| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
Lck (IC50 = 4 nM); Fyn (IC50 = 5 nM)[1]
Src family kinases: Lck (IC₅₀ ≈ 4 nM), Fyn (IC₅₀ ≈ 5 nM), Src (IC₅₀ ≈ 6 nM); non-Src kinases: EGFR (IC₅₀ > 1000 nM), Abl (IC₅₀ > 1000 nM), PKC (IC₅₀ > 1000 nM) (showing high selectivity for Src family kinases) [1] - Src kinase (pancreatic adenocarcinoma cells): PP2 (AG1879) inhibited Src phosphorylation (Tyr416) at concentrations of 5 μM–10 μM, with no effect on total Src protein levels [2] |
|---|---|
| 体外研究 (In Vitro) |
在 10 μM 时,PP2 对细胞增殖的影响不具有统计学意义,表明 PP2 在这种低剂量下对吉西他滨细胞毒性的影响可能是由于吉西他滨诱导的细胞增殖,而不是直接的抗增殖作用。毒性升高。在 20 μM 时,生长逐渐受到抑制,这与其他人类癌细胞系的研究结果一致。虽然我们使用 10 μM PP2,但据文献记载,较高浓度的 PP2 会抑制其他细胞内激酶 [2]。市场上最受欢迎的 Src 家族激酶抑制剂是 PP2。 PP2 的体外 IC50 约为 5 nM,可抑制 Src 家族激酶活性。细胞培养物中 Src 家族激酶的完全抑制通常在 10 μM 剂量下实现 [3]。
在人外周血T细胞中:PP2(AG1879)(1 μM–10 μM)浓度依赖性抑制Lck/FynT依赖的T细胞活化。10 μM浓度下:(1)抗CD3/CD28诱导的IL-2分泌减少约80%(ELISA检测);(2)T细胞表面早期活化标志物CD69的表达降低约75%(流式细胞术);(3)Western blot显示Lck(Tyr394)、Fyn(Tyr417)及下游ZAP-70(Tyr493)的磷酸化水平降低[1] - 在人胰腺腺癌细胞系(AsPC-1、BxPC-3)及吉西他滨耐药亚系(AsPC-1/GemR、BxPC-3/GemR)中:PP2(AG1879)(5 μM–10 μM)可逆转吉西他滨耐药:(1)在AsPC-1/GemR中,吉西他滨的IC₅₀从对照组的~150 nM降至10 μM PP2处理组的~30 nM;(2)耐药亚系中细胞增殖(MTT法)被10 μM PP2处理72小时后抑制约50%;(3)凋亡率(Annexin V/PI染色)从对照组的~5%升至10 μM PP2处理48小时后的~35%;(4)Western blot证实p-Src(Tyr416)、p-FAK(Tyr397)及p-ERK1/2(Thr202/Tyr204)水平降低[2] |
| 体内研究 (In Vivo) |
PP2治疗组肿瘤生长抑制率为25%,吉西他滨治疗组肿瘤生长抑制率为5%(P>0.05)。 PP2 和吉西他滨联合使用时,肿瘤生长抑制率为 98%(P<0.05)。对照组和吉西他滨治疗组 100% 发生肝转移;在PP2治疗组中,88%的肝转移形成。在接受PP2和吉西他滨联合治疗的组中,没有观察到转移(P<0.05)[2]。
|
| 酶活实验 |
一种密切相关的吡唑并嘧啶,PP2,在抑制Lck和FynT方面同样有效。在使用其他Src家族蛋白酪氨酸激酶的进一步选择性测试中,PP1还抑制了Src(170 nM)和Hck(20 nM),而PP2显示出对Hck(5 nM)的强效抑制作用(数据未显示)。相比之下,PP1和PP2在抑制A-431表皮生长因子受体自磷酸化方面的活性都低50-100倍(IC50=0.25和0.48μM)。当发现PP1和PP2对ZAP-70和JAK2以及蛋白激酶A的抑制基本上没有活性时,证明了抑制Lck和FynT的进一步特异性(数据未显示)。由于ZAP-70酶的活性在Lck对493位残基进行磷酸化后可能会增强,因此我们还研究了PP1对ZAP-70的抑制是否会在昆虫细胞中与Lck共表达时发生改变,如前所述。