| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| Other Sizes |
|
| 靶点 |
GSK-3
The target of GSK-3 inhibitor 1 is glycogen synthase kinase 3 (GSK-3), including two isoforms: GSK-3α and GSK-3β. For human GSK-3α, the half-maximal inhibitory concentration (IC₅₀) is 0.08 μM [2] ; for human GSK-3β, the IC₅₀ is 0.06 μM [2] . It shows no significant inhibition of other related kinases (e.g., CDK2, ERK1, JNK2) with IC₅₀ > 10 μM, demonstrating high isoform and kinase selectivity [2] |
|---|---|
| 体外研究 (In Vitro) |
GSK-3 抑制剂 1 可用于诱导、促进或增强内耳组织(如内耳支持细胞或内耳毛细胞)的生长、增殖或再生[2]。
1. GSK-3激酶活性抑制:GSK-3抑制剂1以浓度依赖的方式抑制重组人类GSK-3α和GSK-3β的催化活性。0.1 μM浓度下,较溶媒对照组抑制GSK-3α活性85%,抑制GSK-3β活性90% [2] 2. 诱导内耳支持细胞增殖:体外培养分离的小鼠耳蜗支持细胞,用0.1、0.5、1.0 μM的GSK-3抑制剂1处理7天。0.5 μM浓度使支持细胞增殖率较溶媒组增加2.3倍(BrdU掺入实验);1.0 μM浓度增加3.1倍,且未检测到细胞毒性 [1] 3. 诱导支持细胞向内耳毛细胞分化:用0.5 μM的GSK-3抑制剂1处理小鼠耳蜗支持细胞14天,可诱导其转分化为毛细胞样细胞,免疫荧光染色显示45%的细胞表达毛细胞特异性标志物(Myo7a、钙视网膜蛋白),而溶媒对照组仅3% [1] 4. 促进干细胞/祖细胞自我更新:用0.1–0.5 μM的GSK-3抑制剂1处理人类牙髓干细胞(hDPSCs)5天,自我更新能力增强,克隆形成效率提高1.8–2.5倍(克隆形成实验)。流式细胞术分析证实,干细胞标志物(CD105、CD90)阳性比例保持>95%,表明干细胞表型得以保留 [2] 5. 激活Wnt/β-连环蛋白信号通路:在支持细胞和hDPSCs中,0.5 μM的GSK-3抑制剂1可增加β-连环蛋白的核积累(免疫荧光检测),并使Wnt靶基因(c-Myc、Cyclin D1)的表达上调2.8–3.5倍(实时荧光定量PCR) [1,2] |
| 体内研究 (In Vivo) |
1. 噪声诱导听力损失(NIHL)小鼠的毛细胞再生:将C57BL/6小鼠暴露于110 dB SPL宽带噪声中2小时,诱导耳蜗毛细胞损失。噪声暴露后第3天开始,将GSK-3抑制剂1(溶于10%二甲基亚砜+90%无菌生理盐水)通过耳蜗内注射给药,剂量为0.5 μM(每耳1 μL),每周1次,持续3周。治疗后4周,组织学分析显示,耳蜗基底回外毛细胞数量较溶媒处理的NIHL小鼠增加60%;听性脑干反应(ABR)检测显示,8 kHz频率下听力阈值降低25 dB,表明功能恢复 [1]
2. 小鼠牙髓干细胞增殖:向C57BL/6小鼠牙髓局部注射0.1 μM的GSK-3抑制剂1(5 μL),7天后BrdU标记显示,增殖的牙髓干细胞数量较溶媒对照组增加2.2倍。免疫组织化学染色证实,增殖细胞表达干细胞标志物(CD105),且保留分化潜能 [2] |
| 酶活实验 |
1. 重组GSK-3α/β激酶活性实验:
- 将重组人类GSK-3α或GSK-3β蛋白溶于激酶实验缓冲液(20 mM Tris-HCl,pH 7.5,10 mM MgCl₂,1 mM DTT),终浓度为10 ng/μL [2] - 向激酶溶液中加入系列浓度的GSK-3抑制剂1(0.001–10 μM)或溶媒,随后加入荧光标记的GSK-3特异性肽底物和ATP(终浓度100 μM) [2] - 反应混合物在30°C孵育60分钟,加入含EDTA的终止缓冲液终止反应 [2] - 用酶标仪检测荧光强度(激发光485 nm,发射光520 nm),反映底物的磷酸化水平。计算相对于溶媒对照组的激酶活性抑制百分比,从剂量-反应曲线推导IC₅₀值 [2] |
| 细胞实验 |
1. 耳蜗支持细胞分离及增殖实验:
- 从P3-P5 C57BL/6小鼠幼崽中分离耳蜗,无菌条件下取出柯蒂氏器,通过酶消化和机械吹打分散支持细胞,以5×10³个/孔接种于胶原包被的96孔板 [1] - 细胞在含生长因子的DMEM/F12培养基中培养,用0.1–1.0 μM的GSK-3抑制剂1或溶媒处理,7天培养期的最后24小时向培养基中加入BrdU [1] - 多聚甲醛固定细胞,曲拉通X-100透化后,用抗BrdU抗体进行免疫染色,荧光显微镜下计数BrdU阳性细胞数,计算相对于溶媒对照组的增殖率 [1] 2. 毛细胞转分化实验: - 分离的小鼠耳蜗支持细胞接种于含盖玻片的24孔板,用0.5 μM的GSK-3抑制剂1处理14天,每3天更换一次培养基 [1] - 固定细胞后,用抗Myo7a(毛细胞标志物)和Sox2(支持细胞标志物)一抗进行免疫染色,再加入荧光二抗 [1] - 用图像分析软件量化Myo7a阳性细胞比例,共聚焦显微镜下观察转分化细胞的形态 [1] 3. 干细胞自我更新(克隆形成)实验: - 分离人类牙髓干细胞(hDPSCs),以100个/孔接种于6孔板,用0.1–0.5 μM的GSK-3抑制剂1或溶媒处理 [2] - 培养14天后,甲醇固定克隆,结晶紫染色,计数含>50个细胞的克隆,克隆形成效率计算为(克隆数/接种细胞数)×100% [2] 4. Wnt通路激活实验(实时荧光定量PCR): - 支持细胞或hDPSCs用0.5 μM的GSK-3抑制剂1处理24小时后,提取总RNA [1,2] - 以1 μg总RNA合成互补DNA(cDNA),用c-Myc、Cyclin D1和内参基因GAPDH的特异性引物进行实时荧光定量PCR [1,2] - 采用2^(-ΔΔCt)法计算相对基因表达水平,比较GSK-3抑制剂1处理组与溶媒对照组的差异 [1,2] |
| 动物实验 |
1. 用于毛细胞再生的噪声性听力损失 (NIHL) 小鼠模型:
- 将 8-10 周龄的雄性 C57BL/6 小鼠随机分为三组(每组 n=8):正常对照组(未暴露于噪声)、载体处理的 NIHL 组和GSK-3 抑制剂 1 处理的 NIHL 组 [1] - 将小鼠置于隔音室中,暴露于 110 dB SPL 宽带噪声 2 小时,诱导 NIHL。噪声暴露后 3 天测量 ABR 阈值以确认听力损失 [1] - GSK-3 抑制剂 1 配制成 0.5 μM 的溶液,溶于 10% DMSO + 90% 无菌生理盐水中。在麻醉状态下,使用微量注射器进行耳蜗内注射(每个耳蜗 1 μL)。从噪声暴露后第3天开始,每周注射一次,持续3周[1] - 治疗后4周,重复进行ABR测试以评估听力功能。将小鼠安乐死,解剖耳蜗,固定并用鬼笔环肽(标记毛细胞立体纤毛)染色,用于毛细胞数量的组织学分析[1] 2. 小鼠牙髓干细胞增殖模型: - 将雌性C57BL/6小鼠(6-8周龄)随机分为载体组和GSK-3抑制剂1组(每组n=6)[2] - GSK-3抑制剂1配制成0.1 μM的无菌磷酸盐缓冲液(PBS)溶液。在麻醉状态下,于上颌第一磨牙钻一个小孔,并使用微量注射器将5 μL的测试溶液或载体(PBS)注入牙髓[2]。 在安乐死前24小时(注射后7天),腹腔注射BrdU(50 mg/kg)给小鼠。解剖牙髓组织,固定,石蜡包埋,并进行切片。进行BrdU和CD105的免疫组织化学染色以鉴定增殖干细胞[2]。 |
| 毒性/毒理 (Toxicokinetics/TK) |
1. 体外细胞毒性:用浓度高达 5 μM 的 GSK-3 抑制剂 1 处理耳蜗支持细胞和人牙髓干细胞 (hDPSC) 14 天,未发现细胞活力受到影响(MTT 法),与溶剂对照组相比,细胞活力 >90% [1,2]
2. 体内局部毒性:在噪声性听力损失 (NIHL) 小鼠模型中,耳蜗内注射 GSK-3 抑制剂 1 (0.5 μM) 未引起炎症(苏木精-伊红染色)或邻近耳蜗结构(例如螺旋神经节神经元)的损伤 [1] |
| 参考文献 | |
| 其他信息 |
1. GSK-3 抑制剂 1 是一种小分子糖原合成酶激酶 3 (GSK-3α/β) 抑制剂,已开发用于再生医学领域,包括内耳毛细胞再生(用于治疗听力损失)和干细胞自我更新调节 [1,2]
2. 作用机制:GSK-3 抑制剂 1 选择性地与 GSK-3α/β 的 ATP 结合口袋结合,抑制其激酶活性。这导致β-catenin的稳定和核转位,激活经典的Wnt/β-catenin信号通路,从而促进支持细胞/干细胞的增殖并诱导耳蜗支持细胞向毛细胞的转分化[1,2] 3. 制剂特性:所述专利公开了GSK-3抑制剂1的增溶组合物,包括以DMSO、聚乙二醇或环糊精为增溶剂的水性制剂,旨在提高局部给药(耳蜗内、牙髓注射)的溶解度和生物利用度[1,2] 4. 治疗潜力:它具有治疗感音神经性听力损失(由毛细胞损伤/丢失引起)以及需要增强干细胞自我更新的再生疗法(例如,牙髓再生、组织修复)的潜在用途[1,2] 5. 化学类别:GSK-3抑制剂化合物 1属于 1H-吡咯-2,5-二酮类化合物,其化学结构经过优化,具有高 GSK-3 选择性和低脱靶激酶抑制作用[2] |
| 分子式 |
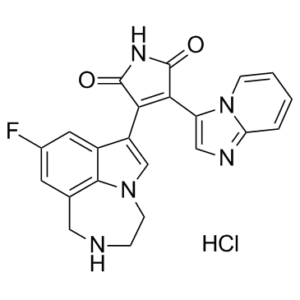
C22H17CLFN5O2
|
|---|---|
| 分子量 |
437.8541
|
| 精确质量 |
337.10223
|
| CAS号 |
603272-51-1
|
| 相关CAS号 |
603272-51-1
|
| PubChem CID |
78357782
|
| 外观&性状 |
Yellow to orange solid
|
| LogP |
8.592
|
| tPSA |
80.12
|
| 氢键供体(HBD)数目 |
3
|
| 氢键受体(HBA)数目 |
5
