| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| Other Sizes |
|
| 靶点 |
Cardiolipin peroxidase[1]
|
|---|---|
| 体外研究 (In Vitro) |
Barth综合征是一种罕见的X连锁遗传性疾病,以线粒体功能障碍为特征。Elamipretide是一种线粒体心磷脂结合剂,可穿透细胞并在线粒体内富集:其靶向定位在线粒体内膜,改善线粒体形态与功能。Elamipretide的作用机制涉及与心磷脂的静电相互作用——心磷脂是对线粒体结构和电子传递链功能至关重要的磷脂。通过与心磷脂结合,elamipretide能稳定线粒体膜、防止氧化损伤并维持膜电位。这种相互作用可保持线粒体嵴的完整性、减少活性氧产生并维持ATP生成。Elamipretide还能增强线粒体呼吸链复合物的活性,促进电子高效传递和ATP合成。此外,elamipretide可抑制线粒体通透性转换孔的开放,在再灌注过程中保护线粒体免受损伤,并减少缺血条件下的细胞死亡。
|
| 体内研究 (In Vivo) |
Elamipretide三乙酸酯(5mg / kg,腹腔注射,每天一次,连续3天)具有神经保护作用,保护海马免受线粒体功能障碍的损害,并减轻脂多糖(HY-D1056)诱导的C57BL / 6小鼠认知障碍模型中的氧化应激和炎症反应[1]。
Elamipretide是一种靶向线粒体心磷脂的结合剂,可定位于线粒体内膜,从而改善线粒体形态与功能。研究显示,Elamipretide能够减轻神经元氧化应激与神经炎症,激活神经线粒体生物合成,增强线粒体呼吸功能,并保护神经细胞免于凋亡。已有报道表明,Elamipretide在创伤性脑损伤、下肢缺血再灌注损伤、II型糖尿病及脊髓损伤模型中具有保护神经元免于丢失和抑制炎症的作用。在达到最大推荐剂量峰值浓度三倍的情况下,未观察到具有临床意义的QTc间期延长。 Elamipretide是一种线粒体心磷脂结合剂,目前正被研究用于治疗涉及线粒体功能障碍的疾病。Elamipretide是一种合成四肽,能选择性结合心磷脂——一种位于线粒体内膜的磷脂。该相互作用可改善线粒体形态与功能。2025年9月,美国食品药品监督管理局(FDA)加速批准FORZINITY™(elamipretide)用于改善体重不低于30公斤的成人与儿童Barth综合征患者的肌力。 ELAMIPRETIDE是一种蛋白质药物,其最高临床试验阶段(针对所有适应症)为III期,并拥有10项研究中的适应症。 |
| 动物实验 |
Animal/Disease Models:LPS-induced cognitive impairment in C57BL/6 mice model[1]
Doses: 5 mg/kg Route of Administration: i.p., once daily for 3 days Experimental Results: Maintained the mitochondrial function, ROS and MDA levels, and SOD activity. Inhibited neural cell apoptosis in hippocampus, enhanced the hippocampal BDNF pathway and synaptic structural complexity. |
| 药代性质 (ADME/PK) |
Absorption
Elamipretide exposure increases proportionally over a dose range of 2 to 80 mg following daily subcutaneous injections with minimal accumulation. Maximum elamipretide concentrations were reached between 0.5 to 1 hour after subcutaneous administration. The absolute bioavailability following subcutaneous administration is approximately 92%. Elamipretide exposure is comparable after subcutaneous injection to the thigh or to the abdomen. Route of Elimination Elamipretide and its metabolites M1 and M2 are excreted in the urine. At 48 hours post-dose, approximately 100% of the elamipretide dose was recovered in the urine as either elamipretide, M1, or M2 in patients with normal renal function. Volume of Distribution Elamipretide is distributed throughout total body water with an approximate volume of distribution of 0.5 L/kg. Protein Binding Protein binding is approximately 39%. Metabolism / Metabolites Elamipretide is metabolized via sequential C-terminal degradation to the M1 tripeptide and M2 dipeptide metabolites, which do not have pharmacological activity. The metabolic pathways have not been fully characterized. |
| 参考文献 |
|
| 其他信息 |
Barth syndrome primarily affects males, typically starts with severe heart failure in infancy, and causes premature death. Patients who survive into adolescence and adulthood often have fatigue, poor stamina, and exercise intolerance. The quality of life and daily functioning of patients with Barth syndrome are significantly affected throughout their lives.
Forzinity works by binding to the inner part of the mitochondria, improving mitochondrial structure and function. FDA granted Forzinity accelerated approval. This pathway can allow earlier approval of medications that treat serious conditions and fill an unmet medical need on the basis of a measure that is considered reasonably likely to predict patient benefit but does not directly assess the benefit to the patient. Forzinity’s accelerated approval is based on improved strength of the muscle used to straighten the leg at the knee. FDA considers this improvement reasonably likely to predict patient benefit, such as an ability to stand more easily or walk farther. As a condition of accelerated approval, FDA is requiring the manufacturer of Forzinity to conduct a post-approval randomized, double-blind, placebo-controlled trial to confirm that the changes seen on knee muscle strength translate into patient benefit.
