| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| Other Sizes |
|
| 靶点 |
NAD+ ; NAD(P)H:quinone oxidoreductase 1 (NQO1); KL1333 modulates NAD+ metabolism, with effects on enzymes involved in NAD+ synthesis and mitochondrial function (specific targets not explicitly defined with IC50/Ki values) [1] [2] [3]
|
|---|---|
| 体外研究 (In Vitro) |
KL1333是NAD细胞内的强大调节剂,NAD是细胞能量觉醒所必需的辅酶。 PGC-1α 由 SIRT1 和 AMPK 激活,而 SIRT1 和 AMPK 则由 NAD+ 水平升高激活[1]。
线粒体脑肌病、乳酸酸中毒和中风样发作(MELAS)是最常见的母系遗传线粒体疾病之一,是由导致线粒体功能障碍的线粒体DNA突变引起的。存在几种治疗选择,包括补充辅酶Q10、维生素和营养素,但目前还没有经过验证的有效治疗方法。在这项研究中,我们研究了一种新型NAD+调节剂KL1333对来自MELAS患者的人成纤维细胞的影响。KL1333是一种口服的小有机分子,与NAD(P)H:醌氧化还原酶1(NQO1)作为底物反应,通过NADH氧化导致细胞内NAD+水平升高。为了阐明KL1333的作用机制,我们使用了C2C12成肌细胞、L6成肌细胞和MELAS成纤维细胞。KL1333诱导的NAD+水平升高触发了SIRT1和AMPK的激活,随后激活了这些细胞中的PGC-1α。在MELAS成纤维细胞中,KL1333增加了ATP水平,降低了乳酸和ROS水平,这些水平在这种疾病中往往失调。此外,线粒体功能分析表明,KL1333增加了线粒体质量、膜电位和氧化能力。这些结果表明,KL1333改善了线粒体的生物合成和功能,因此是治疗MELAS的有前景的治疗剂。[1] 顺铂(CP)是一种用于治疗癌性实体瘤的化疗药物,但它会引起严重的副作用,包括耳毒性。CP诱导耳毒性的主要原因是线粒体活性氧(ROS)水平升高。在这项研究中,我们使用耳蜗的离体器官型培养系统研究了β-拉帕酮衍生物2-异丙基-3H-萘并(1,2-d)咪唑-4,5-二酮(KL1333)对CP诱导的耳毒性的影响。通过免疫组织化学比较CP处理的耳蜗外植体在有或没有KL1333的情况下的毛细胞损伤。通过测量细胞内ROS水平和线粒体膜电位去极化,分析CP诱导的氧化应激和KL1333的预防作用。通过TUNEL法和切割的半胱氨酸天冬氨酸蛋白酶-3的免疫染色检测凋亡信号通路的激活。结果发现,KL1333预处理显著减少了静纤毛变性和毛细胞损失,并防止了CP引起的线粒体ROS水平的增加。耳蜗外植体的免疫组织化学检查显示,CP组的caspase-3免疫阳性率高于对照组,而KL1333+CP组的免疫阳性率明显低于CP组(P<0.05)。因此,KL1333似乎通过减少线粒体ROS产生引起的线粒体损伤来保护Corti器官中的毛细胞免受CP诱导的凋亡。本研究首次报道了KL1333对CP诱导的耳毒性的预防作用。尽管应进行进一步的研究以确定KL1333是否可以维持CP的抗癌作用,但我们的数据谨慎地表明,抗氧化剂KL1333可以用作有效的抗凋亡剂,以预防CP诱导的氧化应激引起的耳毒性,并可能被证明可用于预防CP引起的听力损失[2]。 在MELAS成纤维细胞中,用KL1333(1 μM,处理24-72小时)可增加细胞内NAD+水平,改善线粒体呼吸(通过氧消耗率测量),并减少活性氧(ROS)产生。它还能提高ATP水平,纠正MELAS细胞中观察到的异常线粒体膜电位。蛋白质印迹分析显示线粒体生物合成标志物(如PGC-1α)上调,电子传递链组件的表达改善 [1] 在暴露于顺铂(10 μM)的小鼠耳蜗培养物中,KL1333(0.1-1 μM)预处理可减少顺铂诱导的毛细胞损失(1 μM时减少30-50%),并保护听觉神经元活力。它能抑制顺铂介导的凋亡通路激活,表现为切割的caspase-3水平降低,耳蜗细胞中ROS积累减少 [2] |
| 体内研究 (In Vivo) |
