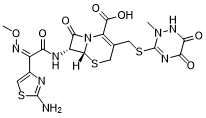
头孢曲松(Ro-139904;Biotrakson;Ro139904)是一种头孢菌素类抗生素,具有广谱抗生素活性,用于治疗各种细菌感染。
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
Short Versus Standard of Care Antibiotic Duration for Children Hospitalized for CAP
CTID: NCT06494072
Phase: Phase 4 Status: Recruiting
Date: 2024-11-20
Antibiotic Instillation in Appendicitis
CTID: NCT05470517
Phase: Phase 2 Status: Completed
Date: 2024-11-06
Early Antibiotics After Aspiration in ICU Patients
CTID: NCT05079620
Phase: Phase 4 Status: Terminated
Date: 2024-11-01
Ceftriaxone Pulse Dose for Post-Treatment Lyme Disease
CTID: NCT06611111
PhaseEarly Phase 1 Status: Not yet recruiting
Date: 2024-10-22
Treatment Responses of Early Syphilis to Ceftriaxone Plus Doxycycline
CTID: NCT05980871
Phase: Phase 4 Status: Active, not recruiting
Date: 2024-09-23
View More
Efficacy of Immunization With 4C-MenB in Preventing Experimental Urethral Infection With Neisseria Gonorrhoeae
CTID: NCT05294588
Phase: Phase 2 Status: Recruiting
Date: 2024-09-19
Trial Comparing Ceftriaxone Plus Azithromycin Versus Ceftriaxone for the Treatment of Gonorrhea
CTID: NCT05027516
Phase: Phase 4 Status: Completed
Date: 2024-08-02
Prophylactic Antibiotic in Subtalar Fusion Surgery
CTID: NCT06527989
Phase: Phase 2/Phase 3 Status: Not yet recruiting
Date: 2024-07-30
Improvement of PPROM Management With Prophylactic Antimicrobial Therapy (iPROMPT)
CTID: NCT06396078
Phase: Phase 4 Status: Recruiting
Date: 2024-07-29
Zoliflodacin in Uncomplicated Gonorrhoea
CTID: NCT03959527
Phase: Phase 3 Status: Completed
Date: 2024-07-26
Tebipenem Trial in Children With Shigellosis
CTID: NCT05121974
Phase: Phase 2 Status: Recruiting
Date: 2024-07-15
Randomized, Embedded, Multifactorial Adaptive Platform Trial for Community- Acquired Pneumonia
CTID: NCT02735707
Phase: Phase 3 Status: Recruiting
Date: 2024-07-12
A Study to Compare Different Antibiotics and Different Modes of Fluid Treatment for Children With Severe Pneumonia
CTID: NCT04041791
Phase: Phase 3 Status: Completed
Date: 2024-07-08
A Study Evaluating Efficacy and Safety of Gepotidacin Compared With Ceftriaxone Plus Azithromycin in the Treatment of Uncomplicated Urogenital Gonorrhea
CTID: NCT04010539
Phase: Phase 3 Status: Completed
Date: 2024-05-30
Antibiotic Treatment for 7 Days Versus 14 Days in Patients With Acute Male Urinary Tract Infection
CTID: NCT02424461
Phase: Phase 3 Status: Completed
Date: 2024-05-01
Screen-and-treat Strategy for Vaginal Flora Abnormalities in Pregnant Women at High Risk of Preterm Birth
CTID: NCT06349122
Phase: Phase 4 Status: Not yet recruiting
Date: 2024-04-05
Potentiated Aminoglycosides in Postoperative Urinary Tract Infection Prophylaxis
CTID: NCT05761405
PhaseEarly Phase 1 Status: Recruiting
Date: 2024-02-08
Mecobalamin Combined With Ceftriaxone Sodium in the Treatment of Sepsis Liver Injury
CTID: NCT06220929
Phase: Phase 4 Status: Not yet recruiting
Date: 2024-01-24
Impact on the Intestinal Microbiota of Treatment With Ceftriaxone in Women's Acute Community Pyelonephritis
