| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|

| 靶点 |
NPC1L1; Nrf2
Ezetimibe (SCH 58235) targets Niemann-Pick C1-Like 1 (NPC1L1) with an IC50 of 0.05 μM (human NPC1L1-mediated cholesterol uptake inhibition) [1] Ezetimibe (SCH 58235) activates nuclear factor erythroid 2-related factor 2 (Nrf2) with an EC50 of 2.3 μM (Nrf2 luciferase reporter activation in HepG2 cells) [1] |
|---|---|
| 体外研究 (In Vitro) |
依折麦布可显着降低总胆固醇、低密度脂蛋白胆固醇和甘油三酯,并小幅但显着地增加高密度脂蛋白胆固醇。依折麦布可减少 Caco-2 细胞中 31% 的胆固醇转运,但不会减少视黄醇的转运。依折麦布导致表面受体 SR-BI、Niemann-Pick 型 C1 Like 1 蛋白 (NPC1L1) 和 ATP 结合盒转运蛋白、亚家族 A (ABCA1) 和核受体视黄酸受体 (ABCA1) 的 mRNA 表达显着降低。通过 Caco-2 细胞中的实时 PCR 分析评估 RAR)gamma、甾醇调节元件结合蛋白 (SREBP)-1 和 -2 以及肝脏 X 受体 (LXR)beta。激酶测定:GST-p62由大肠杆菌制备,0.5μg纯化的GST-p62蛋白用于体外AMPK磷酸化测定。使用 γS-ATP 通过非放射性同位素方法测定 AMPK 对 p62 蛋白的磷酸化。 AMPK 复合物是从 HEK293 细胞中免疫纯化的,用 Flag-AMPKβ1 和 HA-AMPKγ1 转染 myc-AMPKα1 野生型 (WT) 或 myc-AMPKα1 激酶死亡突变体 (KD、D157A)。将 AMPK 复合物添加到含有 20 mM HEPES、pH7.4、1 mM EGTA、0.4 mM EDTA、5 mM MgCl2、0.05 mM DTT、0.5 μg GST-p62、0.2 mM AMP 和 1 mM ATPγS 的反应混合物中。反应在37℃下进行30分钟,然后通过添加20mM EDTA终止。为了检测γS标记的p62蛋白,将反应产物用2.5 mM PNBM在室温下烷基化2小时,并使用抗硫代磷酸盐抗体通过蛋白质印迹分析产物。细胞测定:在与类胡萝卜素 (1 μM) 一起孵育的分化 Caco-2 细胞中,依折麦布 (10 mg/L) 抑制类胡萝卜素转运,对 ɑ-胡萝卜素和 β-胡萝卜素抑制 50%。此外,它还能抑制 β-隐黄质、番茄红素和叶黄素:玉米黄质 (1:1) 的转运。同时,依折麦布抑制胆固醇转运31%。依折麦布降低表面受体 SR-BI、ATP 结合盒转运蛋白、A 亚家族 (ABCA1)、Niemann-Pick C1 型样 1 蛋白 (NPC1L1) 和视黄酸受体 (RAR)γ、甾醇调节元件结合蛋白 SREBP 的表达-1 和 SREBP-2,以及肝 X 受体 (LXR)β。
在棕榈酸(200 μM)诱导脂毒性的人肝癌HepG2细胞中,Ezetimibe (SCH 58235)(1–10 μM)以剂量依赖方式激活Nrf2信号通路,上调Nrf2靶基因的mRNA表达:HO-1(10 μM时3.8倍)、NQO1(10 μM时3.2倍)、GCLC(10 μM时2.5倍)。10 μM时减少细胞内活性氧(ROS)水平45%、脂质过氧化40%,同时抑制肝细胞凋亡(Annexin V⁺细胞从32%降至12%)[1] 在高糖(30 mM)+棕榈酸(150 μM)处理的原代小鼠肝细胞中,Ezetimibe (SCH 58235)(5 μM)增强自噬流,表现为LC3-II/LC3-I比值增加2.8倍、p62蛋白水平降低60%;减少细胞内甘油三酯(TG)积累55%、胆固醇含量48% [2] 在Caco-2细胞中,Ezetimibe (SCH 58235)(0.01–1 μM)抑制NPC1L1介导的[³H]-胆固醇摄取,IC50=0.05 μM,在肠上皮细胞水平阻断胆固醇吸收 [1] |
