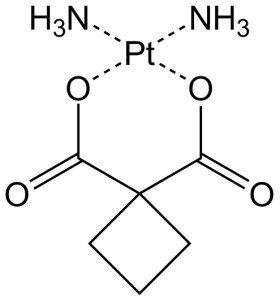
卡铂(以前称为 JM-8、CBDCA、NSC-241240;Paraplat;Paraplatin;Blastocarb;Carboplat;Carbosin;Carbosol;Carbotec;Displata;Ercar)是一种经批准的抗癌药物,通过与DNA(DNA烷基化剂)并干扰癌细胞的细胞修复机制。它用于治疗某些形式的癌症(主要是卵巢癌、肺癌、头颈癌)。它在细胞内被激活,形成反应性铂络合物,与 DNA 中富含 GC 的位点等亲核基团结合,从而诱导链内和链间 DNA 交联,以及 DNA-蛋白质交联。这些卡铂诱导的 DNA 和蛋白质效应导致细胞凋亡和细胞生长抑制。
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
Study of Pembrolizumab/Vibostolimab (MK-7684A) in Combination With Concurrent Chemoradiotherapy Followed by Pembrolizumab/Vibostolimab Versus Concurrent Chemoradiotherapy Followed by Durvalumab in Participants With Stage III Non-small Cell Lung Cancer (MK-7684A-006/KEYVIBE-006)
CTID: NCT05298423
Phase: Phase 3 Status: Recruiting
Date: 2024-12-02
Study of AZD5305 as Monotherapy and in Combination With Anti-cancer Agents in Patients With Advanced Solid Malignancies
CTID: NCT04644068
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2024-12-02
A Study of N9 Chemotherapy in Children With Neuroblastoma
CTID: NCT04947501
PhaseEarly Phase 1 Status: Active, not recruiting
Date: 2024-12-02
Study of Durvalumab Alone or Chemotherapy for Patients With Advanced Non Small-Cell Lung Cancer (PEARL)
CTID: NCT03003962
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-12-02
Study of Pembrolizumab (MK-3475) Combination Therapies in Metastatic Castration-Resistant Prostate Cancer (MK-3475-365/KEYNOTE-365)
CTID: NCT02861573
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2024-12-02
View More
Safety, Effectiveness, and Pharmacokinetics (the Movement of Drug Into, Through and Out of the Body) of BNT327 (an Investigational Therapy) in Combination With Chemotherapy and Other Investigational Agents for Lung Cancer
CTID: NCT06712316
Phase: Phase 2/Phase 3 Status: Not yet recruiting
Date: 2024-12-02
Phase 1b Study of Dato-DXd in Combination With Immunotherapy With or Without Carboplatin in Advanced or Metastatic Non-Small Cell Lung Cancer
CTID: NCT04612751
Phase: Phase 1 Status: Recruiting
Date: 2024-12-02
Neoadjuvant Inhaled Azacytidine With Platinum-Based Chemotherapy and Durvalumab (MEDI4736) - a Combined Epigenetic-Immunotherapy (AZA-AEGEAN) Regimen for Operable Early-Stage Non-Small Cell Lung Cancer (NSCLC)
CTID: NCT06694454
Phase: Phase 1/Phase 2 Status: Not yet recruiting
Date: 2024-12-02
Genetic Testing in Screening Patients With Stage IB-IIIA Non-small Cell Lung Cancer That Has Been or Will Be Removed by Surgery (The ALCHEMIST Screening Trial)
CTID: NCT02194738
Phase: N/A Status: Recruiting
Date: 2024-12-02
Safety and Effectiveness of BNT327, an Investigational Therapy in Combination With Chemotherapy for Patients With Untreated Small-cell Lung Cancer
CTID: NCT06712355
Phase: Phase 3 Status: Not yet recruiting
Date: 2024-12-02
Sacituzumab Tirumotecan (MK-2870) as Monotherapy and in Combination With Pembrolizumab in Participants With Advanced Solid Tumors (MK-2870-008)
CTID: NCT06049212
Phase: Phase 1 Status: Recruiting
Date: 2024-12-02
Phase III, Open-label, First-line Study of Dato-DXd in Combination With Durvalumab and Carboplatin for Advanced NSCLC Without Actionable Genomic Alterations
CTID: NCT05687266
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-12-02
A Study of Pembrolizumab (MK-3475) With or Without V940 in Participants With Non-small Cell Lung Cancer (V940-009/INTerpath-009)
CTID: NCT06623422
Phase: Phase 3 Status: Recruiting
Date: 2024-12-02