CTID: NCT03179384
Phase: Phase 4 Status: Completed
Date: 2023-11-18
Cefixime Plus Doxycycline Compared to Ceftriaxone Plus Azithromycin for Treatment of Gonorrhoea
CTID: NCT06090565
Phase: Phase 4 Status: Completed
Date: 2023-10-23
To Assess the Efficacy and Safety of Ceftriaxone in Patients With Mild to Moderate Parkinson's Disease Dementia
CTID: NCT03413384
Phase: Phase 2 Status: Recruiting
Date: 2023-10-16
PROPER Trial of Pain and Inflammation After Knee Arthroscopy
CTID: NCT05149287
Phase: Phase 2 Status: Terminated
Date: 2023-09-05
Clearance of Asymptomatic Pharyngeal Carriage of Neisseria Gonorrhoeae With or Without Ceftriaxone Treatment: Randomized Non-inferiority Study
CTID: NCT05971550
Phase: Phase 3 Status: Not yet recruiting
Date: 2023-08-02
Piperacillin/Tazobactam Versus ceftriAxone and Metronidazole for Children With Perforated Appendicitis (ALPACA)
CTID: NCT05943223
Phase: Phase 2 Status: Not yet recruiting
Date: 2023-07-13
Cefuroxime vs Ceftriaxone for SSI Prevention in Neurosurgery
CTID: NCT05398081
Phase: Phase 2 Status: Completed
Date: 2023-05-26
Antimicrobial Prophylaxis for Skin Colonization With Propionibacterium Acnes in Primary Open Shoulder Surgery
CTID: NCT02996656
Phase: N/A Status: Recruiting
Date: 2023-05-10
Effect of Intravenous Ceftriaxone and Oral Doxycycline for Lyme Neuroborreliosis
CTID: NCT00138801
Phase: Phase 3 Status: Completed
Date: 2023-05-06
Samu Save Sepsis: Early Goal Directed Therapy in Pre Hospital Care of Patients With Severe Sepsis and/or Septic Shock
CTID: NCT02473263
Phase: Phase 3 Status: Completed
Date: 2023-03-07
Antibiotics to Decrease Post ERCP Cholangitis
CTID: NCT03087656
Phase: Phase 4 Status: Recruiting
Date: 2023-03-06
Prophylactic Antibiotics in Admitted Cirrhotics
CTID: NCT04218695
Phase: Phase 4 Status: Completed
Date: 2023-03-03
Prophylactic Antibiotic Use in Transarterial Chemoembolization for Hepatocellular Carcinoma
CTID: NCT05654896
Phase: Phase 3 Status: Unknown status
Date: 2022-12-16
A Placebo-controlled Efficacy Study of IV Ceftriaxone for Refractory Psychosis
CTID: NCT00591318
Phase: Phase 1/Phase 2 Status: Terminated
Date: 2022-12-02
Comparing the Intravenous Treatment of Skin Infections in Children, Home Versus Hospital
CTID: NCT02334124
Phase: N/A Status: Completed
Date: 2022-11-08
Antimicrobial Treatment in Patients With Ventilator-associated Tracheobronchitis
CTID: NCT03012360
Phase: Phase 4 Status: Unknown status
Date: 2022-08-17
Propranolol on Post Stroke Immune Status and Infection
CTID: NCT05375240
Phase: Phase 2 Status: Unknown status
Date: 2022-05-16
Comparison of Efficacy of Two Combination Regimens for the Neisseria Gonorrhoeae and Chlamydia Coinfection
CTID: NCT05216744
Phase: Phase 2 Status: Completed
Date: 2022-03-17
Experimental Human Infection With Neisseria Gonorrhoeae (LptA Trial)
CTID: NCT04870138
Phase: Phase 1 Status: Completed
Date: 2021-12-02
A Pilot Study Evaluating Penicillin G and Ceftriaxone as Therapies for Presumed Neurosyphilis in HIV Seropositive Individuals