尽管ZAP-70与Lck催化结构域的共表达持续导致ZAP-70对p62底物的比活性提高3-4倍,但PP1在高达100μM的浓度下仍无法抑制这种酶(数据未显示)。为了进行比较,我们还检测了之前描述的两种酪氨酸激酶抑制剂星孢菌素和染料木素的活性。发酵产物星孢菌素之前已被证明是一种有效但非选择性的蛋白激酶抑制剂。在本文报道的实验中,发现星孢菌素是p56lck和p59fynT的纳摩尔抑制剂,也是EGF-R激酶的低微摩尔抑制剂。然而,与PP1和PP2不同,它也是ZAP-70和JAK2酪氨酸激酶的强效抑制剂。为了进一步比较,测试了天然异黄酮染料木素抑制四种酪氨酸激酶的能力。正如预期的那样,它是最无效的抑制剂(表1)。因此,相对于其他报道的酪氨酸激酶抑制剂,新化合物PP1和PP2显示出对Src家族激酶(如p56lck和p59fynT)的有效和选择性抑制[1]。
重组Src家族激酶活性测定实验:将重组人Lck、Fyn、Src、EGFR或Abl激酶结构域与含10 μM ATP(含[γ-³²P]ATP)和Src特异性肽底物(序列:KKEEEEYMMMM)的反应缓冲液(50 mM Tris-HCl pH7.4、10 mM MgCl₂、1 mM DTT)孵育。加入浓度为0.1 nM–1000 nM的PP2(AG1879)(溶剂为对照),反应体系总体积25 μL,37℃孵育30分钟后,取20 μL点样至磷酸纤维素滤纸上终止反应。滤纸用0.75%磷酸洗涤3次以去除未掺入的ATP,通过液体闪烁计数测定放射性强度。计算抑制率,将数据拟合至剂量-反应曲线以确定IC₅₀值[1] |
| 细胞实验 |
通过在96孔微量滴定板(Costar Corp.)中将1×105 PBL(一式三份)与160μl含有10%胎牛血清的RPMI 1640培养基与20μl稀释的试验化合物或单独的培养基混合,评估流感诱导的T细胞增殖(见表2,Ag-Sp(代表特异性抗原))。抗原(流感病毒疫苗Fluzone,Connaught Laboratories)通过离心和洗涤(三次)2ml疫苗通过Centricon-3浓缩器(Amicon,股份有限公司)以去除防腐剂并将剩余材料稀释至40ml(1:20)来制备。然后向每个孔中加入20微升抗原,并在37°C的5%CO2中孵育72小时。然后加入[3H]胸苷(0.5μCi/孔),并在37°C下将平板再孵育18小时。用96孔收获机(Tomtec)收获细胞,用Pharmacia Biotechβ平板计数器测定掺入的[3H]胸苷量。根据培养基对照的增殖抑制百分比与添加的试验化合物浓度的关系图,确定引起50%增殖抑制的浓度(IC50)。结果以重复实验的平均IC50表示(见表2)。通过将5×104新鲜PBL、5×104辐照(5000拉德)混合刺激PBL和RPMI 1640培养基中稀释的试验化合物在96孔测定板的每个孔中混合三份,评估单向混合淋巴细胞反应中的T细胞增殖。在37°C的5%CO2中孵育18小时后,向每个孔中加入0.5μCi的[3H]胸苷,再孵育细胞18小时。然后使用Pharmacia Biotechβ板系统收获细胞。抑制百分比由以下方程式确定:抑制百分比=1-(药物处理细胞的平均cpm/对照刺激细胞的平均cpu)×100[1]。
T细胞活化实验:通过密度梯度离心分离人外周血T细胞,重悬于含10% FBS的RPMI 1640培养基中。细胞(1×10⁶个细胞/mL)用PP2(AG1879)(0 μM、1 μM、5 μM、10 μM)预处理1小时,再用抗CD3(5 μg/mL)+抗CD28(2 μg/mL)刺激24小时。(1)IL-2分泌:收集培养上清,通过夹心ELISA检测IL-2浓度;(2)CD69表达:细胞用抗CD69-PE抗体染色30分钟,流式细胞术分析;(3)信号通路分析:裂解细胞,Western blot检测p-Lck(Tyr394)、p-Fyn(Tyr417)、p-ZAP-70(Tyr493)及总Lck/Fyn/ZAP-70[1] - 胰腺腺癌细胞增殖与凋亡实验:(1)增殖检测:将AsPC-1/GemR/BxPC-3/GemR细胞以5×10³个细胞/孔接种于96孔板,用PP2(AG1879)(0 μM–10 μM)单独处理或与吉西他滨(0 nM–200 nM)联合处理72小时。加入MTT试剂,测定570 nm处吸光度,计算细胞活力及IC₅₀;(2)凋亡检测:细胞用10 μM PP2(AG1879)处理48小时,Annexin V-FITC/PI染色后流式细胞术分析;(3)信号通路分析:PP2(AG1879)处理细胞2小时后裂解,Western blot检测p-Src(Tyr416)、总Src、p-FAK(Tyr397)、p-ERK1/2(Thr202/Tyr204)及β-actin[2] |