Dinutuximab With Chemotherapy, Surgery and Stem Cell Transplantation for the Treatment of Children With Newly Diagnosed High Risk Neuroblastoma
CTID: NCT06172296
Phase: Phase 3 Status: Recruiting
Date: 2024-12-02
A Study to Assess Efficacy and Safety of Pembrolizumab With or Without Sacituzumab Tirumotecan (MK- 2870) in Adult Participants With Resectable Non Small Cell Lung Cancer (NSCLC) Not Achieving Pathological Complete Response (pCR) (MK-2870-019)
CTID: NCT06312137
Phase: Phase 3 Status: Recruiting
Date: 2024-12-02
A Study of MK-1084 in KRAS Mutant Advanced Solid Tumors (MK-1084-001)
CTID: NCT05067283
Phase: Phase 1 Status: Recruiting
Date: 2024-12-02
Sacituzumab Tirumotecan (MK-2870) Versus Pemetrexed and Carboplatin Combination Therapy in Participants With Epidermal Growth Factor (EGFR)-Mutated, Advanced Nonsquamous Non-small Cell Lung Cancer (NSCLC) and Have Progressed on Prior EGFR Tyrosine Kinase Inhibitors (MK-2870-009)
CTID: NCT06305754
Phase: Phase 3 Status: Recruiting
Date: 2024-12-02
TAK-788 as First-Line Treatment Versus Platinum-Based Chemotherapy for Non-Small Cell Lung Cancer (NSCLC) With EGFR Exon 20 Insertion Mutations
CTID: NCT04129502
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-29
A Study Evaluating the Safety, Activity, and Pharmacokinetics of Divarasib in Combination With Other Anti-Cancer Therapies in Participants With Previously Untreated Advanced or Metastatic Non-Small Cell Lung Cancer With a KRAS G12C Mutation
CTID: NCT05789082
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2024-11-29
Testing the Addition of High Dose, Targeted Radiation to the Usual Treatment for Locally-Advanced Inoperable Non-small Cell Lung Cancer
CTID: NCT05624996
Phase: Phase 3 Status: Recruiting
Date: 2024-11-29
Phase I Trial of Bevacizumab and Temsirolimus in Combination With 1) Carboplatin, 2) Paclitaxel, 3) Sorafenib for the Treatment of Advanced Cancer
CTID: NCT01187199
Phase: Phase 1 Status: Active, not recruiting
Date: 2024-11-29
A Study of D3S 001 Monotherapy or Combination Therapy in Subjects With Advanced Solid Tumors With a KRAS p.G12C Mutation
CTID: NCT05410145
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2024-11-29
Carboplatin and Paclitaxel With or Without Ramucirumab in Treating Patients With Locally Advanced, Recurrent, or Metastatic Thymic Cancer That Cannot Be Removed by Surgery
CTID: NCT03694002
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-11-29
Study to Evaluate Adverse Events, Optimal Dose, and Change in Disease Activity, With Livmoniplimab in Combination With Budigalimab Plus Chemotherapy Versus IV Infused Pembrolizumab Plus Chemotherapy in Adult Participants With Untreated Metastatic Non-Squamous Non-Small Cell Lung Cancer (NSCLC)
CTID: NCT06236438
Phase: Phase 2/Phase 3 Status: Recruiting
Date: 2024-11-29
Abiraterone Acetate, Prednisone, and Apalutamide With or Without Ipilimumab or Cabazitaxel and Carboplatin in Treating Patients With Metastatic Castration-Resistant Prostate Cancer
CTID: NCT02703623
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-11-29
Combining Radiation Therapy With Immunotherapy for the Treatment of Metastatic Squamous Cell Carcinoma of the Head and Neck
CTID: NCT05721755
Phase: Phase 3 Status: Recruiting
Date: 2024-11-29
Tiragolumab and Atezolizumab in Patients with Non-squamous Non-small Cell Lung Cancer (NSCLC) and Untreated Brain Metastases
CTID: NCT05746481
Phase: Phase 2 Status: Recruiting
Date: 2024-11-29
Study of Ociperlimab (BGB-A1217) in Combination With Tislelizumab in Advanced Solid Tumors