| 体内研究 (In Vivo) |
在西方、低脂和无胆固醇饮食小鼠中,依泽替米贝分别将血浆胆固醇水平从 964 mg/dL 降低至 374 mg/dL、从 726 mg/dL 降低至 231 mg/dL 和从 516 mg/dL 降低至 178 mg/dL。依折麦布将西方饮食组小鼠的主动脉粥样硬化病变表面积从 20.2% 减少至 4.1%,将低脂胆固醇饮食组小鼠的主动脉粥样硬化病变表面积从 24.1% 减少至 7.0%。在西方和低脂胆固醇组中,依折麦布使颈动脉粥样硬化病变横截面积减少了 97%,在无胆固醇小鼠中减少了 91%。在 apoE-/- 小鼠中,依泽替米贝可抑制胆固醇吸收,降低血浆胆固醇,增加高密度脂蛋白水平,并抑制西方、低脂和无胆固醇饮食条件下动脉粥样硬化的进展。依折麦布可有效抑制胆固醇穿过肠壁的转运,从而降低高胆固醇血症临床前动物模型中的血浆胆固醇。在大鼠中建立了依泽替米贝消除肠道的外分泌胰腺功能,同时维持胆汁流动。依折麦布可降低胆固醇喂养仓鼠的血浆胆固醇和肝脏胆固醇积累,ED(50) 为 0.04 mg/kg。
C57BL/6小鼠喂食高脂高胆固醇(HFHC)饮食16周诱导非酒精性脂肪性肝炎(NASH),口服 Ezetimibe (SCH 58235)(10 mg/kg/天,持续8周),减轻肝脂肪变性(TG含量降低60%)、小叶炎症(炎症细胞浸润评分从3.2降至1.5)、肝细胞气球样变(评分从2.8降至1.2);激活肝脏Nrf2信号(HO-1和NQO1蛋白水平分别增加3.5倍和3.0倍),减少肝脏ROS生成50% [1] 在肥胖糖尿病 Zucker 糖尿病肥胖(ZDF)大鼠中,口服 Ezetimibe (SCH 58235)(5 mg/kg/天,持续12周)改善肝脂肪变性:肝脏TG和胆固醇水平分别降低55%和45%;增强肝脏自噬(LC3-II/LC3-I比值增加2.5倍),血清谷丙转氨酶(ALT)和谷草转氨酶(AST)水平分别降低40%和35% [2] HFHC饮食喂养的小鼠中,Ezetimibe (SCH 58235)(10 mg/kg/天)降低血清总胆固醇(TC)35%、低密度脂蛋白胆固醇(LDL-C)40%、甘油三酯(TG)30%,不影响高密度脂蛋白胆固醇(HDL-C)水平 [1] |
| 酶活实验 |
使用大肠杆菌生产 GST-p62,并将 0.5 μg 纯化蛋白用于体外 AMPK 磷酸化测定。使用 S-ATP 的非放射性同位素方法用于测定 AMPK 对 p62 蛋白的磷酸化。从 HEK293 细胞中免疫纯化 AMPK 复合物,然后将 Flag-AMPKβ1 和 HA-AMPKγ1 转染至 myc-AMPKα1 野生型 (WT) 或 myc-AMPKα1 激酶死亡突变体 (KD、D157A) 细胞中。反应混合物含有 20 mM HEPES、pH 7.4、1 mM EGTA、0.4 mM EDTA、5 mM MgCl2、0.05 mM DTT、0.5 μg GST-p62、0.2 mM AMP 和 1 mM ATPS。然后将 AMPK 复合物添加到混合物中。反应在 37°C 下进行 30 分钟,然后添加 20 mM EDTA 结束反应。反应产物在室温下用 2.5 mM PNBM 烷基化 2 小时,以检测用 S 原子进行 γS 标记的 p62 蛋白 [1],然后使用抗硫代磷酸盐抗体进行蛋白质印迹分析。