在过去的二十年里,人们对罕见病孤儿药物开发的兴趣越来越大。然而,这些疾病的临床试验设计仍然存在障碍。这项1a/1b期研究解决了几个挑战,同时评估了新型口服分子KL1333在健康志愿者和原发性线粒体疾病受试者中的安全性和耐受性。KL1333旨在使对ATP产生至关重要的NAD+:NADH比率正常化。该试验纳入了创新的设计元素,这些元素具有潜在的可移植性,可用于其他罕见疾病,包括患者参与、适应性设计和探索性目标,所有这些元素随后都为正在进行的KL1333第2阶段关键疗效研究的方案提供了信息。结果表明,KL1333是安全且耐受性良好的,具有剂量依赖性胃肠道副作用,并验证了原发性线粒体疾病的潜在新结局指标,包括30秒坐立和患者报告的疲劳量表。重要的是,KL1333的试验数据支持了基于疲劳、功能强度和耐力改善的疗效。此外,该研究强调了使用1期研究来获取有助于优化后期疗效试验设计的数据的价值[3]。
在一项针对成人线粒体疾病患者的1a/1b期临床试验中,单次和多次口服KL1333(50-400 mg/天,持续14天)以剂量依赖性方式增加全血NAD+水平(400 mg/天时最高达2.5倍)。患者在次要结局方面有改善,包括疲劳评分降低,线粒体功能标志物(如血浆酰基肉碱谱)稳定或改善 [3] |
| 酶活实验 |
NQO1氧化测定[1]
用rhNQO1进行NADH氧化测定。NQO1蛋白(2.5 mU)与不同浓度(0.1、0.25、0.5、1、2.5、5、10、25、50和100μM)的KL1333、CoQ10或艾地苯醌在含有0.14%BSA的50 mM Tris-HCl(pH 7.5)缓冲液中混合。通过加入200μM NADH引发反应,并在25°C下测量340 nm处吸光度随时间的变化3分钟(NADH[εNADH]的消光系数=6220 M−1·cm−1)。 细胞色素c还原试验[1] 反应介质由77μM细胞色素c、200μM NADH和每种化合物(KL1333、辅酶Q10或艾地苯醌;0.1-100μM范围)在含有0.14%BSA的50 mM Tris-HCl(pH 7.5)缓冲液中组成。在30°c下,使用NADH作为直接电子供体,细胞色素c作为末端电子受体,测量细胞色素c的还原活性。通过加入rhNQO1(5mU)引发反应。根据550 nm处OD的初始变化率和细胞色素c的消光系数(21.1 mM−1·cm−1),活性计算为细胞色素c减少的μmol/mg/min蛋白质。 NAD+/NADH比值的测量[1] 使用EnzyChrom NAD+/NADH检测试剂盒测量细胞内NAD+和NADH水平。简而言之,细胞在100μl NAD+提取缓冲液(用于NAD+测定)或100μl的NADH提取缓冲液中均化(用于NADH测定)。样品在60°C下加热5分钟,然后与20μl测定缓冲液和100μl相反的提取缓冲液混合以中和提取物。接下来,将样品短暂涡旋,并在14000 rpm下离心5分钟。基于乳酸脱氢酶循环反应,对上清液进行NAD+/NADH测定,其中产生的NADH将四唑盐还原为紫色甲赞产物。通过使用微孔板读数器在570nm处测量甲赞的增加来定量NAD+和NADH水平。 为了制备无细胞酶系统,将1μMKL1333与200μM NADH和200μM NAD在含有0.14%BSA的50 mM Tris-HCl(pH 7.5)缓冲液中混合(总体积,200μl)。通过加入rhNQO1(10mU)引发反应,并将测定混合物在37°C下孵育1小时。将20微升反应混合物与100μl NAD+提取缓冲液或100μl的NADH提取缓冲液混合。样品在60°C下加热5分钟,然后与20μl测定缓冲液和100μl相反的提取缓冲液混合以中和提取物。接下来,将样品短暂涡旋,并在14000 rpm下离心5分钟。上清液进行NAD+/NADH测定,并通过监测570 nm处的吸光度在微孔板读数器上测量NAD+和NADH水平。 SIRT1活性的测量[1] 使用SIRT1活性测定试剂盒测量SIRT1活性。将细胞接种到6孔板中(2×105个细胞/孔)。第二天,取出培养基,用1或2μM的KL1333处理细胞。1小时后,收集细胞并在裂解缓冲液中裂解。通过将细胞裂解物加入到含有SIRT1测定缓冲液、氟底物肽(100 mM)和NAD+(100 mmol)的反应混合物中来引发反应。在微孔板荧光计上以2-3分钟的间隔测量荧光强度30分钟(激发,350 nm;发射,460 nm)。SIRT1活性在反应速度的线性范围内计算,并根据WT对照细胞中的蛋白质浓度进行归一化。 |
| 细胞实验 |
萤光素酶测定[1]