CTID: NCT04047862
Phase: Phase 1 Status: Completed
Date: 2024-11-29
Debio 0123 in Combination With Carboplatin and Etoposide in Adult Participants With Small Cell Lung Cancer That Recurred or Progressed After Previous Standard Platinum-Based Therapy
CTID: NCT05815160
Phase: Phase 1 Status: Recruiting
Date: 2024-11-29
Pembrolizumab and Concurrent Chemoradiotherapy or Radiation Therapy in Treating Patients With Small Cell Lung Cancer
CTID: NCT02402920
Phase: Phase 1 Status: Active, not recruiting
Date: 2024-11-29
Testing the Addition of Trastuzumab or Trastuzumab/Pertuzumab to the Usual Chemotherapy for HER2 Positive Endometrial Cancer
CTID: NCT05256225
Phase: Phase 2/Phase 3 Status: Recruiting
Date: 2024-11-29
A Study to Learn About the Study Medicine PF-07985045 When Given Alone or With Other Anti-cancer Therapies in People With Advanced Solid Tumors That Have a Change in a Gene
CTID: NCT06704724
Phase: Phase 1 Status: Not yet recruiting
Date: 2024-11-29
A Study to Investigate the Efficacy and Safety of Trastuzumab Deruxtecan as the First Treatment Option for Unresectable, Locally Advanced/Metastatic Non-Small Cell Lung Cancer With HER2 Mutations
CTID: NCT05048797
Phase: Phase 3 Status: Recruiting
Date: 2024-11-29
Study of Oral Debio 0123 in Combination With Carboplatin in Participants With Advanced Solid Tumors
CTID: NCT03968653
Phase: Phase 1 Status: Recruiting
Date: 2024-11-29
A Study of Osimertinib With or Without Chemotherapy Versus Chemotherapy Alone as Neoadjuvant Therapy for Patients With EGFRm Positive Resectable Non-Small Cell Lung Cancer
CTID: NCT04351555
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-27
Pirfenidone Combined With Standard First-Line Chemotherapy in Advanced-Stage Lung NSCLC
CTID: NCT03177291
Phase: Phase 1 Status: Active, not recruiting
Date: 2024-11-27
Safety and Preliminary Effectiveness of BNT327, an Investigational Therapy for Breast Cancer, When Given in Combination with Chemotherapy
CTID: NCT06449222
Phase: Phase 2 Status: Recruiting
Date: 2024-11-27
A Study to Evaluate Dostarlimab Plus Carboplatin-paclitaxel Versus Placebo Plus Carboplatin-paclitaxel in Participants With Recurrent or Primary Advanced Endometrial Cancer
CTID: NCT03981796
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-27
A Trial to Learn if the Combination of Fianlimab, Cemiplimab, and Chemotherapy is Safe and Works Better Than the Combination of Cemiplimab and Chemotherapy in Adult Patients With Non-Small Cell Lung Cancer That Can be Treated With Surgery
CTID: NCT06161441
Phase: Phase 2 Status: Recruiting
Date: 2024-11-27
Study of Pembrolizumab (MK-3475) Plus Chemotherapy vs. Placebo Plus Chemotherapy for Previously Untreated Locally Recurrent Inoperable or Metastatic Triple Negative Breast Cancer (MK-3475-355/KEYNOTE-355)
CTID: NCT02819518
Phase: Phase 3 Status: Completed
Date: 2024-11-27
Study of MK-4830 as Monotherapy and in Combination With Pembrolizumab (MK-3475) in Participants With Advanced Solid Tumors (MK-4830-001)
CTID: NCT03564691
Phase: Phase 1 Status: Active, not recruiting
Date: 2024-11-27
Comparing Proton Therapy to Photon Radiation Therapy for Esophageal Cancer
CTID: NCT03801876
Phase: Phase 3 Status: Recruiting
Date: 2024-11-27
A Study of Subcutaneous (SC) Pembrolizumab Coformulated With Hyaluronidase (MK-3475A) vs Intravenous Pembrolizumab in Adult Participants With Metastatic Non-small Cell Lung Cancer (NSCLC) (MK-3475A-D77)