|
| 可旋转键数目(RBC) |
2
|
| 重原子数目 |
31
|
| 分子复杂度/Complexity |
788
|
| 定义原子立体中心数目 |
0
|
| SMILES |
Cl[H].FC1=C([H])C2C([H])([H])N([H])C([H])([H])C([H])([H])N3C([H])=C(C4C(N([H])C(C=4C4=C([H])N=C5C([H])=C([H])C([H])=C([H])N45)=O)=O)C(=C1[H])C3=2
|
| InChi Key |
CXXAOCQHGIGIBJ-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C22H16FN5O2.ClH/c23-13-7-12-9-24-4-6-27-11-15(14(8-13)20(12)27)18-19(22(30)26-21(18)29)16-10-25-17-3-1-2-5-28(16)17;/h1-3,5,7-8,10-11,24H,4,6,9H2,(H,26,29,30);1H
|
| 化学名 |
3-(6-fluoro-1,10-diazatricyclo[6.4.1.04,13]trideca-2,4,6,8(13)-tetraen-3-yl)-4-imidazo[1,2-a]pyridin-3-ylpyrrole-2,5-dione;hydrochloride
|
| 别名 |
GSK-3 inhibitor 1
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 请将本产品存放在密封且受保护的环境中,避免吸湿/受潮。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO: ~12.5 mg/mL (~28.55 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 1.25 mg/mL (2.85 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 12.5 mg/mL澄清的DMSO储备液加入到400 μL PEG300中,混匀;再向上述溶液中加入50 μL Tween-80,混匀;然后加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 1.25 mg/mL (2.85 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 12.5 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2839 mL | 11.4194 mL | 22.8389 mL | |
| 5 mM | 0.4568 mL | 2.2839 mL | 4.5678 mL | |
| 10 mM | 0.2284 mL | 1.1419 mL | 2.2839 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Status | Interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT04239092 | Active Recruiting |
Drug: 9-ING-41 Drug: Irinotecan |
Refractory Cancer Cancer Pediatric |
Actuate Therapeutics Inc. | June 5, 2020 | Phase 1 |
| NCT05239182 | Active Recruiting |
Drug: 9-ING-41 Drug: Retifanlimab |
Pancreatic Adenocarcinoma | Anwaar Saeed | January 26, 2022 | Phase 2 |
| NCT03678883 | Recruiting | Drug: 9-ING-41 Drug: Doxorubicin |
Cancer Sarcoma |
Actuate Therapeutics Inc. | January 4, 2019 | Phase 2 |
| NCT05010629 | Recruiting | Drug: 9-ING-41 Drug: Carboplatin |
Metastatic Cancer Salivary Gland Cancer |
Glenn J. Hanna | September 14, 2021 | Phase 2 |
| NCT05077800 | Recruiting | Drug: 9-ING-41 Drug: Losartan |
Pancreatic Adenocarcinoma Pancreatic Adenocarcinoma Metastatic |
Colin D. Weekes, M.D. | March 21, 2022 | Phase 2 |