NPC1L1介导的胆固醇摄取实验:Caco-2细胞培养于含10%胎牛血清的DMEM培养基,接种到24孔板(2×10⁵个细胞/孔),分化21天。血清饥饿2小时后,用系列浓度的 Ezetimibe (SCH 58235)(0.01–1 μM)处理30分钟,加入[³H]-胆固醇(1 μCi/孔),37°C孵育1小时。冰浴PBS终止反应,洗涤细胞2次,裂解液裂解后闪烁计数检测放射性,计算胆固醇摄取抑制率和IC50 [1] Nrf2荧光素酶报告基因实验:HepG2细胞共转染Nrf2响应性荧光素酶报告质粒和海肾荧光素酶质粒(内参)。24小时后,用系列浓度的 Ezetimibe (SCH 58235)(0.1–10 μM)处理18小时。裂解细胞后双荧光素酶试剂盒检测荧光素酶活性,计算相对荧光素酶活性(萤火虫/海肾),确定Nrf2激活的EC50 [1] |
| 细胞实验 |
Huh7 人肝细胞在 37°C、95% 空气/5% CO2 环境中使用含有 10% FBS、100 单位/mL 青霉素和 100 μg/mL 链霉素的高葡萄糖 DMEM 培养。依泽替米贝(10 μM,1 小时)和棕榈酸(0.5 mM,24 小时)在治疗或不治疗的情况下给予肝细胞[2]。
肝细胞脂毒性与Nrf2激活实验:HepG2细胞(5×10⁴个细胞/孔)接种到24孔板,孵育过夜后,用棕榈酸(200 μM)+ Ezetimibe (SCH 58235)(1–10 μM)处理24小时。DCFH-DA染色检测细胞内ROS,丙二醛(MDA)法检测脂质过氧化,Annexin V-FITC/PI染色检测凋亡。提取RNA和蛋白,qPCR和Western blot检测Nrf2靶基因(HO-1、NQO1、GCLC)[1] 肝细胞自噬与脂质积累实验:分离原代小鼠肝细胞,接种到6孔板(1×10⁶个细胞/孔),用高糖(30 mM)+棕榈酸(150 μM)+ Ezetimibe (SCH 58235)(5 μM)处理24小时。Western blot分析自噬标志物(LC3、p62),比色法试剂盒量化细胞内甘油三酯和胆固醇水平 [2] |
| 动物实验 |
小鼠:我们使用10周龄的雄性C57BL/6J小鼠。随机选取三组小鼠:普通饲料组、MCD饲料加载体对照组和MCD饲料加依折麦布治疗组,每组7-10只。饲养环境温度保持在23±2℃,湿度保持在60%±10%,光照周期为12小时光照/12小时黑暗。MCD饲料组小鼠每日灌胃给予依折麦布10 mg/kg,持续4周。普通饲料组和MCD饲料加载体对照组小鼠每日灌胃给予等量的磷酸盐缓冲液,持续4周。治疗期间,每周测量一次小鼠体重。4周后,对小鼠进行镇静并处死,通过心脏穿刺取血。组织采集后,一部分用福尔马林固定,然后包埋于石蜡中;另一部分则立即用液氮冷冻,并保存在-70°C。
大鼠:实验在特定无病原体(SPF)级动物房内进行,光照/黑暗周期为12小时。实验采用雄性OLETF大鼠(n=11)和年龄匹配的LETO大鼠(n=3)。OLETF大鼠是一种晚发性高血糖症模型,具有慢性病程、轻度肥胖和糖尿病临床发作的特征。动物可自由摄取食物和水。大鼠在12周龄时随机分组,分别灌胃给予PBS或依泽替米贝(10 mg/kg/天),持续20周。实验期间大鼠禁食,之后腹腔注射佐来替尔/罗普宁(Zoletil/Rompun)使其安乐死。解剖肝脏后,立即将肝脏组织置于液氮中冷冻,然后储存于-80°C,待从腹主动脉抽取血液后进行后续分析。 NASH小鼠模型:6周龄C57BL/6小鼠(每组n=8)喂食高脂高胆固醇(HFHC)饮食16周以诱导NASH。然后,将依泽替米贝(SCH 58235)悬浮于0.5%羧甲基纤维素溶液中,以10 mg/kg/天的剂量通过灌胃给药,持续8周。对照组给予溶剂。治疗结束后,采集血液样本,检测血清脂质谱(TC、LDL-C、HDL-C、TG)和肝功能指标(ALT、AST)。采集肝组织,检测肝脏TG、胆固醇、ROS和Nrf2靶蛋白水平;对脂肪变性、炎症和气球样变进行组织病理学分析[1] 肥胖糖尿病大鼠模型:8周龄ZDF大鼠(每组n=7)饲喂标准饲料。依泽替米贝(SCH 58235)悬浮于0.5%羧甲基纤维素溶液中,以5 mg/kg/天的剂量灌胃给药,持续12周。对照组给予溶剂。采集血液样本,测定血清ALT、AST和血脂水平。取肝组织,定量分析TG和胆固醇含量,并通过Western blot分析自噬标志物(LC3、p62)[2] |
| 药代性质 (ADME/PK) |
吸收、分布和排泄