CTID: NCT05722015
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-27
Study of Tislelizumab in Combination With Chemotherapy Compared to Chemotherapy Alone for Participants With Urothelial Carcinoma
CTID: NCT03967977
Phase: Phase 3 Status: Recruiting
Date: 2024-11-27
Study of IMNN-001 (Also Known as GEN-1) With NACT for Treatment of Ovarian Cancer (OVATION 2)
CTID: NCT03393884
Phase: Phase 1/Phase 2 Status: Active, not recruiting
Date: 2024-11-26
A Study to Investigate the Efficacy and Safety of Dato-DXd With or Without Osimertinib Compared With Platinum Based Doublet Chemotherapy in Participants With EGFR-Mutated Locally Advanced or Metastatic Non-Small Cell Lung Cancer
CTID: NCT06417814
Phase: Phase 3 Status: Recruiting
Date: 2024-11-26
Study of Sacituzumab Govitecan-hziy and Pembrolizumab Versus Treatment of Physician's Choice and Pembrolizumab in Patients With Previously Untreated, Locally Advanced Inoperable or Metastatic Triple-Negative Breast Cancer
CTID: NCT05382286
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-26
A Study to Compare the Efficacy of Nivolumab and Relatlimab Plus Chemotherapy vs Pembrolizumab Plus Chemotherapy for Stage IV/Recurrent Non-squamous Non-small Cell Lung Cancer With PD-L1 1-49%
CTID: NCT06561386
Phase: Phase 3 Status: Recruiting
Date: 2024-11-26
KEYMAKER-U01 Substudy 01A: Efficacy and Safety Study of Pembrolizumab (MK-3475) With or Without Chemotherapy When Used With Investigational Agents in Treatment-naïve Participants With Stage IV Non-small Cell Lung Cancer (NSCLC) (MK-3475-01A/KEYMAKER-U01A)
CTID: NCT04165070
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2024-11-26
Phase II IMNN-001 (Also Known as GEN-1) on SLL With BEV and NACT, Newly Diagnosed Advanced Ovarian, Fallopian Tube or Primary Peritoneal Cancer
CTID: NCT05739981
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2024-11-26
Study of Tislelizumab and Platinum-based Chemotherapy Combination With H1 Receptor Antagonist(Diphenhydramine)in Advanced and Metastatic Non-Small Cell Lung Cancer
CTID: NCT06704620
Phase: Phase 3 Status: Not yet recruiting
Date: 2024-11-26
Testing the Addition of BMS-986016 (Relatlimab) to the Usual Immunotherapy After Initial Treatment for Recurrent or Metastatic Nasopharyngeal Cancer
CTID: NCT06029270
Phase: Phase 2 Status: Recruiting
Date: 2024-11-26
DAREONᵀᴹ-8: A Study to Test How Well Different Doses of BI 764532 in Addition to Standard of Care Are Tolerated by People With Advanced Small Cell Lung Cancer
CTID: NCT06077500
Phase: Phase 1 Status: Recruiting
Date: 2024-11-26
Testing the Use of Investigational Drugs Atezolizumab and/or Bevacizumab With or Without Standard Chemotherapy in the Second-Line Treatment of Advanced-Stage Head and Neck Cancers
CTID: NCT05063552
Phase: Phase 2/Phase 3 Status: Recruiting
Date: 2024-11-26
A Trial With Chemotherapy, Immunotherapy, and Radiotherapy for Patients With Newly Diagnosed Stage IV Small Cell Lung Cancer
CTID: NCT04951115
Phase: Phase 2 Status: Terminated
Date: 2024-11-26
Study of Dato-Dxd as Monotherapy and in Combination With Anti-cancer Agents in Patients With Advanced Solid Tumours (TROPION-PanTumor03)
CTID: NCT05489211
Phase: Phase 2 Status: Recruiting
Date: 2024-11-26
A Study Comparing Two Carboplatin Containing Regimens for Children and Young Adults With Previously Untreated Low Grade Glioma
CTID: NCT02455245
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-26
A Study of ZN-c3 in Patients With Ovarian Cancer
