHESO、OACP和YES-6),并分别用CA3单独、5-FU单独或CA3与5-FU在指定浓度下联合处理。图6A的结果表明,尽管CA3对这四种细胞系的生长产生了剂量依赖性的降低,但CA3与5-FU的结合对其生长的抑制作用显著,尤其是CA3与5-FU的结合作用最大。为了进一步研究CA3对EAC细胞的抑制是否依赖于YAP1,我们用CA3单独、5-FU单独或联合处理Dox+ (YAP1诱导)和Dox - SKGT4 (PIN20YAP1)细胞,我们发现CA3单独与YAP1低SKGT-4细胞(Dox−)相比,优先抑制YAP1高SKGT-4细胞(Dox+)的生长,并且呈剂量依赖性(图6B)。此外,CA3和5-FU联合处理对Dox+和Dox−SKGT-4细胞的生长均产生最大的抑制作用(图6b)。这些结果表明,CA3与5-FU在GAC细胞生长抑制中的协同作用。 1. 在YAP1诱导型异种移植模型中的抗肿瘤活性:将具有YAP1诱导表达能力的SKGT-4细胞(PIN20YAP1)皮下接种于裸鼠(每组5只,双侧接种)。小鼠分为有无多西环素(DOX)处理组(以诱导YAP1表达),并给予CA3 (CIL-56)或溶媒对照处理。结果显示,在DOX+组(YAP1过表达肿瘤)中,CA3 (CIL-56)处理显著降低肿瘤重量,且对小鼠体重无显著影响,表明其耐受性良好[1] 2. 在JHESO异种移植模型中的抗肿瘤活性:将JHESO细胞(1.5×10⁶个/位点)皮下接种于裸鼠(每组5只,双侧接种)。小鼠分别接受CA3 (CIL-56)单独处理、5-FU单独处理或两者联合处理。结果显示,联合处理组的肿瘤体积缩小程度显著大于单独用药组或溶媒对照组。肿瘤组织的免疫组织化学(immunohistochemistry, IHC)分析表明,CA3 (CIL-56)(单独或与5-FU联合)可降低YAP1、SOX9和Ki67(增殖标志物)的表达[1] |
| 酶活实验 |
SOX9 荧光素酶报告基因之前已有描述。之前也描述过的 5 × -UAS-荧光素酶报告基因和 Gal4-TEAD4 构建体是从 MD 安德森癌症中心的 Johnson 博士获得的。如前所述,用 SOX9 荧光素酶报告基因和 Renilla 载体或 5 × -UAS-荧光素酶报告基因和 Gal4-TEAD4 与 CMV-β-gal 构建体瞬时共转染食管腺癌细胞。
|
| 细胞实验 |
将 SKGT-4 和 JHESO 细胞接种到 DMEM 中的 6 孔板(1 × 105/孔)中,培养 24 小时以允许细胞贴壁。然后将细胞按照推荐的不同剂量暴露于 0.1% DMSO(对照)或 CA3 中 48 小时。然后收集细胞,用甲醇固定,洗涤,用RNase A处理,用碘化丙啶进行DNA染色,并进行DNA直方图和细胞周期阶段分布的流式细胞术分析。
细胞增殖试验[1] 用0.1%二甲亚砜(对照)、不同剂量的<强>CA3处理EAC细胞及其耐药对应细胞。对于联合处理实验,按指示用<强>CA3、5-FU或不同浓度的联合处理细胞6天,并使用前面描述的MTS法评估细胞活力。所有试验一式三份,至少重复三次。 流式细胞术及凋亡分析[1] 采用流式细胞术对EAC细胞凋亡进行分析。简而言之,将SKGT-4和JHESO细胞在Dulbecco改良Eagle培养基中接种到六孔板上(每孔1 × 105),培养24小时以使细胞附着。然后用0.1%二甲亚砜(对照)或CA3按不同剂量处理细胞48小时。接下来,收集细胞,用甲醇固定,清洗,用RNase A处理,用碘化丙啶染色,用流式细胞仪分析细胞的DNA直方图和细胞周期分布。 蛋白的提取和Western blot分析[1] 从经CA3处理的EAC细胞中分离出蛋白质,并按照前面描述的方法进行Western blotting分析。 