空腹成年人单次服用10 mg依泽替米贝后,血浆峰浓度 (Cmax) 为3.4-5.5 ng/mL,达峰时间为4-12小时 (Tmax)。主要药理活性代谢物依泽替米贝葡糖醛酸苷的Cmax为45-71 ng/mL,Tmax为1-2小时。食物摄入对依泽替米贝的吸收影响甚微,但与高脂餐同服可使Cmax增加38%。由于依泽替米贝不溶于适用于静脉注射的水性介质,因此无法确定其真实生物利用度。 口服放射性标记的依泽替米贝后,约78%和11%分别从粪便和尿液中排出。粪便中主要成分为未代谢的原药,约占给药剂量的 69%;尿液中主要成分为依泽替米贝-葡萄糖醛酸苷,约占给药剂量的 9%。粪便中原药回收率高提示胆汁中分泌的依泽替米贝-葡萄糖醛酸苷吸收率低和/或水解率低。 依泽替米贝的相对分布容积为 107.5 升。 目前尚无依泽替米贝清除率的药代动力学数据。 依泽替米贝是首个新型选择性胆固醇吸收抑制剂。该药物及其活性葡萄糖醛酸苷代谢物通过抑制一种尚未鉴定的膜转运蛋白,阻碍肠道对膳食和肝脏排泄的胆汁胆固醇的重吸收。口服依泽替米贝后,其吸收迅速,且不受食物成分的影响。该药物不经细胞色素P450系统代谢,而是在肠道内进行广泛的葡萄糖醛酸化。因此,依泽替米贝的血浆浓度约占血浆中总依泽替米贝浓度的10%。依泽替米贝及其葡萄糖醛酸化物在肠道内的循环显著延长了这些化合物在其作用部位的停留时间。老年患者和肾功能不全患者的依泽替米贝葡萄糖醛酸化物清除似乎受损,血浆浓度升高1.5至2倍。迄今为止,尚无药物相互作用研究发现依泽替米贝或合用药物的药代动力学发生显著变化。依泽替米贝通过抑制肠道对胆固醇的吸收来降低血浆胆固醇水平。由于依泽替米贝存在广泛的肠肝循环,因此只需相对较低的剂量即可有效。在血液和胆汁中,依泽替米贝主要以葡萄糖醛酸苷结合物的形式存在,该结合物在肠细胞中形成。目前,尚不清楚哪些机制负责这种高效的肠肝循环。Abcc2、Abcc3 和 Abcg2 是 ABC 转运蛋白,它们在肝脏和肠道中均有表达,能够转运葡萄糖醛酸化化合物。本研究旨在探讨这些转运蛋白在依泽替米贝葡萄糖醛酸苷 (Ez-gluc) 肠肝循环中的作用。我们利用表达 ABCC2、ABCC3 和 ABCG2 的 Sf21 昆虫细胞的质膜囊泡进行了转运研究。此外,我们还利用野生型和 Abcc3-/- 小鼠的肠道外植体,在 Ussing 室装置中研究了定向转运。最后,在野生型、Abcc3-/-、Abcc2-/-、Abcg2-/- 和 Abcg2-/-/Abcc2-/- 小鼠中,通过十二指肠给药依泽替米贝,测定了 Ez-gluc 的胆汁排泄情况。Ez-gluc 呈剂量依赖性地抑制了 ABCC3、ABCC2 和 ABCG2 介导的转运。在 Ussing 室中,与野生型小鼠相比,Abcc3-/- 小鼠十二指肠(2.2%)、空肠(23%)和回肠(23%)组织中从基底外侧回收的 Ez-gluc 显著减少。与野生型小鼠相比,Abcc3-/- 小鼠(34%)、Abcc2-/- 小鼠(56%)和 Abcg2-/-/Abcc2-/- 小鼠(2.5%)的胆汁中 Ez-gluc 的排泄量也显著减少。这些数据表明,依泽替米贝-葡糖苷酸的肠肝循环强烈依赖于Abcc3、Abcc2和Abcg2的联合功能。 目前尚不清楚依泽替米贝是否会分泌到人乳中。在大鼠研究中,哺乳幼鼠体内依泽替米贝的总暴露量可达母体血浆中观察到的浓度的一半。 口服后,依泽替米贝被吸收并广泛与一种具有药理活性的酚类葡糖苷酸(依泽替米贝-葡糖苷酸)结合。空腹成年人单次服用10 mg依泽替米贝后,依泽替米贝的平均血浆峰浓度(Cmax)在4至12小时内达到3.4至5.5 ng/mL(Tmax)。依泽替米贝-葡糖苷酸的平均Cmax值在1至2小时内达到45至71 ng/mL(Tmax)。在 5 至 20 mg 剂量范围内,未观察到剂量比例的显著偏差。由于依泽替米贝几乎不溶于适用于注射的水性介质,因此无法确定其绝对生物利用度。 代谢/代谢物 在人体内,依泽替米贝通过小肠和肝脏中的 II 期葡萄糖醛酸结合反应迅速且广泛地代谢,形成其主要酚类代谢物——依泽替米贝葡萄糖醛酸苷。