| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
Vitamin D receptor
|
|---|---|
| 体外研究 (In Vitro) |
在接受肾切除术的小鼠中,多克骨化醇(100 或 300 皮克/克体重)可将血清钙和甲状旁腺激素 (PTH) 水平恢复至正常。在接受肾切除术的小鼠中,多克骨化醇(300 pg/g bw)可显着降低纤维性骨炎。在给予高盐 (HS) 饮食的大鼠中,多克骨化醇显着减少心脏肥大并增强心脏功能。在喂食高盐 (HS) 饮食的大鼠中,多克骨化醇治疗导致组织心房钠尿因子 (ANF) mRNA 水平和血浆脑钠尿肽 (BNP) 水平显着降低。此外,多克骨化醇可显着降低蛋白激酶 C-α (PKCα) 水平,表明维生素 D 缺乏与 PKC 介导的心脏肥大之间可能存在联系。在饮食诱导的肥胖 (DIO) 小鼠中,多克骨化醇可减少蛋白尿、足细胞损伤、系膜生长和细胞外基质蛋白积累。在 DIO 小鼠中,doxercalciferol 还可以减少促纤维化生长因子、促炎细胞因子、氧化应激和巨噬细胞浸润。此外,doxercalciferol 抑制 DIO 小鼠的肾素-血管紧张素-醛固酮系统激活,其中包括血管紧张素 II 1 型受体和盐皮质激素受体。在小鼠中,多克骨化醇和氯沙坦的组合最有效地预防白蛋白尿,恢复肾小球滤过屏障的结构,并以剂量依赖性方式显着降低肾小球硬化。当多克骨化醇和氯沙坦联合使用时,小鼠的糖尿病肾脏几乎没有表现出形态或分子变化。
|
| 体内研究 (In Vivo) |
在 5/6 肾切除 (NX) 大鼠中,第 6 周时,多克骨化醇(0.083、0.167 或 0.333 μg/kg,腹腔注射)可升高血清磷。此外,多克骨化醇(0.167 和 0.333 μg/kg)可增强脉搏波速度 (PWV) 的增加)在第 6 周的 5/6 肾切除 (NX) 大鼠中,并在第 2 周和第 6 周升高血清钙和 Ca × P。多克骨化醇将血清 PTH 降低至 SHAM 水平,并防止 PTH 上升至 0.083 μg/kg[1]。在饲喂高脂肪饮食的 NON 小鼠中,多克骨化醇(125 ng/kg,腹腔注射,每周 3 次)增加 VDR mRNA 水平的表达和 TRPV5 的肾表达。在接受 HF 饮食的小鼠中,多克骨化醇还可以减少蛋白尿,阻止足细胞损失,并减少细胞外基质蛋白的积累。在饲喂 HF 饮食的小鼠中,多克骨化醇可阻断肾素-血管紧张素-醛固酮系统表达的增加,并抑制促纤维化生长因子(TGF-β、PAI-1 和结缔组织生长因子 (CTGF))的表达。此外,Doxercalciferol 还可抑制巨噬细胞的浸润,降低 NF-κb 活性,停止促炎细胞因子的表达,并阻止高脂饮食小鼠肾脂质的积累[2]。当对链脲佐菌素诱导的糖尿病小鼠每周 3 次腹膜内 (ip) 给药时,多克骨化醇 (30 ng/kg) 显着减轻足细胞损失和细胞凋亡,并减少肾小球纤维化[3]。
|
| 动物实验 |
大鼠:采用肾切除术后一周的雄性Sprague-Dawley 5/6肾切除(NX)大鼠(约200 mg)。肾切除术采用典型的两步手术切除方法。为诱导继发性甲状旁腺功能亢进,从肾切除术后两周开始,大鼠饲喂高磷饮食(0.9%磷和0.6%钙)。第0天:假手术组(SHAM)和5/6 NX组大鼠(每组n = 7–10)每周三次腹腔注射赋形剂(5%乙醇/95%丙二醇;0.4 mL/kg)或VDRA(帕立骨化醇或多西骨化醇;0.083、0.167或0.333 μg/kg),持续41天(每组n = 6–10)。选择这些剂量是因为,在该慢性肾脏病模型中,治疗两周或六周后,较低剂量(0.021 和 0.042 μg/kg;腹腔注射)的任一化合物均不能抑制甲状旁腺激素(PTH)水平。在第 0 天至第 41 天(给药后 24 小时)采集血液样本。在第 0 天、第 13 天和第 41 天(给药后 24 小时),对动物进行氯胺酮(50 mg/kg)麻醉,并从尾静脉采集血液样本,用于测定 PTH 和血清生化指标[1]。
|
| 药代性质 (ADME/PK) |
代谢/代谢物
多西骨化醇经胃肠道吸收后,在肝脏中经CYP27活化,生成1α,25-(OH)2D2(主要代谢物)和1α,24-二羟基维生素D2(次要代谢物)。多西骨化醇的活化不需要肾脏参与。 生物半衰期 32至37小时。 |
| 参考文献 |
|
| 其他信息 |
多西骨化醇是一种羟基类固醇,也是一种合成的维生素D2类似物,它在体内代谢活化后会生成1α,25-二羟基维生素D2 (1α,25-(OH)2D2),这是一种天然存在的、具有生物活性的维生素D2形式。它用于治疗继发性甲状旁腺功能亢进症,这是一种慢性肾病患者体内产生过量甲状旁腺激素(PTH;一种控制血液中钙含量的天然物质)的疾病。它具有维生素原、骨密度保护剂和激素原的作用。它是一种维生素D和羟基类固醇。多西骨化醇适用于治疗接受透析的慢性肾脏病患者的继发性甲状旁腺功能亢进症,以及治疗3期或4期慢性肾脏病患者的继发性甲状旁腺功能亢进症。多西骨化醇由健赞公司(Genzyme Corporation)以商品名Hectoral销售,由Catalent Pharma Solutions, Inc.生产。