1. YAP1/Tead转录活性实验(荧光素酶报告基因实验):将293T细胞或EAC细胞(SKGT-4、JHESO)共转染Gal4-Tead质粒、5×UAS-荧光素酶报告质粒和YAP1 cDNA质粒(以过表达YAP1)。转染后,用不同浓度(0.5µM、1µM或其他指定剂量)的CA3 (CIL-56)处理细胞48h,随后检测荧光素酶活性,以评估CA3 (CIL-56)对YAP1/Tead转录活性的抑制作用。为检测通路特异性,将细胞转染Super-TOP/Wnt、CBF1/Notch或AP-1通路的报告质粒,再经CA3 (CIL-56)处理并检测荧光素酶活性[1] 2. 细胞增殖/活力实验(MTS实验):将EAC细胞(SKGT-4、Flo-1、JHESO、OACP、Yes-6,包括SKGT-4 DOX+和DOX−细胞、Flo-1-P和Flo-1 XTR细胞)接种于适宜的培养板中。细胞贴壁后,用CA3 (CIL-56)(0.1µM、0.5µM、1µM)单独、5-FU单独或两者联合(指定浓度)处理6天(协同实验)或其他指定时长。向各孔中加入CellTiter Aqueous One Solution(MTS试剂),检测490nm处的吸光度(OD490),并以0.1% DMSO对照组为参照,计算细胞增殖/活力百分比[1] 3. 细胞周期与凋亡分析(流式细胞术):将SKGT-4和JHESO细胞接种于6孔板中,用CA3 (CIL-56)(0.5µM、1µM)或0.1% DMSO处理24h和48h。收集细胞、固定后用碘化丙啶染色,通过流式细胞术分析DNA直方图以确定细胞周期时相分布,并计算肿瘤细胞死亡指数[1] 4. 免疫印迹实验:用指定剂量的CA3 (CIL-56)处理EAC细胞(SKGT-4、JHESO、Flo-1-P、Flo-1 XTR),制备细胞裂解液,经凝胶电泳分离蛋白后转移至膜上,用抗YAP1、SOX9、phospho-EGFR、phospho-S6或其他相关蛋白的抗体进行孵育。采用标准免疫印迹流程进行检测,并对蛋白水平进行半定量分析[1] 5. 实时定量PCR(qPCR):用指定剂量的CA3 (CIL-56)处理SKGT-4和JHESO细胞,提取总RNA并合成互补DNA(cDNA)。使用YAP1特异性引物进行qPCR,以合适的内参基因(如GAPDH)为参照,采用2⁻ΔΔCt法计算YAP1 mRNA的相对表达量[1] 6. 免疫荧光染色:用CA3 (CIL-56)或对照处理JHESO细胞,固定并透化后,加入抗YAP1和SOX9的一抗孵育,再用荧光标记的二抗孵育。通过共聚焦显微镜观察并定量YAP1和SOX9的表达[1] 7. 肿瘤球形成实验:将Flo-1-P和Flo-1 XTR细胞以低密度接种于超低吸附培养板中,在培养开始时加入指定浓度的CA3 (CIL-56)。培养8~10天后,在显微镜下计数直径>50µm的肿瘤球,拍摄球体制备图像,通过球体数量量化评估CSC的自我更新能力[1] 8. ALDH1+细胞比例分析:用ALDH1标记试剂盒对Flo-1、Flo-1 XTR细胞及经CA3 (CIL-56)(0.5µM处理48h)处理的Flo-1 XTR细胞进行染色,通过流式细胞术检测并定量ALDH1+细胞(CSC标志物)的百分比[1] |
| 动物实验 |
PBS;1 mg/kg;腹腔注射