体外研究表明,负责依泽替米贝葡萄糖醛酸化的主要人肝脏和/或肠道尿苷 5′-二磷酸 (UDP)-葡萄糖醛酸转移酶 (UGT) 为 UGT1A1、1A3 和 2B15。依泽替米贝的氧化反应(I期)可生成SCH 57871,人空肠微粒体中也检测到痕量的苄基葡糖醛酸苷(SCH 488128)。依泽替米贝葡糖醛酸苷占血浆中总循环化合物的80-90%,并保留一定的药理活性,可抑制肠道胆固醇的吸收。在人体内,依泽替米贝及其葡糖醛酸苷约占血浆中总药物的93%。血浆浓度-时间曲线呈现多个峰值,提示存在肠肝循环,约20%的药物因肠肝循环而被重吸收。依泽替米贝主要在小肠和肝脏中通过葡糖醛酸结合(II期反应)代谢,随后经胆汁和肾脏排泄。在所有评估物种中均观察到极少的氧化代谢(I期反应)。在人体内,依泽替米贝迅速代谢为依泽替米贝-葡萄糖醛酸苷。依泽替米贝和依泽替米贝-葡萄糖醛酸苷是血浆中检测到的主要药物衍生物,分别约占血浆中总药物的10%至20%和80%至90%。依泽替米贝和依泽替米贝-葡萄糖醛酸苷均以约22小时的半衰期从血浆中消除。血浆浓度-时间曲线呈现多个峰值,提示存在肠肝循环。口服(14)C-依泽替米贝(20 mg)后,血浆中总依泽替米贝(依泽替米贝+依泽替米贝-葡萄糖醛酸苷)约占总放射性的93%。 48小时后,血浆中未检测到放射性物质。在10天的收集期内,分别约有78%和11%的给药放射性物质从粪便和尿液中回收。依泽替米贝是粪便中的主要成分,占给药剂量的69%;依泽替米贝-葡萄糖醛酸苷是尿液中的主要成分,占给药剂量的9%。 依泽替米贝已知的代谢产物包括依泽替米贝-葡萄糖醛酸苷。 生物半衰期 依泽替米贝和依泽替米贝-葡萄糖醛酸苷的半衰期均约为22小时。 依泽替米贝和依泽替米贝-葡萄糖醛酸苷均以约22小时的半衰期从血浆中消除。 |
| 毒性/毒理 (Toxicokinetics/TK) |
肝毒性
单独使用依折麦布或与其他降脂药联合使用时,血清酶升高发生率较低(0.5%至1.5%),但大多数升高是自限性的,且不伴有黄疸或其他症状。在大型随机对照试验中,依折麦布单药治疗并未导致血清ALT升高发生率高于安慰剂组。然而,依折麦布与他汀类药物联合使用时,血清转氨酶升高或因肝功能异常而停药的可能性略有增加。已有依折麦布引起临床上明显的急性肝损伤的报道,但这种情况较为罕见。此外,由于该药常与其他降胆固醇药物联合使用,因此在这些报道中依折麦布的作用并不总是明确。依折麦布引起临床上明显的肝损伤的潜伏期为2至10个月,血清酶升高模式从肝细胞型到胆汁淤积型不等。服用依折麦布和他汀类药物联合用药的患者中曾报道出现自身免疫性肝炎样损伤,但依折麦布在这些反应中的作用难以确定(病例1)。曾报道过一例因依折麦布引起的胆管消失综合征病例,该患者尽管出现黄疸,仍继续服用依折麦布数月。 可能性评分:C(可能是临床上明显的肝损伤的罕见病因)。 妊娠和哺乳期影响 ◉ 哺乳期用药概述 来自两位母亲的数据表明,依折麦布及其活性代谢物在乳汁中的含量极低,且根据药代动力学模型预测,婴儿血清中的药物浓度远低于成人。依折麦布在哺乳期似乎是可以接受的。哺乳期妇女应避免将依折麦布与他汀类药物(例如阿托伐他汀、辛伐他汀)联合使用。 ◉ 对母乳喂养婴儿的影响 截至修订日期,未找到相关的已发表信息。 ◉ 对泌乳和母乳的影响 截至修订日期,未找到相关的已发表信息。 蛋白结合 依折麦布和依折麦布-葡糖苷酸与人血浆蛋白的结合率均>90%。依折麦布的平均体外蛋白结合率范围为99.5%至99.8%,依折麦布-葡糖苷酸的平均体外蛋白结合率范围为87.8%至92.0%。 相互作用 根据一项小型研究,依折麦布与华法林发生药代动力学或药效学相互作用的可能性不大。上市后监测数据显示,依折麦布与华法林合用时,国际标准化比值(INR)可能升高;然而,大多数患者同时还服用其他药物。若患者正在服用华法林,则应监测依折麦布的INR。 可能存在药代动力学相互作用(依折麦布血浆峰浓度和AUC升高,环孢素AUC升高)。严重肾功能不全患者的依折麦布暴露量可能更高。依折麦布和辛伐他汀固定复方制剂(尤其是高剂量时)与环孢素合用会增加肌病/横纹肌溶解的风险。