将C2C12成肌细胞接种到6孔板中(2×105个细胞/孔)。第二天,使用Turbofect转染试剂将细胞与pGL3小鼠PGC-1α荧光素酶报告基因和编码Renilla荧光素酶的pRL-SV40共转染。让转染的细胞稳定24小时,然后用1μM的KL1333处理24小时。使用双荧光素酶报告检测系统对细胞裂解物进行荧光素酶检测。使用光度计测量萤光素酶活性。萤火虫荧光素酶活性与肾肾萤光素酶活性进行了标准化。 ATP水平的量化[1] 为了测量细胞内ATP水平,使用了ATP测定试剂盒。将人成纤维细胞接种到6孔板中(1.5×105个细胞/孔)。第二天,细胞用1μMKL1333或1μM艾地苯醌处理24小时,然后在100μl细胞裂解缓冲液中裂解。细胞裂解物以13000rpm离心10分钟。使用光度计测量上清液中的ATP水平。 细胞内乳酸水平的测定[1] 使用乳酸比色测定试剂盒测量细胞内乳酸水平。人成纤维细胞用1μM的KL1333处理24小时,然后在测定缓冲液中均质化。细胞裂解物与含有测定缓冲液、底物混合物和酶混合物的反应混合物反应,然后在室温下孵育30分钟。通过监测450nm处的光密度来分析每个样品中的乳酸水平。 细胞内ROS水平的测量[1] 人成纤维细胞用1μMKL1333处理24小时,然后在37°C下用2μM CM-H2DCFDA孵育30分钟。然后用磷酸盐缓冲盐水(PBS)洗涤细胞两次,并重新悬浮在500μl PBS中。使用流式细胞仪分析每个样品中的ROS水平(激发,488 nm;发射,530 nm)。 线粒体质量和线粒体膜电位的测量[1] 用1μMKL1333处理人成纤维细胞24小时。处理后,用PBS洗涤细胞并胰蛋白酶处理,在400 g下离心5分钟,然后重新悬浮在PBS中。细胞悬浮液与200 nM MitoTracker Green FM(激发,488 nM;发射,530 nM)混合以评估线粒体质量,或与200 nMTMRM(激发,488nm;发射)混合以测量线粒体膜电位。然后将细胞在37°C的CO2培养箱中染色30分钟,用PBS洗涤,并使用流式细胞仪进行分析。 在MELAS成纤维细胞研究中,将细胞接种于多孔板,用KL1333(0.1-5 μM)或溶媒处理24-72小时。采用比色法测量NAD+水平,通过细胞外通量分析评估线粒体呼吸。使用发光法量化ATP水平,用荧光探针检测ROS产生。通过蛋白质印迹分析线粒体蛋白和生物合成标志物 [1] 在耳蜗培养物中,从小鼠内耳获取器官型外植体,用KL1333(0.1-1 μM)预处理1小时,随后用顺铂(10 μM)处理24小时。通过免疫荧光染色评估毛细胞存活,通过蛋白质印迹分析凋亡标志物 [2] |
| 动物实验 |
This double-blind, randomized, placebo-controlled, single and multiple oral dose phase 1a/1b study was conducted in four parts (A, B, C and D). Parts A, B and D included a total of eight cohorts of healthy volunteers, while Part C, conducted at completion of Parts A and B, included one cohort of subjects with genetically confirmed PMDs. The primary objective of the study was to evaluate the safety and tolerability of KL1333 in healthy subjects and people with PMD. Other objectives were to explore pharmacokinetics (PK), food effect and pharmacodynamics (PD) of KL1333. An overview of the study design is shown in Fig. 1, Supplementary Table 1 and the Supplementary material. The investigational medicinal products were provided as 25 and 100 mg KL1333 encapsulated tablets and matching placebo tablets.