多西骨化醇是一种维生素D2类似物。 多西骨化醇是一种合成的维生素D类似物,具有潜在的抗肿瘤活性。在肝脏中,多西骨化醇转化为具有生物活性的维生素D代谢物,这些代谢物控制肠道对膳食钙的吸收、肾脏对钙的肾小管重吸收,并与甲状旁腺激素(PTH)共同作用,促进骨骼中钙的动员。这些维生素D代谢物通过与靶组织中的特定受体蛋白相互作用,直接作用于成骨细胞,刺激骨骼生长;同时作用于甲状旁腺,抑制甲状旁腺激素(PTH)的合成和分泌。该药物还被证实能够抑制视网膜母细胞瘤的生长,并可能对前列腺癌细胞具有一定的抗增殖活性。 药物适应症 多西骨化醇适用于治疗接受透析的慢性肾脏病患者的继发性甲状旁腺功能亢进症,以及治疗3期或4期慢性肾脏病患者的继发性甲状旁腺功能亢进症。 FDA标签 作用机制 骨化三醇(1α,25-(OH)2D3)和1α,25-(OH)2D2调节血钙水平,使其维持在维持身体基本功能所需的水平。具体而言,具有生物活性的维生素D代谢物控制着膳食钙的肠道吸收、肾脏对钙的肾小管重吸收,并与甲状旁腺激素(PTH)共同促进骨骼中钙的动员。它们直接作用于骨细胞(成骨细胞)以刺激骨骼生长,并作用于甲状旁腺以抑制甲状旁腺激素(PTH)的合成和分泌。这些功能是通过这些具有生物活性的代谢物与各种靶组织中的特定受体蛋白相互作用而实现的。在慢性肾脏病(CKD)患者中,由于缺乏或25-羟基维生素D-1α-羟化酶活性不足,导致具有生物活性的维生素D代谢物生成不足,进而引起继发性甲状旁腺功能亢进,从而促进代谢性骨病的发生发展。 |
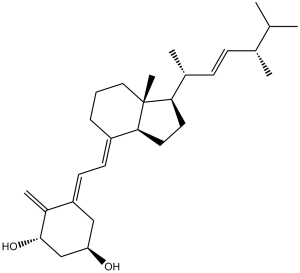
| 分子式 |
C28H44O2
|
|
|---|---|---|
| 分子量 |
412.65
|
|
| 精确质量 |
412.334
|
|
| 元素分析 |
C, 81.50; H, 10.75; O, 7.75
|
|
| CAS号 |
54573-75-0
|
|
| 相关CAS号 |
trans-Doxercalciferol;74007-20-8;Impurity of Doxercalciferol;127516-23-8
|
|
| PubChem CID |
5281107
|
|
| 外观&性状 |
White to yellow/brown solid powder
|
|
| 密度 |
1.0±0.1 g/cm3
|
|
| 沸点 |
538.7±50.0 °C at 760 mmHg
|
|
| 熔点 |
138-140ºC
|
|
| 闪点 |
224.0±24.7 °C
|
|
| 蒸汽压 |
0.0±3.3 mmHg at 25°C
|
|
| 折射率 |
1.541
|
|
| LogP |
8.15
|
|
| tPSA |
40.46
|
|
| 氢键供体(HBD)数目 |
2
|
|
| 氢键受体(HBA)数目 |
2
|
|
| 可旋转键数目(RBC) |
5
|
|
| 重原子数目 |
30
|
|
| 分子复杂度/Complexity |
712
|
|
| 定义原子立体中心数目 |
7
|
|
| SMILES |
C=C1[C@H](C[C@@H](C/C1=C/C=C2[C@]3([C@@](C)([C@H](CC3)[C@@H](/C=C/[C@@H](C(C)C)C)C)CCC/2)[H])O)O
|
|
| InChi Key |
HKXBNHCUPKIYDM-CGMHZMFXSA-N
|
|
| InChi Code |
InChI=1S/C28H44O2/c1-18(2)19(3)9-10-20(4)25-13-14-26-22(8-7-15-28(25,26)6)11-12-23-16-24(29)17-27(30)21(23)5/h9-12,18-20,24-27,29-30H,5,7-8,13-17H2,1-4,6H3/b10-9+,22-11+,23-12-/t19-,20+,24+,25+,26-,27-,28+/m0/s1
|
|
| 化学名 |
(1R,3S,5Z)-5-[(2E)-2-[(1R,3aS,7aR)-1-[(E,2R,5R)-5,6-dimethylhept-3-en-2-yl]-7a-methyl-2,3,3a,5,6,7-hexahydro-1H-inden-4-ylidene]ethylidene]-4-methylidenecyclohexane-1,3-diol