CTID: NCT00000648
Phase: N/A Status: Completed
Date: 2021-10-27
Strategies to Reduce Mortality Among HIV-infected and HIV-exposed Children Admitted With Severe Acute Malnutrition
CTID: NCT05051163
Phase: Phase 2/Phase 3 Status: Unknown status
Date: 2021-09-30
New AntiBiotic Treatment Options for Uncomplicated Anogenital GOnorrhoea
CTID: NCT03294395
Phase: Phase 3 Status: Completed
Date: 2021-08-06
Assessing Parenteral Antibiotics Versus Single-time Intra-operative Intra-dermal Antibiotic Administration for Prevention of SSI
CTID: NCT04975945
Phase: Phase 4 Status: Unknown status
Date: 2021-07-26
Ceftriaxone and Jaundice in Neonates
CTID: NCT03133637
Phase: Status: Completed
Date: 2021-04-06
Experimental Human Infection With Neisseria Gonorrhoeae
CTID: NCT03840811
Phase: Phase 1 Status: Completed
Date: 2021-03-23
Antibiotics as an Adjuvant in Patients With Acute Severe Ulcerative Colitis
CTID: NCT03794765
Phase: Phase 2 Status: Completed
Date: 2020-12-11
Noninferiority Comparison of Prophylactic Open Fracture Antimicrobial Regimens
CTID: NCT03560232
Phase: Phase 4 Status: Terminated
Date: 2020-09-11
First Line Antimicrobials in Children With Complicated Severe Acute Malnutrition
CTID: NCT03174236
Phase: Phase 3 Status: Unknown status
Date: 2020-05-19
Comparison of Efficacy of Cefotaxime, Ceftriaxone, and Ciprofloxacin for the Treatment of SBP in Patients With LC
CTID: NCT01265173
Phase: Phase 4 Status: Completed
Date: 2020-04-06
Ceftriaxone in the Management of Bipolar Depression
CTID: NCT00566111
Phase: N/A Status: Terminated
Date: 2020-03-31
Ceftriaxone as Home IV for Staph Infections
CTID: NCT04141787
Phase: Phase 4 Status: Unknown status
Date: 2019-10-28
A Randomized Trial of Preoperative Prophylactic Antibiotics Prior to Kidney Stone Surgery (Percutaneous Nephrolithotomy [PCNL])
CTID: NCT02384200
Phase: Phase 4 Status: Completed
Date: 2019-08-22
Antibiotic Prophylaxis for Transrectal Prostate Biopsy
CTID: NCT01659866
Phase: Phase 4 Status: Completed
Date: 2019-06-25
Study of Lyme Neuroborreliosis
CTID: NCT01635530
Phase: Phase 4 Status: Completed
Date: 2019-05-13
A Study Evaluating the Effect of Esomeprazole on SYN-004 Degradation of Ceftriaxone In Adults With an Ileostomy
CTID: NCT02473640
Phase: Phase 1/Phase 2 Status: Completed
Date: 2018-11-27
Study on Early Lyme Neuroborreliosis
CTID: NCT00910533
Phase: N/A Status: Unknown status
Date: 2018-10-16
Antibiotics for Klebsiella Liver Abscess Study
CTID: NCT01723150
Phase: Phase 4 Status: Completed
Date: 2018-08-27
The Importance of Anti-anaerobic Therapy for Acute Pelvic Inflammatory Disease (PID)
CTID: NCT01160640
Phase: Phase 2 Status: Completed
Date: 2018-07-10
Comparison of Delafloxacin Versus Ceftriaxone for the Treatment of Uncomplicated Gonorrhea
CTID: NCT02015637
Phase: Phase 3 Status: Terminated
Date: 2018-06-20
Impact of the Choice of 3rd Generation Cephalosporins on the Emergence of Resistance in the Microbiota Intestinal.