CTID: NCT04516447
Phase: Phase 1 Status: Recruiting
Date: 2024-11-26
Chemotherapy and Pelvic Radiation Therapy With or Without Additional Chemotherapy in Treating Patients With High-Risk Early-Stage Cervical Cancer After Radical Hysterectomy
CTID: NCT00980954
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-26
A Study to Evaluate Immunotherapy Combinations in Participants With Lung Cancer
CTID: NCT03846310
Phase: Phase 1 Status: Active, not recruiting
Date: 2024-11-25
Sonocloud-9 in Association With Carboplatin Versus Standard-of-Care Chemotherapies (CCNU or TMZ) in Recurrent GBM
CTID: NCT05902169
Phase: Phase 3 Status: Recruiting
Date: 2024-11-25
Testing What Happens When an Immunotherapy Drug (Pembrolizumab) is Given by Itself Compared to the Usual Treatment of Chemotherapy With Radiation After Surgery for Recurrent Head and Neck Squamous Cell Carcinoma
CTID: NCT04671667
Phase: Phase 2 Status: Recruiting
Date: 2024-11-25
Nivolumab and Ipilimumab in Treating Patients With Esophageal and Gastroesophageal Junction Adenocarcinoma Undergoing Surgery
CTID: NCT03604991
Phase: Phase 2/Phase 3 Status: Suspended
Date: 2024-11-25
A Trial to Learn How Effective and Safe Odronextamab is Compared to Standard of Care for Adult Participants With Previously Treated Aggressive B-cell Non-Hodgkin Lymphoma
CTID: NCT06230224
Phase: Phase 3 Status: Recruiting
Date: 2024-11-25
Phase Ib Study of the Safety of T-DXd and Immunotherapy Agents With and Without Chemotherapy in Advanced or Metastatic HER2+, Non-squamous NSCLC
CTID: NCT04686305
Phase: Phase 1 Status: Recruiting
Date: 2024-11-25
A Phase III Study of Dato-DXd With or Without Durvalumab Compared With Investigator's Choice of Chemotherapy in Combination With Pembrolizumab in Patients With PD-L1 Positive Locally Recurrent Inoperable or Metastatic Triple-negative Breast Cancer
CTID: NCT06103864
Phase: Phase 3 Status: Recruiting
Date: 2024-11-25
Chemotherapy Combined With Immunotherapy vs Immunotherapy Alone for Older Adults With Stage IIIB-IV Lung Cancer, The ACHIEVE Trial
CTID: NCT06096844
Phase: Phase 3 Status: Recruiting
Date: 2024-11-25
Durvalumab With Chemotherapy as First Line Treatment in Patients With Advanced Biliary Tract Cancers (aBTCs)
CTID: NCT05771480
Phase: Phase 3 Status: Recruiting
Date: 2024-11-25
Organoid-based Functional Precision Therapy for Advanced Breast Cancer
CTID: NCT06102824
Phase: Phase 2 Status: Recruiting
Date: 2024-11-25
Pembrolizumab/Vibostolimab (MK-7684A) or Atezolizumab in Combination With Chemotherapy in First Line Treatment of Extensive-Stage Small Cell Lung Cancer (MK-7684A-008, KEYVIBE-008)
CTID: NCT05224141
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-25
Safety, Preliminary Effectiveness of BNT327, an Investigational Therapy for Patients With Small-cell Lung Cancer in Combination With Chemotherapy
CTID: NCT06449209
Phase: Phase 2 Status: Recruiting
Date: 2024-11-25
A Study Evaluating the Efficacy and Safety of Tislelizumab Versus Chemotherapy in Advanced Non-Squamous Non-small Cell Lung Cancer
CTID: NCT03663205
Phase: Phase 3 Status: Completed
Date: 2024-11-25
Cemiplimab, Low-Dose Paclitaxel and Carboplatin for the Treatment of Recurrent/Metastatic Squamous Cell Carcinoma of the Head and Neck
CTID: NCT04862650
Phase: Phase 2 Status: Recruiting
Date: 2024-11-22
Neoadjuvant and Adjuvant Treatment in Resectable Non-small Cell Lung Cancer
CTID: NCT05061550
Phase: Phase 2 Status: Recruiting