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: (1). 本产品在运输和储存过程中需避光。 (2). 该产品在溶液状态不稳定,请现配现用。 |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (6.06 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (6.06 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL 澄清 DMSO 储备液加入到 900 μL 玉米油中并混合均匀。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4234 mL | 12.1168 mL | 24.2336 mL | |
| 5 mM | 0.4847 mL | 2.4234 mL | 4.8467 mL | |
| 10 mM | 0.2423 mL | 1.2117 mL | 2.4234 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
Link: https://clinicaltrials.gov/ct2/show/NCT02859896
Conditions:Secondary Hyperparathyroidism-Chronic Kidney DiseaseLink: https://clinicaltrials.gov/ct2/show/NCT00502268
Conditions:Coronary Calcification|Endstage Renal Disease|Parathyroid HormoneLink: https://clinicaltrials.gov/ct2/show/NCT00022412
Conditions:Prostate Cancer
Title:Doxercalciferol in Treating Patients With Myelodysplastic Syndrome or Chronic Myelomonocytic Leukemia
Status:Completed
updateDate:2019-11-19
Ctid:NCT00052832
Link: https://clinicaltrials.gov/ct2/show/NCT00052832
Conditions:Leukemia|Myelodysplastic Syndromes|Myelodysplastic/Myeloproliferative DiseasesLink: https://clinicaltrials.gov/ct2/show/NCT00889629
Conditions:Chronic Kidney Disease|Kidney TransplantationLink: https://clinicaltrials.gov/ct2/show/NCT02282813
Conditions:Chronic Kidney Disease|Hyperparathyroidism, Secondary|Vitamin D DeficiencyLink: https://clinicaltrials.gov/ct2/show/NCT00285467
Conditions:Renal OsteodystrophyLink: https://clinicaltrials.gov/ct2/show/NCT00528788
Conditions:Hyperparathyroidism, Secondary|Kidney Failure, ChronicLink: https://clinicaltrials.gov/ct2/show/NCT00463021
Conditions:Secondary HyperparathyroidismLink: https://clinicaltrials.gov/ct2/show/NCT00454350
Conditions:Secondary HyperparathyroidismLink: https://clinicaltrials.gov/ct2/show/NCT00646282
Conditions:Hyperparathyroidism, SecondaryLink: https://clinicaltrials.gov/ct2/show/NCT00792857
Conditions:Chronic Kidney Disease|Secondary Hyperparathyroidism|Chronic Renal Insufficiency|Chronic Renal FailureLink: https://clinicaltrials.gov/ct2/show/NCT00601107
Conditions:Moderate to Severe Chronic Plaque PsoriasisLink: https://clinicaltrials.gov/ct2/show/NCT00749736
Conditions:Chronic Kidney DiseaseLink: https://clinicaltrials.gov/ct2/show/NCT00511017
Conditions:Solid Tumors
|
|
|
|
|