由于依折麦布和环孢素的暴露量增加,合用时应谨慎,并监测环孢素浓度。如果同时使用,固定复方制剂中依折麦布和辛伐他汀的剂量每日不应超过10 mg。 可能存在药代动力学(依折麦布AUC降低)和药效学(降低低密度脂蛋白胆固醇的作用)相互作用。依折麦布应在服用胆汁酸螯合剂前至少2小时或后至少4小时服用。 与非诺贝特或吉非贝齐同时使用时,观察到药代动力学相互作用(血浆依折麦布浓度升高)。贝特类药物可能增加胆固醇排泄到胆汁中,导致胆结石,并且已有研究表明依折麦布可增加动物胆囊胆汁中的胆固醇含量。临床研究显示,接受依折麦布联合非诺贝特治疗的患者中,1.7%接受了胆囊切除术;而接受非诺贝特单药治疗的患者中,这一比例为0.6%。目前,在积累更多人体数据之前,不建议将依折麦布与非诺贝特以外的其他贝特类药物联合使用。如果怀疑接受依折麦布联合非诺贝特治疗的患者患有胆结石,应进行胆囊检查,并考虑其他降脂治疗方案。 有关依折麦布的更多相互作用(完整)数据(共 6 项),请访问 HSDB 记录页面。 在 C57BL/6 小鼠中进行的 8 周毒性研究(10 mg/kg/天口服)显示,依折麦布 (SCH 58235) 未引起体重(变化 <5%)、肝功能(ALT、AST)或肾功能(肌酐、BUN)的显著变化[1]。 在 ZDF 大鼠中进行的 12 周研究(5 mg/kg/天口服)显示,肝脏、肾脏、脾脏或肠道均未观察到明显的组织病理学异常。血清电解质和血液学参数均在正常范围内[2] 依泽替米贝(SCH 58235)在人体内的血浆蛋白结合率为90-95%[1] |
| 参考文献 |
|
| 其他信息 |
治疗用途
依泽替米贝可单独使用或与其他降脂药(例如羟甲基戊二酰辅酶A [HMG-CoA] 还原酶抑制剂(他汀类药物)、非诺贝特)联合使用,作为饮食疗法的辅助药物,用于治疗原发性高胆固醇血症和混合型血脂异常、纯合子家族性高胆固醇血症和/或纯合子家族性植物甾醇血症。/美国产品标签包含/ 依泽替米贝可单独使用或与他汀类药物联合使用,作为饮食疗法的辅助药物,用于降低原发性(杂合子家族性和非家族性)高胆固醇血症中升高的血清总胆固醇、低密度脂蛋白胆固醇 (LDL-C) 和载脂蛋白 B (apo B) 浓度。依泽替米贝与辛伐他汀固定复方制剂可作为饮食疗法的辅助药物,用于治疗原发性高胆固醇血症或混合型血脂异常,以降低升高的血清总胆固醇、低密度脂蛋白胆固醇、载脂蛋白B、甘油三酯和非高密度脂蛋白胆固醇水平,并升高高密度脂蛋白胆固醇水平。依泽替米贝也可与非诺贝特联合使用,作为饮食疗法的辅助药物,用于治疗混合型血脂异常,以降低升高的血清总胆固醇、低密度脂蛋白胆固醇、载脂蛋白B和非高密度脂蛋白胆固醇水平。/美国产品标签/ 依泽替米贝可作为饮食疗法的辅助药物,用于降低纯合子家族性谷甾醇血症患者升高的血清谷甾醇和菜油甾醇水平。 /美国产品标签包含/ 依折麦布可与阿托伐他汀或辛伐他汀联合使用,以降低纯合子家族性高胆固醇血症患者升高的血清总胆固醇和低密度脂蛋白胆固醇浓度,作为其他降脂疗法(例如血浆低密度脂蛋白分离术)的辅助治疗,或在其他降脂疗法不可用时使用。/美国产品标签包含/ 这是一项回顾性研究,纳入了所有接受依折麦布单药治疗高胆固醇血症且有随访临床和血脂结果的儿科患者。在确定的36例患者中,26例的脂蛋白谱提示家族性高胆固醇血症(FH),10例的脂蛋白谱提示家族性混合型高脂血症(FCHL)。经过平均105天(范围32-175天)的依折麦布治疗后,FH患者的总胆固醇(TC)水平从7.3±1.0 mmol/L降至5.7±1.0 mmol/L(P<0.0001),低密度脂蛋白胆固醇(LDL-C)水平从5.3±0.9 mmol/L降至3.9±0.8 mmol/L(P<0.0001)。在FCHL患者中,TC水平从6.4±2.0 mmol/L降至5.6±0.4 mmol/L(P≤0.002),LDL-C水平从4.7±1.0 mmol/L降至3.8±0.6 mmol/L(P≤0.005)。所有患者的平均低密度脂蛋白胆固醇(LDL-C)值下降了1.5 ± 0.9 mmol/L,降幅为28%。依折麦布治疗后,甘油三酯和高密度脂蛋白胆固醇水平无显著变化。