| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1mg |
|
||
| Other Sizes |
|
| 靶点 |
Oxytocin ( IC50 = 0.65 nM )
|
|---|---|
| 参考文献 | |
| 其他信息 |
瑞托西班是一种二肽。
瑞托西班已用于研究早产和产科早产治疗的临床试验。 药物适应症 治疗自发性早产 |
| 分子式 |
C27H34N4O5
|
|---|---|
| 分子量 |
494.58266
|
| 精确质量 |
494.253
|
| 元素分析 |
C, 65.57; H, 6.93; N, 11.33; O, 16.17
|
| CAS号 |
820957-38-8
|
| 相关CAS号 |
(S)-Retosiban
|
| PubChem CID |
11340891
|
| 外观&性状 |
Off-white to yellow solid powder
|
| LogP |
2.191
|
| tPSA |
108.47
|
| 氢键供体(HBD)数目 |
1
|
| 氢键受体(HBA)数目 |
6
|
| 可旋转键数目(RBC) |
6
|
| 重原子数目 |
36
|
| 分子复杂度/Complexity |
819
|
| 定义原子立体中心数目 |
4
|
| SMILES |
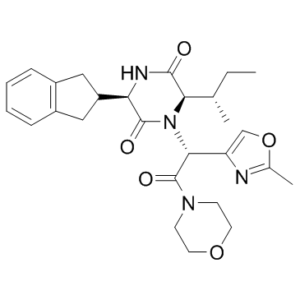
[C@H](C1=COC(C)=N1)(N1C(=O)[C@@H](C2CC3C=CC=CC=3C2)NC(=O)[C@H]1[C@@H](C)CC)C(N1CCOCC1)=O
|
| InChi Key |
PLVGDGRBPMVYPB-FDUHJNRSSA-N
|
| InChi Code |
InChI=1S/C27H34N4O5/c1-4-16(2)23-25(32)29-22(20-13-18-7-5-6-8-19(18)14-20)26(33)31(23)24(21-15-36-17(3)28-21)27(34)30-9-11-35-12-10-30/h5-8,15-16,20,22-24H,4,9-14H2,1-3H3,(H,29,32)/t16-,22+,23+,24+/m0/s1
|
| 化学名 |
(3R,6R)-6-[(2S)-butan-2-yl]-3-(2,3-dihydro-1H-inden-2-yl)-1-[(1R)-1-(2-methyl-1,3-oxazol-4-yl)-2-morpholin-4-yl-2-oxoethyl]piperazine-2,5-dione
|
| 别名 |
GSK221,149; GSK 221,149; GSK-221,149; GSK221149; GSK 221149; GSK-221149; Retosiban; GSK221149A; GSK 221149A; GSK-221149A
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO: ~100 mg/mL (~202.2 mM )
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (5.05 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (5.05 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.5 mg/mL (5.05 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0219 mL | 10.1096 mL | 20.2192 mL | |
| 5 mM | 0.4044 mL | 2.0219 mL | 4.0438 mL | |
| 10 mM | 0.2022 mL | 1.0110 mL | 2.0219 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT01867996 | Completed | Drug: Retosiban Drug: EFZ 600 mg |
Obstetric Labour, Premature | GlaxoSmithKline | June 11, 2013 | Phase 1 |
| NCT02377414 | Completed | Drug: Placebo Drug: Retosiban solution for Infusion |
Obstetric Labour, Premature | GlaxoSmithKline | March 2, 2015 | Phase 1 |
| NCT01702376 | Completed | Drug: Retosiban 100 mg Drug: Retosiban 800 mg Drug: Placebo |
Obstetric Labour, Premature | GlaxoSmithKline | October 3, 2012 | Phase 1 |
| NCT02292784 | Completed | Drug: Retosiban Drug: Atosiban Drug: Placebo |
Obstetric Labour, Premature | GlaxoSmithKline | June 1, 2015 | Phase 3 |
| NCT00404768 | Completed | Drug: GSK221149A Drug: Placebo |
Obstetric Labour, Premature | GlaxoSmithKline | October 12, 2007 | Phase 2 |