CTID: NCT02659033
Phase: Phase 3 Status: Completed
Date: 2018-05-21
Penile Fracture: A Comparison of Erectile Function After Immediate Repair Versus Delayed Repair
CTID: NCT03449940
Phase: N/A Status: Completed
Date: 2018-03-01
RCT to Determine the Efficacy of Combining Hemospray With Medical Treatment in Acute Variceal Bleeding
CTID: NCT03061604
Phase: N/A Status: Completed
Date: 2017-11-14
Efficacy and Safety Study of Oral Solithromycin Compared to Intramuscular Ceftriaxone Plus Oral Azithromycin in the Treatment of Patients With Gonorrhea
CTID: NCT02210325
Phase: Phase 3 Status: Completed
Date: 2017-09-11
A Study to Evaluate the Efficacy and Safety of Intravenous Ceftaroline Versus Intravenous Ceftriaxone in the Treatment of Adult Hospitalised Patients With Community-Acquired Bacterial Pneumonia in Asia
CTID: NCT01371838
Phase: Phase 3 Status: Completed
Date: 2017-09-06
Optimising Antibiotic Treatment for Sick Malnourished Children
CTID: NCT02746276
Phase: Phase 2 Status: Unknown status
Date: 2017-07-02
A Glutamate Transporter GLT1, in the Treatment of Bipolar Disorder
CTID: NCT00512616
Phase: Phase 2 Status: Withdrawn
Date: 2017-07-02
Is Spontaneous Bacterial Peritonitis Still Responding to 3rd Generation Cephalosporins?
CTID: NCT02443285
Phase: Phase 3 Status: Unknown status
Date: 2017-06-20
Prehospital Antibiotics Against Sepsis Trial
CTID: NCT01988428
Phase: N/A Status: Completed
Date: 2017-06-15
Antibiotic Treatment of Multiple Erythema Migrans
CTID: NCT01163994
Phase: N/A Status: Unknown status
Date: 2017-05-05
Crossover Study to Compare the Pharmacokinetics of Subcutaneous and Intravenous Ceftriaxone Administration
CTID: NCT02561442
Phase: Phase 2/Phase 3 Status: Completed
Date: 2017-04-25
Randomized, Open-label Phase 2 Study of Oral AZD0914 in the Treatment of Gonorrhea
CTID: NCT02257918
Phase: Phase 2 Status: Completed
Date: 2017-03-22
Efficacy and Safety of Ertapenem Sodium (MK-0826) Following Colorectal Surgery in Chinese Adults (MK-0826-056)
CTID: NCT01254344
Phase: Phase 3 Status: Completed
Date: 2017-03-22
Comparative Study of Ceftaroline vs. Ceftriaxone in Adults With Community-Acquired Pneumonia
CTID: NCT00509106
Phase: Phase 3 Status: Completed
Date: 2017-03-14
Intravenous Azithromycin Plus Intravenous Ceftriaxone Followed by Oral Azithromycin With Intravenous Levofloxacin Followed by Oral Levofloxacin for the Treatment of Moderate to Severely Ill Hospitalized Patients With Community Acquired Pneumonia
CTID: NCT00035347
Phase: Phase 4 Status: Completed
Date: 2017-03-10
A Study to Assess Beta-Lactam in the Treatment of Hospitalized Patients With Bacterial Pneumonia
CTID: NCT00111644
Phase: Phase 2 Status: Completed
Date: 2016-11-02
Gatifloxacin Versus Ceftriaxone in the Treatment of Enteric Fever
CTID: NCT01421693
Phase: Phase 4 Status: Completed
Date: 2016-10-04
The Op
PNEUMONIA DOSING IN CRITICALLY ILL PATIENTS
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2021-11-25
An open label randomized controlled trial comparing the effect of ceftriaxone plus azithromycin versus ceftriaxone for the treatment of Neisseria gonorrhoeae on the resistome
CTID: null
Phase: Phase 4 Status: Completed
Date: 2021-11-04
Multicenter, randomized, open-label non-inferiority trial, comparing two antibiotic therapy periods (3 versus 7 days) in patients with mild leptospirosis and seen at the hospital in 5 French overseas departments (Martinique, Guadeloupe, French Guiana, Reunion, Mayotte)
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2021-07-28
Short course antibiotic treatment of Gram-negative bacteremia: A multicenter, randomized, non-blinded, non-inferiority interventional study