患者接受依折麦布治疗长达3.5年,未出现与药物相关的不良反应。开始使用依折麦布治疗后平均13.6个月(范围1-44个月),LDL-C水平仍维持在4.0 ± 0.6 mmol/L。在这项针对高胆固醇血症儿童和青少年的小型回顾性研究中,依折麦布在降低低密度脂蛋白胆固醇(LDL-C)水平方面安全有效。 药物警告 依折麦布与羟甲基戊二酰辅酶A(HMG-CoA)还原酶抑制剂(他汀类药物)联用时,禁用于活动性肝病患者或血清转氨酶(转氨酶)浓度不明原因的持续升高患者。 在Zetia对照临床试验数据库(安慰剂对照)中,纳入了2396例患者,中位治疗持续时间为12周(范围0至39周)。Zetia组和安慰剂组分别有3.3%和2.9%的患者因不良反应而停药。导致Zetia治疗中断且发生率高于安慰剂组的最常见不良反应为:关节痛(0.3%);头晕(0.2%);γ-谷氨酰转移酶升高(0.2%)。在包含 2396 例患者的 Zetia 单药治疗对照临床试验数据库中,最常报告的不良反应(发生率≥2% 且高于安慰剂组)为:上呼吸道感染(4.3%)、腹泻(4.1%)、关节痛(3.0%)、鼻窦炎(2.8%)和四肢疼痛(2.7%)。 在包含 11308 例患者的 Zetia + 他汀类药物对照临床试验数据库中,中位治疗持续时间为 8 周(范围 0 至 112 周),Zetia + 他汀类药物组有 4.0% 的患者和单用他汀类药物组有 3.3% 的患者因不良反应而停药。在接受Zetia联合他汀类药物治疗的患者组中,导致治疗中断且发生率高于单用他汀类药物的最常见不良反应包括:丙氨酸氨基转移酶升高(0.6%)、肌痛(0.5%)、疲乏、天冬氨酸氨基转移酶升高、头痛和四肢疼痛(均为0.2%)。在包含11308例患者的Zetia联合他汀类药物对照临床试验数据库中,最常报告的不良反应(发生率≥2%且高于单用他汀类药物)包括:鼻咽炎(3.7%)、肌痛(3.2%)、上呼吸道感染(2.9%)、关节痛(2.6%)和腹泻(2.5%)。 Zetia上市后监测经验显示,曾有肌病和横纹肌溶解症的病例报告。大多数发生横纹肌溶解症的患者在开始服用Zetia之前已服用过他汀类药物。然而,已有报道显示,单独使用依替拉韦治疗以及将依替拉韦与已知会增加横纹肌溶解风险的药物(例如贝特类药物)联用时,均可能发生横纹肌溶解。如果确诊或怀疑患者患有肌病,应立即停用依替拉韦以及患者同时服用的任何他汀类药物或贝特类药物。出现肌肉症状且肌酸磷酸激酶 (CPK) 水平超过正常值上限 (ULN) 10 倍以上提示肌病。 有关依泽替米贝(共 15 条)的更多药物警告(完整)数据,请访问 HSDB 记录页面。 药效学 依泽替米贝已被证实可降低高脂血症患者的总胆固醇 (total-C)、低密度脂蛋白胆固醇 (LDL-C)、载脂蛋白 B (Apo B)、非高密度脂蛋白胆固醇 (non-HDL-C) 和甘油三酯 (TG) 水平,并升高高密度脂蛋白胆固醇 (HDL-C) 水平。与单独使用依泽替米贝或他汀类药物或非诺贝特相比,依泽替米贝与这两种药物联合使用时,其治疗效果更为显著。在针对纯合子和杂合子家族性高胆固醇血症患者以及植物甾醇血症患者的临床试验中,推荐治疗剂量的依折麦布可有效降低低密度脂蛋白胆固醇(LDL-C)水平15-20%,同时使高密度脂蛋白胆固醇(HDL-C)水平升高2.5-5%。中重度肝功能损害导致依折麦布暴露量增加的影响尚未评估——符合这些标准的患者应避免使用依折麦布。上市后报告显示,服用依折麦布的患者可能出现肌病和横纹肌溶解症,且在同时接受或近期接受过他汀类药物治疗的患者中,这种风险似乎会加剧。 依折麦布(SCH 58235)是一种选择性NPC1L1抑制剂,可阻断肠道胆固醇吸收和肝脏胆固醇重吸收[1]。 其对非酒精性脂肪性肝炎(NASH)和肝脂肪变性的保护作用涉及两个关键机制:抑制NPC1L1以减少脂质积累,以及激活Nrf2以增强抗氧化防御并减少氧化应激[1]。 它通过上调自噬通量来调节肝脏自噬,促进脂滴降解,并减少肥胖/糖尿病模型中的细胞内脂质积累[2]。 临床上,依折麦布适用于治疗高胆固醇血症,可单独使用或与他汀类药物联合使用,以降低血清胆固醇水平。低密度脂蛋白胆固醇 (LDL-C) 水平 [1] 临床前研究支持其作为非酒精性脂肪性肝病 (NAFLD) 和非酒精性脂肪性肝炎 (NASH) 治疗药物的潜力 [1][2] |