Date: 2024-11-22
A Study of the Drugs Selumetinib Versus Carboplatin/Vincristine in Patients With Neurofibromatosis and Low-Grade Glioma
CTID: NCT03871257
Phase: Phase 3 Status: Recruiting
Date: 2024-11-22
A Study of PT217 in Patients with Neuroendocrine Carcinomas Expressing DLL3 (the SKYBRIDGE Study)
CTID: NCT05652686
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2024-11-22
Veliparib and Carboplatin in Treating Patients With HER2-Negative Metastatic Breast Cancer
CTID: NCT01251874
Phase: Phase 1 Status: Active, not recruiting
Date: 2024-11-22
Sacituzumab Govitecan in Primary HER2-negative Breast Cancer
CTID: NCT04595565
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-22
Study of SAR444881 Administered Alone and in Combination With Other Therapeutics in Participants With Advanced Solid Tumors
CTID: NCT04717375
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2024-11-22
A Study of I-DXd in Combination With Atezolizumab With or Without Carboplatin as First-Line Induction or Maintenance in Subjects With Extensive Stage-Small Cell Lung Cancer (IDeate-Lung03)
CTID: NCT06362252
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2024-11-22
Study of Pemetrexed + Platinum Chemotherapy With or Without Pembrolizumab (MK-3475) in Adults With Tyrosine Kinase Inhibitor- (TKI)-Resistant Epidermal Growth Factor Receptor- (EGFR)-Mutated Metastatic Non-squamous Non-small Cell Lung Cancer (NSCLC) (MK-3475-789/KEYNOTE-789)
CTID: NCT03515837
Phase: Phase 3 Status: Completed
Date: 2024-11-22
A Study of Pembrolizumab (MK-3475) Plus Carboplatin and Paclitaxel as First-line Treatment of Recurrent/Metastatic Head and Neck Squamous Cell Carcinoma (MK-3475-B10/KEYNOTE B10)
CTID: NCT04489888
Phase: Phase 4 Status: Completed
Date: 2024-11-22
Veliparib With or Without Carboplatin in Treating Patients With Stage III or IV Breast Cancer
CTID: NCT01149083
Phase: Phase 2 Status: Completed
Date: 2024-11-22
A Phase 2 Study of EIK1001 in Combo With Pembrolizumab and Chemotherapy in Patients With Stage 4 NSCLC
CTID: NCT06246110
Phase: Phase 2 Status: Recruiting
Date: 2024-11-21
A Study of ASP-1929 Photoimmunotherapy in Combination with Pembrolizumab in First-line Treatment of Locoregional Recurrent Squamous Cell Carcinoma of the Head and Neck with No Distant Metastases
CTID: NCT06699212
Phase: Phase 3 Status: Not yet recruiting
Date: 2024-11-21
Study of Pembrolizumab (MK-3475) Versus Chemotherapy in Mismatch Repair Deficient (dMMR) Advanced or Recurrent Endometrial Carcinoma (MK-3475-C93/KEYNOTE-C93/GOG-3064/ENGOT-en15)
CTID: NCT05173987
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-21
Clinical Trial Evaluating the Safety, Tolerability and Preliminary Efficacy of BNT116 Alone and in Combinations in Patients with Advanced Non-small Cell Lung Cancer
CTID: NCT05142189
Phase: Phase 1 Status: Recruiting
Date: 2024-11-21
Pembrolizumab With Chemotherapy and MK-4830 for Treating Participants With Ovarian Cancer (MK-4830-002)
CTID: NCT05446870
Phase: Phase 2 Status: Completed
Date: 2024-11-21
MEDI5752 in Combination With Carboplatin Plus Pemetrexed in Unresectable Pleural Mesothelioma
CTID: NCT06097728
Phase: Phase 3 Status: Recruiting
Date: 2024-11-21
Evaluating the Addition of the Immunotherapy Drug Atezolizumab to Standard Chemotherapy Treatment for Advanced or Metastatic Neuroendocrine Carcinomas That Originate Outside the Lung
CTID: NCT05058651
Phase: Phase 2/Phase 3 Status: Recruiting
Date: 2024-11-21