CTID: null
Phase: Phase 4 Status: Trial now transitioned
Date: 2020-02-11
Short-course antibiotic regimen compared to conventional antibiotic treatment for gram-positive cocci infective endocarditis: randomized clinical trial
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2019-12-10
A Phase III, Randomized, Multicenter, Open-Label Study in Adolescent and Adult Participants Comparing the Efficacy and Safety of Gepotidacin to Ceftriaxone Plus Azithromycin in the Treatment of Uncomplicated Urogenital Gonorrhea Caused by Neisseria gonorrhoeae
CTID: null
Phase: Phase 3 Status: GB - no longer in EU/EEA, Completed
Date: 2019-12-04
Partial oral antibiotic treatment for bacterial brain abscess: An open-label randomised non-inferiority trial (ORAL)
CTID: null
Phase: Phase 4 Status: Trial now transitioned
Date: 2019-10-02
A Phase 2/3 Open-label, Randomized, Active-controlled Clinical Study to Evaluate the Safety, Tolerability, Efficacy and Pharmacokinetics of MK-7655A in Pediatric Participants From Birth to Less Than 18 Years of Age With Confirmed or Suspected Gram-negative Bacterial Infection
CTID: null
Phase: Phase 2, Phase 3 Status: Restarted, Completed
Date: 2019-06-26
Shortened Antibiotic Treatment in Community-Acquired Pneumonia: A Nationwide Danish Randomized Controlled Trial
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2019-04-29
Pharmacokinetics of different antibiotics in cerebrospinal fluid in children with malignant brain tumors – a pilot study
CTID: null
Phase: Phase 1 Status: Ongoing
Date: 2018-09-27
The value of post-operative antibiotic therapy after laparoscopic appendectomy for complicated acute appendicitis (other than for generalized peritonitis): a prospective, randomized, placebo-controlled Phase III study.
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2018-06-05
Shorter treatment of catheter related urinary tract infections
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2018-03-07
Phase II clinical trial to evaluate an antibiotic regimen pharmacokinetic applicable to outpatient parenteral antimicrobial therapy in Enterococcus faecalis infective endocarditis
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2018-02-26
Right Dose, Right Now: Randomized Clinical Trial
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2018-01-11
Investigation of Pharmacokinetic of Antiinfective Therapy in Healthy Subjects and Severely Burned Patients Admitted to the ICU
CTID: null
Phase: Phase 4 Status: Trial now transitioned
Date: 2017-07-26
Non-inferiority multicentre randomized controlled trial comparing short versus standard course postoperative antibiotic treatment for complex acute appendicitis
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2017-03-14
A multicentre, randomized, investigator-blind, active-controlled study to evaluate the safety, tolerability, pharmacokinetics and efficacy of ceftobiprole versus intravenous standard-of-care cephalosporin treatment with or without vancomycin in paediatric patients aged from 3 months to less than 18 years with hospital-acquired pneumonia or community-acquired pneumonia requiring hospitalisation
CTID: null
Phase: Phase 3 Status: Completed
Date: 2016-12-02
Antimicrobial treatment in patients with ventilator-associated tracheobronchitis: a prospective randomized placebo-controlled double-blind multicenter trial
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2016-09-06
PREvention of Complications to Improve Outcome in elderly patients with acute Stroke. A randomised, open, phase III, clinical trial with blinded outcome assessment.