The two healthy cohorts in Part D, receiving the same daily dose of 150 mg divided in two or three doses, were added later to refine the tolerability profile of KL1333, after reviewing preliminary results from Parts B and C.[3]
Culture of mouse cochlear explants [2] Cochlear explant and culture were performed as previously described. The cultured organs of Corti were divided into four treatment groups: untreated control (CT, n = 5), CP alone (n = 5), KL1333 pretreatment plus CP (KL1333 + CP, n = 5), and KL1333 alone (n = 5). After 16 h equilibration in a humidified atmosphere of 5 % CO2 at 37°C, the appropriate organs were treated with 1 μM KL1333 diluted into culture medium from DMSO solution. After 1 h incubation, 30 μM CP, or vehicle control, was added. Incubation continued for 30 h before collecting the explants for analyses. Histological evaluation [2] To evaluate the protective effects of KL1333 on CP-induced ototoxicity, we examined the morphology of inner hair cells (IHCs) and outer hair cells (OHCs) within the treated organs of Corti. At the end of incubation, the organs were washed with phosphate-buffered saline (PBS), fixed for 15 min with 4 % paraformaldehyde (PFA, pH 7.4) in PBS, and washed with PBS three times. All samples were stained with Alexa Fluor 488-conjugated phalloidin (1:1000) in PBS-Tx, then washed three times with PBS, and mounted on glass slides using Fluoromount. For immunohistochemical quantification, IHCs and OHCs were each counted in three fields near the apex, middle and basal of each cochlear explant. Perfectly shaped hair cells were counted over a 200-μm length of basilar membrane at 30 % (apical), 50 % (middle), and 70 % (basal) positions from the apical end of the cochlear duct, respectively. Each experiment was performed independently and repeated at least five times (n = 5). Images were captured using a Zeiss Axio Imager A2 fluorescence microscope. |
| 药代性质 (ADME/PK) |
In human subjects, plasma exposure (Cmax and AUC) increased proportionally to the dose after oral administration of KL1333 (50-400 mg). The median time to peak concentration (Tmax) was 2-3 hours, and the terminal half-life was approximately 6-8 hours. The drug has good oral bioavailability and can be detected in whole blood and tissues related to mitochondrial function [3].
|
| 毒性/毒理 (Toxicokinetics/TK) |
In the phase 1a/1b clinical trials, KL1333 was well tolerated at doses up to 400 mg/day. No serious adverse events were reported; mild adverse events included transient nausea and headache. No significant changes were observed in liver and kidney function tests, and no evidence of drug interaction was found.[3] In vitro studies showed that KL1333 (at concentrations up to 5 μM) did not induce cytotoxicity in MELAS fibroblasts or normal cochlear cells, indicating good safety.[1][2]
|
| 参考文献 |
|
| 其他信息 |
KL1333 is a novel NAD+ regulator derived from β-lappatone, designed to improve mitochondrial function by enhancing NAD+ availability. It has shown potential in treating mitochondrial diseases such as MELAS and in preventing cisplatin-induced ototoxicity by reducing oxidative stress and apoptosis. Phase 1 clinical trials support its safety and ability to modulate human NAD+ metabolism, paving the way for further research into mitochondrial diseases [1][2][3]. KL-1333 is a small molecule drug that has completed Phase II clinical trials (covering all indications) and has two investigational indications. Our results indicate that KL1333 enhances mitochondrial biosynthesis and function by upregulating the major mitochondrial regulator PGC-1α. PGC-1α is an important target in mitochondrial medicine research because