| 分子式 |
C24H21F2NO3
|
|---|---|
| 分子量 |
409.4
|
| 精确质量 |
409.148
|
| 元素分析 |
C, 70.41; H, 5.17; F, 9.28; N, 3.42; O, 11.72
|
| CAS号 |
163222-33-1
|
| 相关CAS号 |
Ezetimibe;163222-33-1
|
| PubChem CID |
150311
|
| 外观&性状 |
White to off-white solid powder
|
| 密度 |
1.3±0.1 g/cm3
|
| 沸点 |
654.9±55.0 °C at 760 mmHg
|
| 熔点 |
164-166℃
|
| 闪点 |
349.9±31.5 °C
|
| 蒸汽压 |
0.0±2.1 mmHg at 25°C
|
| 折射率 |
1.624
|
| LogP |
3.26
|
| tPSA |
60.77
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
5
|
| 可旋转键数目(RBC) |
6
|
| 重原子数目 |
30
|
| 分子复杂度/Complexity |
567
|
| 定义原子立体中心数目 |
3
|
| SMILES |
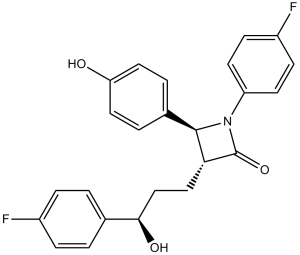
FC1C([H])=C([H])C(=C([H])C=1[H])N1C([C@]([H])(C([H])([H])C([H])([H])[C@@]([H])(C2C([H])=C([H])C(=C([H])C=2[H])F)O[H])[C@@]1([H])C1C([H])=C([H])C(=C([H])C=1[H])O[H])=O
|
| InChi Key |
OLNTVTPDXPETLC-XPWALMASSA-N
|
| InChi Code |
InChI=1S/C24H21F2NO3/c25-17-5-1-15(2-6-17)22(29)14-13-21-23(16-3-11-20(28)12-4-16)27(24(21)30)19-9-7-18(26)8-10-19/h1-12,21-23,28-29H,13-14H2/t21-,22+,23-/m1/s1
|
| 化学名 |
(3R,4S)-1-(4-fluorophenyl)-3-[(3S)-3-(4-fluorophenyl)-3-hydroxypropyl]-4-(4-hydroxyphenyl)azetidin-2-one
|
| 别名 |
SCH-58235; SCH 58235; SCH-58235; SCH58235; trade names: Zetia, Ezetrol
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|---|
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4426 mL | 12.2130 mL | 24.4260 mL | |
| 5 mM | 0.4885 mL | 2.4426 mL | 4.8852 mL | |
| 10 mM | 0.2443 mL | 1.2213 mL | 2.4426 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
CHOlesterol Lowering and Residual Risk in Type 2 Diabetes
CTID: NCT04369664
Phase: Phase 4 Status: Completed
Date: 2024-10-17
|
|
|
|