| 动物实验 |
溶于1% DMSO;5 mg/kg;腹腔注射。
将HT29细胞接种到SCID小鼠的脾脏中。按照先前描述的方法(Ueda等,1979)进行脾脏池内给药。小鼠用异氟烷短暂麻醉后,以10 μL的体积进行脾脏池内注射GRP(1 nmol,n = 9)或NMDA(1 nmol,n = 4;5 nmol,n = 4;10 nmol,n = 6;20 nmol,n = 8;40 nmol,n = 4)。然后,对小鼠进行30分钟的视频记录,并计数其后爪抓挠的次数。为了研究三叉神经NMDA受体在抓挠行为中的作用,我们在颊部注射氯喹前30分钟,分别向小鼠颊部注射D-AP5(10.1 nmol,n = 8)、CP101,606(30.5 nmol,n = 8)或PP2(1 nmol,n = 7)。在GRPR拮抗剂实验中,我们同时向小鼠颊部注射RC-3095(5 nmol)和GRP(1 nmol,n = 5)或NMDA(20 nmol,n = 6),注射体积为10 μL,并记录30分钟内的抓挠次数。对于化学诱导疼痛,将辣椒素(98.2 nmol,n = 5)以 10 μL 的体积皮内注射到左侧面颊,并计数前肢擦拭的次数。[4] Sprague-Dawley 大鼠接受短暂性(90 分钟)大脑中动脉闭塞 (MCAO),并在存活 1 天后进行评估。MCAO 后 30 分钟给予 PP2(1.5 mg/kg,腹腔注射)或载体。使用磁共振成像 (MRI) 和氯化三苯基四氮唑 (TTC) 染色检查病变,并根据 Bederson 等人的方法,使用神经功能评分确定功能结果。Acta Neurol Scand. 2004 年 9 月;110(3):175-9。https://pubmed.ncbi.nlm.nih.gov/15285775/ |
| 参考文献 |
|
| 其他信息 |
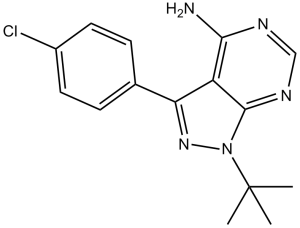
PP2 属于吡唑并嘧啶类化合物,其结构为吡唑并[3,4-d]嘧啶-4-胺,在 1 位和 3 位分别带有叔丁基和 4-氯苯基取代基。它是一种强效的 ATP 竞争性 Src 家族蛋白酪氨酸激酶抑制剂。它具有多种功能,包括作为 EC 2.7.10.2(非特异性蛋白酪氨酸激酶)抑制剂、β-肾上腺素能拮抗剂和抗衰老剂。PP2 是一种吡唑并嘧啶类化合物,属于芳香胺类化合物,也属于单氯苯类化合物。
PP2 (AG1879) 是一种对 Src 家族激酶(Lck、Fyn、Src)具有高度选择性的小分子抑制剂,对非 Src 激酶的活性极低。它被广泛用作研究Src家族介导的信号通路的工具化合物,尤其是在T细胞活化(其中Lck/Fyn是关键调节因子)方面[1]。 在吉西他滨耐药的胰腺腺癌细胞中,PP2 (AG1879)通过抑制Src激酶活化和下游FAK/ERK信号通路恢复吉西他滨敏感性,表明其具有作为辅助药物克服胰腺癌化疗耐药性的潜力。然而,由于缺乏体内疗效和毒性数据,它尚未被开发用于临床[2]。 参考文献[3](侧重于AP23846,一种不同的Src抑制剂)和[4](侧重于瘙痒中NMDA受体GluN2B的磷酸化)不包含与PP2 (AG1879)相关的信息[3][4]。 |
| 分子式 |
C15H16CLN5
|
|
|---|---|---|
| 分子量 |
431.53
|
|
| 精确质量 |
301.109
|
|
| 元素分析 |
C, 59.70; H, 5.34; Cl, 11.75; N, 23.21
|
|
| CAS号 |
172889-27-9
|
|
| 相关CAS号 |
|
|
| PubChem CID |
4878
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| 密度 |
1.4±0.1 g/cm3
|
|
| 沸点 |
493.5±40.0 °C at 760 mmHg
|
|
| 熔点 |
214-216ºC
|
|
| 闪点 |
252.3±27.3 °C
|
|
| 蒸汽压 |
0.0±1.3 mmHg at 25°C
|
|
| 折射率 |
1.676
|
|
| LogP |
3.22
|
|
| tPSA |
69.62
|
|
| 氢键供体(HBD)数目 |
1
|
|