Phase I/II Study of PEGylated Arginine Deiminase (ADI-PEG20) With Carboplatin and Cabazitaxel in Men With Aggressive Variant Prostate Cancers (AVPC)
CTID: NCT06085729
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2024-11-20
Adding Certolizumab to Chemotherapy + Nivolumab in People Who Have Lung Cancer That Can Be Treated With Surgery
CTID: NCT04991025
Phase: Phase 2 Status: Recruiting
Date: 2024-11-20
A Study of Neoadjuvant Atezolizumab Plus Chemotherapy Versus Placebo Plus Chemotherapy in Patients With Resectable Stage II, IIIA, or Select IIIB Non-Small Cell Lung Cancer (IMpower030)
CTID: NCT03456063
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-20
Adjuvant Dynamic Marker-Adjusted Personalized Therapy Trial Optimizing Risk Assessment and Therapy Response Prediction in Early Breast Cancer - Triple Negative Breast Cancer
CTID: NCT01815242
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-11-20
Immunotherapy in Combination With Chemoradiation in Patients With Advanced Solid Tumors
CTID: NCT03509012
Phase: Phase 1 Status: Active, not recruiting
Date: 2024-11-20
A Study of Tobemstomig Plus Platinum-Based Chemotherapy vs Pembrolizumab Plus Platinum-Based Chemotherapy in Participants With Previously Untreated Non-Small Cell Lung Cancer
CTID: NCT05775289
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-11-20
N10: A Study of Reduced Chemotherapy and Monoclonal Antibody (mAb)-Based Therapy in Children With Neuroblastoma
CTID: NCT06528496
Phase: Phase 2 Status: Recruiting
Date: 2024-11-20
DAREON™-7: A Study to Test How Well Different Doses of BI 764532 in Addition to Chemotherapy Are Tolerated by People With Advanced Neuroendocrine Cancers
CTID: NCT06132113
Phase: Phase 1 Status: Recruiting
Date: 2024-11-20
Durvalumab With or Without Olaparib as Maintenance Therapy After First-Line Treatment of Advanced and Recurrent Endometrial Cancer
CTID: NCT04269200
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-20
Cabazitaxel, Carboplatin, and Cetrelimab Followed by Niraparib With or Without Cetrelimab for the Treatment of Aggressive Variant Metastatic Prostate Cancer
CTID: NCT04592237
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-11-20
Study of Pembrolizumab With Maintenance Olaparib or Maintenance Pemetrexed in First-line (1L) Metastatic Nonsquamous Non-Small-Cell Lung Cancer (NSCLC) (MK-7339-006, KEYLYNK-006)
CTID: NCT03976323
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-19
Efficacy and Safety of Pembrolizumab Plus Investigational Agents in Combination With Chemotherapy as First-Line Treatment in Extensive-Stage Small Cell Lung Cancer (ES-SCLC) (MK-3475-B99/ KEYNOTE-B99)
CTID: NCT04924101
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-11-19
A Study of Pembrolizumab (MK-3475) With or Without Maintenance Olaparib in First-line Metastatic Squamous Non-small Cell Lung Cancer (NSCLC, MK-7339-008/KEYLYNK-008)
CTID: NCT03976362
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-19
An Investigational Immuno-therapy Trial of Nivolumab, or Nivolumab Plus Ipilimumab, or Nivolumab Plus Platinum-doublet Chemotherapy, Compared to Platinum Doublet Chemotherapy in Patients With Stage IV Non-Small Cell Lung Cancer (NSCLC)
CTID: NCT02477826
Phase: Phase 3 Status: Completed
Date: 2024-11-19
Mirvetuximab Soravtansine (MIRV) With Carboplatin in Second-line Treatment of Folate Receptor Alpha (FRα) Expressing, Platinum-sensitive Epithelial Ovarian Cancer
CTID: NCT05456685