Forzinity is administered subcutaneously (under the skin) once daily. The most common side effects identified in clinical trials were mild-to-moderate injection site reactions. Serious reactions to Forzinity have also been reported.
|
| 分子式 |
C38H61N9O11
|
|---|---|
| 分子量 |
819.94
|
| 精确质量 |
819.449
|
| CAS号 |
1849610-71-4
|
| 相关CAS号 |
736992-21-5;1334953-95-5 (acetate);2244098-12-0 (HCl); 1606994-55-1
|
| PubChem CID |
155977505
|
| 序列 |
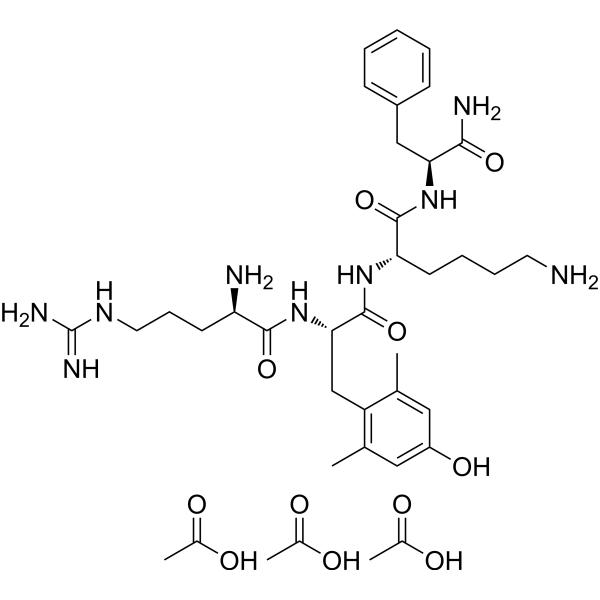
H-D-Arg-Tyr(2,6-diMe)-Lys-Phe-NH2.3CH3CO2H; D-arginyl-2,6-dimethyl-L-tyrosyl-L-lysyl-L-phenylalaninamide acetic acid
|
| 短序列 |
RXKF
|
| 外观&性状 |
Solid powder
|
| tPSA |
379
|
| 氢键供体(HBD)数目 |
12
|
| 氢键受体(HBA)数目 |
14
|
| 可旋转键数目(RBC) |
19
|
| 重原子数目 |
58
|
| 分子复杂度/Complexity |
1020
|
| 定义原子立体中心数目 |
4
|
| SMILES |
CC1=CC(=CC(=C1C[C@@H](C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC2=CC=CC=C2)C(=O)N)NC(=O)[C@@H](CCCN=C(N)N)N)C)O.CC(=O)O.CC(=O)O.CC(=O)O
|
| InChi Key |
ZTOJICBVNLHTEQ-QTSNMJAOSA-N
|
| InChi Code |
InChI=1S/C32H49N9O5.3C2H4O2/c1-19-15-22(42)16-20(2)23(19)18-27(41-29(44)24(34)11-8-14-38-32(36)37)31(46)39-25(12-6-7-13-33)30(45)40-26(28(35)43)17-21-9-4-3-5-10-21;3*1-2(3)4/h3-5,9-10,15-16,24-27,42H,6-8,11-14,17-18,33-34H2,1-2H3,(H2,35,43)(H,39,46)(H,40,45)(H,41,44)(H4,36,37,38);3*1H3,(H,3,4)/t24-,25+,26+,27+;;;/m1.../s1
|
| 化学名 |
acetic acid;(2S)-6-amino-2-[[(2S)-2-[[(2R)-2-amino-5-(diaminomethylideneamino)pentanoyl]amino]-3-(4-hydroxy-2,6-dimethylphenyl)propanoyl]amino]-N-[(2S)-1-amino-1-oxo-3-phenylpropan-2-yl]hexanamide
|
| 别名 |
MTP-131 triacetate; Elamipretide Triacetate; 1849610-71-4; Elamipretide (triacetate); RX-31 triacetate; SS-31 triacetate
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 请将本产品存放在密封且受保护的环境中,避免吸湿/受潮。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
H2O : ≥ 100 mg/mL (121.96 mM) DMSO : 100 mg/mL (121.96 mM; with sonication)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 1 mg/mL (1.22 mM)(饱和度未知) in 10% DMSO 40% PEG300 5% Tween-80 45% Saline (这些助溶剂从左到右依次添加,逐一添加),澄清溶液 。
例如,若需制备1 mL的工作液,可将100 μL 10.0 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀;再向上述体系中加入 50 μL Tween-80,混合均匀;然后再继续加入 450 μL 生理盐水 定容至 1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 1 mg/mL (1.22 mM)(饱和度未知) in 10% DMSO 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加),澄清溶液。 例如,若需制备1 mL的工作液,可将100 μL 10.0 mg/mL 的澄清 DMSO 储备液加到 900 μL玉米油中,混合均匀。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.2196 mL | 6.0980 mL | 12.1960 mL | |
| 5 mM | 0.2439 mL | 1.2196 mL | 2.4392 mL | |
| 10 mM | 0.1220 mL | 0.6098 mL | 1.2196 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。