| 氢键受体(HBA)数目 |
4
|
|
| 可旋转键数目(RBC) |
2
|
|
| 重原子数目 |
21
|
|
| 分子复杂度/Complexity |
364
|
|
| 定义原子立体中心数目 |
0
|
|
| SMILES |
ClC1C([H])=C([H])C(=C([H])C=1[H])C1C2=C(N([H])[H])N=C([H])N=C2N(C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H])N=1
|
|
| InChi Key |
PBBRWFOVCUAONR-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C15H16ClN5/c1-15(2,3)21-14-11(13(17)18-8-19-14)12(20-21)9-4-6-10(16)7-5-9/h4-8H,1-3H3,(H2,17,18,19)
|
|
| 化学名 |
1-(tert-butyl)-3-(4-chlorophenyl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine
|
|
| 别名 |
AG 1879; AG-1879; PP2; PP-2; 1-(tert-butyl)-3-(4-chlorophenyl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine; AG 1879; 1-tert-butyl-3-(4-chlorophenyl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine; 1-tert-butyl-3-(4-chlorophenyl)pyrazolo[3,4-d]pyrimidin-4-amine; PP 2; AG1879.
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 3 mg/mL (9.94 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 30.0 mg/mL 澄清 DMSO 储备液加入到 900 μL 玉米油中并混合均匀。 配方 2 中的溶解度: 4% DMSO+30% PEG 300+ddH2O:5 mg/mL 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3173 mL | 11.5867 mL | 23.1734 mL | |
| 5 mM | 0.4635 mL | 2.3173 mL | 4.6347 mL | |
| 10 mM | 0.2317 mL | 1.1587 mL | 2.3173 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT06012734 | Not yet recruiting | Drug: LB-100 Drug: Atezolizumab |
Metastatic Microsatellite-stable Colorectal Cancer |
The Netherlands Cancer Institute | December 2023 | Phase 1 |
| NCT06065462 | Recruiting | Drug: Dostarlimab Drug: LB-100 |
Ovarian Clear Cell Carcinoma | M.D. Anderson Cancer Center | November 10, 2023 | Phase 1 Phase 2 |
| NCT00807365 | Terminated Has Results | Drug: GHRH | Elderly | Johns Hopkins University | December 17, 2007 | Phase 2 |
| NCT00005592 | Completed | Biological: 90-Y-ibritumomab tiuxetan Biological: rituximab |
Lymphoma | University of Alabama at Birmingham | November 1999 | Phase 2 |
|
|
|