CTID: null
Phase: Phase 3 Status: Ongoing, Completed
Date: 2016-02-03
Randomized, Embedded, Multifactorial, Adaptive Platform trial for Community-Acquired Pneumonia (COVID-19)
CTID: null
Phase: Phase 4 Status: Trial now transitioned, Temporarily Halted, GB - no longer in EU/EEA, Ongoing
Date: 2015-09-16
A Phase 2/3, Randomized, Open-Label, Multi-center
CTID: null
Phase: Phase 2 Status: Temporarily Halted, Completed
Date: 2014-10-10
A randomised controlled trial to compare the clinical effectiveness and safety of gentamicin and ceftriaxone in the treatment of gonorrhoea.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2014-08-05
Essai randomisé contre placebo, multicentrique, de non-infériorité comparant l’efficacité d’un traitement antibiotique court de 14 jours versus 21 jours dans les prostatites aiguës non nosocomiales, à germes sensibles aux fluoroquinolones - PROSTASHORT
CTID: null
Phase: Phase 4 Status: Completed
Date: 2014-03-12
A Multicenter, Randomized, Double-Blinded Comparative Study to Evaluate the Efficacy, Safety, and Pharmacokinetics of Daptomycin Versus Active Comparator in Pediatric Subjects With Acute Hematogenous Osteomyelitis Due to Gram-Positive Organisms
CTID: null
Phase: Phase 3 Status: Completed
Date: 2014-01-02
A Multicenter, Multinational, Randomized, Double-blind Study to Evaluate the Efficacy and Safety of Ceftaroline fosamil Versus Ceftriaxone Plus Vancomycin in Adult Subjects with Community-acquired Bacterial Pneumonia at Risk for Infection Due to Methicillin-resistant Staphylococcus aureus
CTID: null
Phase: Phase 2 Status: Prematurely Ended, Completed
Date: 2012-10-22
Prospective, Randomized, open label, European, multicenter study of the efficacy of the linezolid-rifampin combination versus standard of care in the treatment of Gram-positive prosthetic hip joint infection
CTID: null
Phase: Phase 2 Status: Completed
Date: 2012-10-08
A Multicenter, Randomized, Observer-Blinded, Active-Controlled Study Evaluating the Safety, Tolerability, Pharmacokinetics, and Efficacy of Ceftaroline Versus Ceftriaxone in Pediatric Subjects With Community-acquired Bacterial Pneumonia Requiring Hospitalization
CTID: null
Phase: Phase 2 Status: Completed
Date: 2012-07-18
Neuroborrelioosin epidemiologia, taudinkuva, diagnostiikka ja hoito
CTID: null
Phase: Phase 4 Status: Completed
Date: 2012-06-07
Individualizing duration of antibiotic therapy in hospitalized patients with community-acquired pneumonia: a non-inferiority, randomized, controlled trial.
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2011-12-20
Estudio de la eficacia de la administración prolongada de antibióticos betalactámicos frente a la administración intermitente en el tratamiento de la infección causada por microorganismo sensibles pero con concentraciones mínimas inhibitorias altas
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2011-10-27
Etude multicentrique, de non infériorité, randomisée, ouverte, évaluant l’efficacité de deux Durées d’Antibiothérapie (6 semaines versus 12 semaines) dans le Traitement des Infections sur Prothèses Ostéo-articulaires, avec changement prothétique (en 1 temps ou 2 temps long) ou non (lavage articulaire)
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2011-07-26
A prospective single blind randomised controlled study to compare the outcomes of patients with diabetes and clinically non-infected ischaemic and neuropathic foot ulcers treated with and without oral antibiotics
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2011-07-13
Is a short course of azithromycin effective in the treatment of mild to moderate pelvic inflammatory disease (PID)?
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2011-06-22
Evaluation de l'efficacité et de la tolérance d'un traitement court de 7 jours par ceftriaxone intraveineux le 1er jour puis par cefixime per os du 2éme au 7éme jours dans la prise en charge aux urgences de la pyélonéphrite aigue simple chez la femme de 18 à 65 ans.