JHESO 食管腺癌异种移植小鼠模型 体内异种移植小鼠模型[1] 体内实验均按照机构动物护理和使用委员会 (IACUC) 的规定进行。将 SKGT-4 (PIN20YAP1) 细胞 (1 × 10⁶) 接种到裸鼠体内(每组 n = 5),其中部分细胞经强力霉素诱导表达 YAP1(Dox− 组),部分细胞经强力霉素诱导表达 YAP1(Dox+ 组)。Dox+ 组小鼠饮用含 2.5% 蔗糖和 2.5% 强力霉素的水,而 Dox− 组小鼠饮用仅含 2.5% 蔗糖的水。 10天后,Dox+组小鼠腹腔注射CA3(1 mg/kg/只),每周三次,共3周。 在JHESO异种移植EAC模型中,将2 × 10⁶个JHESO细胞皮下注射到裸鼠体内(n = 5/组)。约10天后,小鼠接受腹腔注射CA3(1 mg/kg/只)、5-FU(30 mg/kg/只)或二者联合用药,每周三次,共3周。对照组注射磷酸盐缓冲液(100 µl/只)。小鼠的肿瘤体积、肿瘤重量和体重按先前描述的方法进行测量(4)。所有测量结果均采用非配对 Student t 检验进行比较。 1. YAP1 诱导型 SKGT-4 异种移植实验: - 细胞制备:培养并收集处于对数生长期的 YAP1 诱导型 SKGT-4 细胞(PIN20YAP1)。 - 异种移植建立:将 SKGT-4 (PIN20YAP1) 细胞皮下接种到裸鼠(每组 5 只)的两个部位(左侧和右侧腹部)。 - 治疗分组:将小鼠分为接受或未接受强力霉素 (DOX) 治疗(以诱导 YAP1 表达)的组,并分别用 CA3 (CIL-56) 或载体对照进行治疗。 CA3 (CIL-56) 的具体剂量和给药频率/途径未明确说明,但治疗持续了 6 周。 - 终点:6 周后,处死小鼠,切除肿瘤并称重。定期测量小鼠体重以监测毒性。收集肿瘤组织进行YAP1、SOX9和Ki67的免疫组织化学(IHC)分析[1] 2. JHESO异种移植实验: - 细胞制备:培养JHESO EAC细胞,并制备每点1.5×10⁶个细胞用于注射。 - 异种移植瘤建立:将JHESO细胞皮下接种于裸鼠(每组5只)的两个部位(左侧和右侧腹部)。 - 治疗分组:将小鼠随机分为四组:载体对照组、CA3 (CIL-56)单药组、5-FU)单药组或CA3 (CIL-56)+5-FU联合组。 CA3 (CIL-56) 和 5-FU 的具体剂量以及给药频率/途径并未明确说明,但治疗持续至研究终点。 - 研究终点:定期使用游标卡尺测量肿瘤体积(肿瘤体积 = 长 × 宽² / 2)。监测小鼠体重以评估耐受性。研究结束时,收集肿瘤组织进行 YAP1、SOX9 和 Ki67 的免疫组化分析 [1] |
| 毒性/毒理 (Toxicokinetics/TK) |
唯一与毒性相关的观察结果是,在两种异种移植模型中,使用CA3 (CIL-56)(单独使用或与5-FU联合使用)治疗均未导致小鼠体重显著下降,表明在测试剂量下体内耐受性良好。文献[1]中未提供CA3 (CIL-56)的半数致死量(LD50)、肝毒性、肾毒性、药物相互作用或血浆蛋白结合率的数据。
|
| 参考文献 | |
| 其他信息 |
越来越多的证据表明,Hippo共激活因子Yes相关蛋白1 (YAP1) 是癌症干细胞 (CSC) 特性、肿瘤进展和治疗耐药性的主要介质,并且通常是许多致癌通路的终末节点。因此,靶向YAP1可能成为治疗多种YAP1高表达肿瘤(包括食管腺癌)的新型治疗策略。然而,目前尚缺乏有效的YAP1抑制剂。本文鉴定了一种小分子化合物(CA3),它不仅对YAP1/Tead转录活性具有显著的抑制作用,而且在体外和体内均表现出对食管腺癌细胞生长,尤其是YAP1高表达食管腺癌细胞的强效抑制作用。值得注意的是,放射抗性细胞会获得强大的癌症干细胞(CSC)特性和侵袭性表型,而CA3可以通过抑制细胞增殖、诱导细胞凋亡、减少肿瘤球形成以及降低ALDH1+细胞比例来有效抑制这些表型。此外,CA3与5-FU联用可协同抑制食管腺癌细胞的生长,尤其是在YAP1高表达的食管腺癌细胞中。综上所述,这些发现表明CA3是一种新型的YAP1抑制剂,主要靶向具有CSC特性的YAP1高表达和耐药性食管腺癌细胞。Mol Cancer Ther; 17(2); 443-54. ©2017 AACR.