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-11-19
Study of Pembrolizumab (MK-3475) in Combination With Adjuvant Chemotherapy With or Without Radiotherapy in Participants With Newly Diagnosed Endometrial Cancer After Surgery With Curative Intent (MK-3475-B21 / KEYNOTE-B21 / ENGOT-en11 / GOG-3053)
CTID: NCT04634877
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-19
HOT: HIPEC in Ovarian Cancer as Initial Treatment
CTID: NCT02124421
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-11-19
Study of Chemotherapy With Pembrolizumab (MK-3475) Followed by Maintenance With Olaparib (MK-7339) for the First-Line Treatment of Women With BRCA Non-mutated Advanced Epithelial Ovarian Cancer (EOC) (MK-7339-001/KEYLYNK-001/ENGOT-ov43/GOG-3036)
CTID: NCT03740165
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-19
Chemotherapy Followed by Radiation Therapy in Treating Younger Patients With Newly Diagnosed Localized Central Nervous System Germ Cell Tumors
CTID: NCT01602666
Phase: Phase 2 Status: Completed
Date: 2024-11-19
A Trial to Compare the Efficacy, Safety, Pharmacokinetics and Immunogenicity of HD204 to Avastin® in Advanced Non-squamous Non-small Cell Lung Cancer Patients
CTID: NCT03390686
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-19
Cryoablation in Combination (or Not) With Pembrolizumab and Pemetrexed-carboplatin in 1st-line Treatment for Patients With Metastatic Lung Adenocarcinoma
CTID: NCT04339218
Phase: Phase 3 Status: Recruiting
Date: 2024-11-19
Study of Pembrolizumab With Concurrent Chemoradiation Therapy Followed by Pembrolizumab With or Without Olaparib in Stage III Non-Small Cell Lung Cancer (NSCLC) (MK-7339-012/KEYLYNK-012)
CTID: NCT04380636
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-11-19
A Study of the Drugs Selumetinib vs. Carboplatin and Vincristine in Patients With Low-Grade Glioma
CTID: NCT04166409
Phase: Phase 3 Status: Recruiting
Date: 2024-11-19
Pembrolizumab, Carboplatin, and Paclitaxel in Treating Patients With Stage III-IV Ovarian, Primary Peritoneal, or Fallopian Tube Cancer
CTID: NCT02520154
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-11-19
Utility of Adjusting Chemotherapy Dose & Dosing Schedule with the SALVage Weekly Dose-dense Regimen in Patients with Poor Prognostic OVARian Cancers Based on the Tumor Unfavorable Primary Chemosensitivity and Incomplete Debulking Surgery
CTID: NCT06476184
Phase: Phase 3 Status: Recruiting
Date: 2024-11-18
A Global Phase III Study of Rilvegostomig or Pembrolizumab Plus Chemotherapy for First-Line Treatment of Metastatic Squamous Non-small Cell Lung Cancer (NSCLC)
CTID: NCT06692738
Phase: Phase 3 Status: Not yet recruiting
Date: 2024-11-18
A Phase III, Randomised Study of Adjuvant Dato-DXd in Combination With Rilvegostomig or Rilvegostomig Monotherapy Versus Standard of Care, Following Complete Tumour Resection, in Participants With Stage I Adenocarcinoma NSCLC Who Are ctDNA-positive or Have High-risk Pathological Features
CTID: NCT06564844
Phase: Phase 3 Status: Recruiting
Date: 2024-11-18
Study Evaluating Effectiveness and Safety of Zimberelimab and Domvanalimab in Lung Cancer
CTID: NCT04736173
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-11-18
A Study to Evaluate Glofitamab Monotherapy and Glofitamab + Chemoimmunotherapy in Pediatric and Young Adult Participants With Relapsed/Refractory Mature B-Cell Non-Hodgkin Lymphoma
CTID: NCT05533775
Phase: Phase 1/Phase 2 Status: Recruiting