it plays a crucial role in mitochondrial function and metabolism; specifically, it activates multiple transcription factors involved in the expression of mitochondrial and metabolic genes. The activity of PGC-1α can be regulated by post-translational modifications. As shown in Figure 4, we found that KL1333 activates PGC-1α through dual deacetylation and phosphorylation, which may be mediated by SIRT1 and AMPK, respectively. SIRT1 and AMPK are important metabolic sensors that play multiple roles in glucose and lipid metabolism, mitochondrial biosynthesis and transcriptional regulation. Therefore, the potential of KL1333 as an activator of the SIRT1/AMPK/PGC-1α signaling pathway may be extended to age-related diseases and metabolic diseases, such as neurodegenerative diseases, diabetes and non-alcoholic fatty liver. In summary, our results indicate that KL1333 can improve energy balance, reduce oxidative stress, restore mitochondrial function and alleviate the harmful effects of mitochondrial diseases by pharmacologically regulating NAD+ through its action on NQO1. [1] All of these studies have shown that β-lappaone can induce cancer cell-specific apoptosis without causing non-specific cytotoxicity to normal tissues. As a β-lappaone derivative, KL1333 exhibits similar functions to β-lappaone in fibroblasts of MELAS patients. It can increase intracellular ATP levels and mitochondrial membrane potential while reducing reactive oxygen species (ROS) levels. Our results largely confirm these findings. Although further confirmatory studies are needed to determine whether KL1333 can maintain the anticancer effect of CP, we cautiously anticipate that KL1333 has the positive potential to be an effective candidate drug for preventing hearing impairment in patients receiving CP chemotherapy, while potentially maximizing the anticancer effect of CP. [2]
|
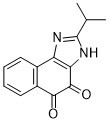
| 分子式 |
C14H12N2O2
|
|---|---|
| 分子量 |
240.257283210754
|
| 精确质量 |
240.089
|
| 元素分析 |
C, 69.99; H, 5.03; N, 11.66; O, 13.32
|
| CAS号 |
1800405-30-4
|
| PubChem CID |
91820639
|
| 外观&性状 |
Reddish brown to red solid powder
|
| LogP |
2.3
|
| tPSA |
62.8
|
| 氢键供体(HBD)数目 |
1
|
| 氢键受体(HBA)数目 |
3
|
| 可旋转键数目(RBC) |
1
|
| 重原子数目 |
18
|
| 分子复杂度/Complexity |
380
|
| 定义原子立体中心数目 |
0
|
| SMILES |
O=C1C(C2C=CC=CC=2C2=C1NC(C(C)C)=N2)=O
|
| InChi Key |
AJFWITSBVLLDCC-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C14H12N2O2/c1-7(2)14-15-10-8-5-3-4-6-9(8)12(17)13(18)11(10)16-14/h3-7H,1-2H3,(H,15,16)
|
| 化学名 |
2-(1-Methylethyl)-3H-naphth(1,2-d)imidazole-4,5-dione
|
| 别名 |
KL-1333; KL 1333; 1800405-30-4; 2-isopropyl-3H-naphtho[1,2-d]imidazole-4,5-dione; NA2ZOL5UGM; UNII-NA2ZOL5UGM; 2-(1-Methylethyl)-3H-naphth(1,2-d)imidazole-4,5-dione; 2-Isopropyl-3H-naphtho(1,2-d)imidazole-4,5-dione; KL1333;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ~100 mg/mL (~416.22 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (10.41 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (10.41 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.1622 mL | 20.8108 mL | 41.6216 mL | |
| 5 mM | 0.8324 mL | 4.1622 mL | 8.3243 mL | |
| 10 mM | 0.4162 mL | 2.0811 mL | 4.1622 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
 KL1333 increases the intracellular NAD+/NADH ratio.
KL1333 induces PGC-1α activation.Front Neurol. 2018; 9: 552. |
|---|
 KL1333 activates SIRT1 and AMPK.
KL1333 regulates intracellular ATP, lactate, and ROS levels in MELAS fibroblasts.Front Neurol. 2018; 9: 552. |
 KL1333 activates AMPK in a NQO1-dependent manner.
KL1333 increases the NAD+/NADH ratio and activates SIRT1, AMPK, and PGC-1α in MELAS fibroblasts.Front Neurol. 2018; 9: |