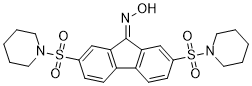
1. 背景:Yes 相关蛋白 1 (YAP1) 是 Hippo 通路中的一种共激活因子,是多种癌症(包括食管腺癌 (EAC))中癌症干细胞 (CSC) 特性、肿瘤进展和治疗耐药性的关键介质。YAP1 通常是多个致癌通路的末端节点,使其成为潜在的治疗靶点。然而,此前缺乏有效的 YAP1 抑制剂 [1] 2. 作用机制:CA3 (CIL-56) 主要通过特异性抑制 YAP1/Tead 转录复合物发挥其抗肿瘤作用。这种抑制作用导致下游效应,包括 YAP1 及其靶基因 SOX9 的下调、CSC 特性的抑制(肿瘤球形成减少、ALDH1+ 细胞减少)、诱导细胞周期阻滞和凋亡以及抑制细胞增殖。在放射抗性EAC细胞中,CA3 (CIL-56) 还能下调磷酸化EGFR和磷酸化S6,而磷酸化EGFR和磷酸化S6与治疗耐药性相关[1] 3. 治疗潜力:CA3 (CIL-56) 对YAP1高表达的EAC细胞和放射抗性EAC细胞(具有增强的CSC特性)表现出特别的疗效。它与5-FU(一种常用的EAC化疗药物)的协同作用表明,CA3 (CIL-56) 可能是一种有前景的YAP1驱动的、治疗耐药性EAC的候选药物[1] 4. 化学结构:CA3 (CIL-56) 的化学结构见文献[1]中的图1E |
| 分子式 |
C23H27N3O5S2
|
|
|---|---|---|
| 分子量 |
489.61
|
|
| 精确质量 |
489.139
|
|
| 元素分析 |
C, 56.42; H, 5.56; N, 8.58; O, 16.34; S, 13.10
|
|
| CAS号 |
300802-28-2
|
|
| 相关CAS号 |
|
|
| PubChem CID |
654092
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| 密度 |
1.5±0.1 g/cm3
|
|
| 沸点 |
741.7±70.0 °C at 760 mmHg
|
|
| 闪点 |
402.4±35.7 °C
|
|
| 蒸汽压 |
0.0±2.6 mmHg at 25°C
|
|
| 折射率 |
1.714
|
|
| LogP |
3.66
|
|
| tPSA |
124
|
|
| 氢键供体(HBD)数目 |
1
|
|
| 氢键受体(HBA)数目 |
8
|
|
| 可旋转键数目(RBC) |
4
|
|
| 重原子数目 |
33
|
|
| 分子复杂度/Complexity |
870
|
|
| 定义原子立体中心数目 |
0
|
|
| SMILES |
S(C1C=CC2=C(/C(/C3C=C(C=CC2=3)S(N2CCCCC2)(=O)=O)=N/O)C=1)(N1CCCCC1)(=O)=O
|
|
| InChi Key |
XYZXEEIUKQGUHB-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C23H27N3O5S2/c27-24-23-21-15-17(32(28,29)25-11-3-1-4-12-25)7-9-19(21)20-10-8-18(16-22(20)23)33(30,31)26-13-5-2-6-14-26/h7-10,15-16,27H,1-6,11-14H2
|
|
| 化学名 |
N-[2,7-bis(piperidin-1-ylsulfonyl)fluoren-9-ylidene]hydroxylamine
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 2.5 mg/mL (5.11 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 悬浮液; 超声和加热处理
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.08 mg/mL (4.25 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.08 mg/mL (4.25 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 配方 4 中的溶解度: 5% DMSO+40% PEG 300+5% Tween 80+50% ddH2O: 0.5mg/ml 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0424 mL | 10.2122 mL | 20.4244 mL | |
| 5 mM | 0.4085 mL | 2.0424 mL | 4.0849 mL | |
| 10 mM | 0.2042 mL | 1.0212 mL | 2.0424 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT04643327 | Recruiting | Drug: Levetiracetam Drug: Placebo |
Parkinson Disease Memory Impairment |
The University of Queensland | February 9, 2021 | Phase 2 |
| NCT03896659 | Recruiting | Drug: Hydrocortisone Oral Drug: Placebo Oral Tablet |
Depression Hydrocortisone |
University of Texas Southwestern Medical Center |
October 1, 2019 | Phase 4 |
| NCT04951700 | Recruiting | Other: Other | Aging Schizophrenia |
University of Texas Southwestern Medical Center |
July 1, 2021 | |
| NCT01522560 | Completed | Other: Feedback group Other: Control group |
Pediatric Anesthesia Department |
The Cleveland Clinic | July 2011 | Not Applicable |
Identification of novel YAP1 inhibitor CA3 and determination of its effects on YAP1 high esophageal adenocarcinoma cells.Mol Cancer Ther.2018 Feb;17(2):443-454. |
|---|
CA3 potently inhibits esophageal adenocarcinoma cell growth and induces tumor cell death.Mol Cancer Ther.2018 Feb;17(2):443-454. |
CA3 inhibits YAP1 expression and transcriptional activity in esophageal adenocarcinoma cell lines, especially those with high YAP1.Mol Cancer Ther.2018 Feb;17(2):443-454. |
CA3 preferentially inhibits CSC properties enriched in radiation-resistant esophageal adenocarcinoma cells.Mol Cancer Ther.2018 Feb;17(2):443-454. |
|---|
CA3 suppresses ALDH1+cell tumor sphere and exerts strong antitumor effects in inducible high YAP xenograft model.Mol Cancer Ther.2018 Feb;17(2):443-454. |
CA3 synergizes with 5-FU in inhibiting growth of esophageal adenocarcinoma cellsin vitroandin vivo.Mol Cancer Ther.2018 Feb;17(2):443-454. |