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2011-04-18
Preventive Antibiotics in Stroke Study
CTID: null
Phase: Phase 4 Status: Completed
Date: 2010-05-05
PLEASE Persistent Lyme Empiric Antibiotic Study Europe. A prospective, randomised study comparing two prolonged oral antibiotic strategies after initial intravenous ceftriaxone therapy for patients with symptoms of proven or possible persistent Lyme disease
CTID: null
Phase: Phase 4 Status: Completed
Date: 2010-04-29
Intraveneous induction theraphy followed by oral theraphy against exclusive oral theraphy: randomized trial for the treatment of Whipple's disease
CTID: null
Phase: Phase 3 Status: Completed
Date: 2010-01-05
A Phase 3, Multicenter, Randomized, and Double-blind Study to Evaluate the Safety of Tigecycline versus a Ceftriaxone Regimen in the Treatment of Complicated Intra-abdominal Infections and Community-acquired Pneumonia in Pediatric Subjects Ages 8 to 17 Years Old
CTID: null
Phase: Phase 3 Status: Completed
Date: 2009-09-15
treatment of healthcare-associated pneumonia: a prospective, multicenter study
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2009-03-23
Estudio piloto, en fase IV para evaluar la influencia de la asociación de descontaminación intestinal selectiva (DIS) con norfloxacino a antibioterapia estándar sobre la traslocación bacteriana y la actividad inflamatoria en pacientes con peritonitis bacteriana espontánea (PBE)
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2008-07-31
A Phase 3, Multicenter, Randomized, Double-blind, Comparative Study to Evaluate the Safety and Efficacy of Ceftaroline versus Ceftriaxone in the Treatment of Adult Subjects with Community-Acquired Pneumonia
CTID: null
Phase: Phase 3 Status: Completed
Date: 2008-03-25
A national, prospective, randomized, open label study to asses the efficacy and safety of IV/PO moxifloxacin vs IV ceftriaxone + IV azithromicin followed by PO amoxicilline/clavulanate and PO claritromycin in subjects with community-acquired pneumonia
CTID: null
Phase: Phase 3 Status: Prematurely Ended
Date: 2007-12-10
'A Phase 3, Multicenter, Randomized, Double-blind, Comparative Study to Evaluate the Safety and Efficacy of Ceftaroline versus Ceftriaxone, with Adjunctive Clarithromycin, in the Treatment of Adult Subjects with Community-Acquired Pneumonia '
CTID: null
Phase: Phase 3 Status: Completed
Date: 2007-11-22
Efficacy assessment of ceftriaxone therapy in patients with amyotrophic lateral sclerosis.
CTID: null
Phase: Phase 2 Status: Completed
Date: 2007-09-25
Pharmacokinetics of Cetriaxon in bone
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2007-05-22
Randomized, Double-Blind, Multicenter Study of Ceftobiprole Medocaril Versus Ceftriaxone with/without Linezolid in Treatment of Subjects Hospitalized With Community-Acquired Pneumonia
CTID: null
Phase: Phase 3 Status: Completed
Date: 2006-10-10
A Multicenter, Open-Label, Randomized Comparative Study of Tigecycline vs Ceftriaxone Sodium Plus Metronidazole for the Treatment of Hospitalized Subjects With Complicated Intra-abdominal Infection.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2005-12-12
A Phase II, prospective, randomized, double-blind, active-controlled, parallel group, multi-center 'proof of concept' trial in adult patients with community-acquired pneumonia requiring hospitalization without evidence of Legionella
CTID: null
Phase: Phase 2 Status: Completed
Date: 2005-02-18
CEFTRIAXONE NEONATAL THERAPY: A RANDOMIZED CONTROLLED TRIAL TO EVALUATE THE EFFECT OF CEFTRIAXONE ON HYPERBILIRUBINEMIA
CTID: null
Phase: Phase 4 Status: Ongoing
Date:
A MULTI-CENTER, RANDOMIZED, OPEN-LABEL, NON INFERIORITY TRIAL TO EVALUATE THE EFFICACY AND SAFETY OF A SINGLE, ORAL DOSE OF ZOLIFLODACIN COMPARED TO A COMBINATION OF A SINGLE INTRAMUSCULAR DOSE OF CEFTRIAXONE AND A SINGLE ORAL DOSE OF AZITHROMYCIN IN THE TREATMENT OF PATIENTS WITH UNCOMPLICATED GONORRHOEA
CTID: null
Phase: Phase 3 Status: Ongoing
Date: