| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
Akt
|
|---|---|
| 体外研究 (In Vitro) |
合成化合物SC79抑制PHAKT-GFP质膜易位,但增强细胞质溶胶中Akt的磷酸化和活化。
SC79增强Akt三种亚型的磷酸化,并在多种细胞类型中提高Akt的活化。
SC79特异性增强Akt磷酸化和激活受体酪氨酸激酶和gpcr介导的信号传导。
SC79直接结合Akt并将其转化为更容易被上游激酶磷酸化的活性构象。
SC79减少神经元兴奋毒性并防止中风引起的神经元死亡。[1]
在 HEK293、HeLa、HL60、NB4 和 HsSulton(B 细胞)细胞中,SC79 减少 PHAKTM-GFP 质膜易位并增加所有三种 Akt 亚型的磷酸化。 SC79 减轻神经元兴奋毒性并阻止中风引起的神经元死亡。[1] SC79 增加 MitoSox 阳性细胞产生超氧化物的能力,同时减少 BRAT1 敲低细胞的增殖。 [2] |
| 体内研究 (In Vivo) |
在永久性局灶性脑缺血的小鼠模型中,SC79(0.04 mg/g,腹腔注射)可激活细胞质中的 Akt,并模拟 Akt 信号传导的主要细胞功能,从而增加神经元存活率。 [1]
为了确定SC79是否可以导致Akt过度激活并防止完整生物体中谷氨酸介导的神经毒性,我们使用了缺血性卒中模型。研究人员对小鼠进行大脑中动脉闭塞(MCAO),引起闭塞区域大量细胞死亡。大多数中风的临床症状,如瘫痪、失语、视觉障碍和记忆丧失,都是由缺氧-葡萄糖剥夺(OGD)引起的神经元死亡引起的,这是血液流动不足的结果。脑卒中诱发的OGD在受影响的脑区诱导突触间隙谷氨酸的显著升高,进而导致大量兴奋性毒性引起的神经元细胞死亡(27,28)。Akt被认为是治疗中风引起的神经元死亡的潜在靶点(29-32)。与此一致的是,在小鼠中使用SC79进行i.p.预处理可以有效地预防中风诱导的Akt失活(图4 C和D)。因此,SC79可以保护皮质区和纹状体免受兴奋性毒性诱导的脑损伤。SC79的作用是强大的,单剂量SC79, 0.04 mg/g体重(相当于0.5 μM),在MCAO后24小时将新皮质病变大小减少35%,MCAO后1周减少40%以上(图4E)。当多次注入SC79时,观察到更为剧烈的效果(图4F)。 大鼠围手术期给予SC79(一种选择性Akt激活剂,可透过细胞和血脑屏障)0.05 mg/kg × 3 i.p.或载药i.p.。MCAO 1小时,再灌注2小时后,测定14C-α-氨基异丁酸(14C- aib,分子量104 Da)的传递系数(Ki)和3h -葡聚糖体积(分子量7万Da)分布,测定血脑屏障破坏程度。同一时间点,采用四氮唑染色法测定梗死面积。在另一组大鼠中,给予更高剂量的SC79 (0.5 mg/kg × 3)以确定梗死面积。与未给药的MCAO/再灌注动物相比,SC79增加了缺血再灌注皮质(IR-C, +32%, p < 0.05)和对侧皮质(CC, +35%, p < 0.05)的Ki。SC79对右旋糖酐分布体积无显著影响。SC79治疗显著降低皮质梗死占总皮质面积的百分比(12.7±1.7% vs 6.9±0.9%,p < 0.001)。SC79剂量增加10倍对皮质梗死面积无显著影响。与我们的假设相反,我们的数据表明,尽管血脑屏障破坏增加,但SC79减少了缺血再灌注皮层的梗死面积。我们的数据表明Akt的激活对于治疗窗口内早期脑缺血再灌注神经元存活的重要性,并且神经保护的机制可能与SC79的血脑屏障作用无关。[3] 研究人员在这里报道,Akt激活剂SC79保护肝细胞免受TNF-α-诱导的凋亡,并保护小鼠免受d-半乳糖(d-Gal)/脂多糖(LPS)诱导的TNF-α-介导的肝损伤和损伤。SC79不仅在TNF-α刺激下增强核因子-κB (NF-κB)促生存信号,而且增加细胞FLICE (FADD-like il -1β-转换酶)抑制蛋白L和S (FLIPL/S)的表达,从而抑制procaspase-8的活化。此外,预处理PI3K/Akt抑制剂LY294002可逆转sc79诱导的所有肝保护作用。这些结果强烈提示SC79对TNF-α-诱导的肝细胞凋亡具有保护作用,提示SC79可能是一种有希望改善肝损伤发展的治疗药物。SC79可保护肝细胞免受TNF-α介导的凋亡,并可保护小鼠免受Gal/ lps诱导的肝损伤和损伤。SC79对TNF-α的细胞保护作用是通过akt介导的NF-κB活化和FLIPL/S上调来实现的。[5] 先前的研究表明,Akt的激活可能减轻蛛网膜下腔出血(SAH)后的早期脑损伤(EBI)。本研究旨在确定铁代谢是否参与SAH后Akt激活的有益作用。因此,我们使用了一个新的分子SC79来激活实验性SAH大鼠模型中的Akt。将大鼠随机分为sham、SAH、SAH +载药、SAH + SC79 4组。结果证实,SC79有效增强了SAH后颞叶氧化应激的防御,减轻了EBI。有趣的是,我们发现SC79磷酸化Akt可减少SAH后细胞表面转铁蛋白受体介导的铁摄取,并促进铁转运蛋白介导的铁转运。结果表明,SC79降低了脑组织中的铁含量。此外,受损的Fe-S簇生物发生得到恢复,含Fe-S簇酶活性的丧失得到恢复,表明受损的线粒体功能恢复到健康水平。这些发现表明,铁稳态的破坏可能导致EBI, Akt的激活可能调节铁代谢以减轻铁毒性,进一步保护SAH后的神经元免受EBI。[6] |
| 酶活实验 |
Hela 细胞血清饥饿 1 小时,并用 IGF (100ng/mL) 或 SC79 (4 μg/mL) 处理 30 分钟。将蛋白酶抑制剂添加到裂解缓冲液中,其中含有 250 mM 蔗糖、20 mM HEPES、10 mM KCl、1.5 mM MgCl2、1 mM EDTA 和 1 mM EGTA。用 25G 针多次穿过细胞,然后将其置于冰上 20 分钟。此时,收集整个细胞裂解物。将细胞裂解物以 100,000 g 离心 30 分钟。通过收集上清液获得胞质级分。代表膜部分的沉淀在裂解缓冲液中洗涤。 SDS-PAGE 用于分离总细胞裂解物、胞质和膜部分。然后使用蛋白质印迹法检查磷酸 Akt (S473)、总 Akt、微管蛋白(胞质标记物)和 Orai1(膜标记物)的存在。
Akt质膜易位抑制剂的筛选。为了验证我们基于细胞的高通量筛选方法,我们首先使用生物活性化合物文库(大约3000种化合物)进行了中试筛选。筛选是在哈佛医学院化学和细胞生物学研究所(ICCB)进行的。实验的每一步都在高吞吐量模式下进行(图S3)。根据我们的初步数据,我们将血清饥饿(0.1%血清)的细胞与化合物一起培养30分钟,然后用IGF诱导PHAkt-GFP膜易位。我们的目标是鉴定直接抑制PtdIns(3,4,5)P3/Akt信号通路的化合物。因此,我们选择使用较短的孵育时间来排除间接阻断GFP-PH膜易位的化合物(例如,通过影响转录或翻译)。从先导筛选中,我们鉴定出21种阳性击中化合物(图S4和表S1)。正如预期的那样,几种已知的PI3激酶抑制剂和非特异性抑制PI3激酶活性的化合物被鉴定为阳性命中化合物,验证了我们的高通量筛选策略和方法。然后我们使用几个合成化合物文库进行了高通量筛选。ICCB化合物来自多种来源,包括商业文库、面向多样性的有机合成文库、已知的生物活性化合物和由不同合成策略产生的历史化合物。当ICCB购买化合物文库时,他们选择了富含复杂杂环化合物和高分子量化合物(平均分子量为350-400道尔顿)的集合,因为这些类型的化合物更有可能在高通量筛选中提供有趣的结果。此外,他们还试图减少潜在“有害”化合物的数量,即那些含有可能使它们不稳定或有毒的基团的化合物。特别是,它们消除了不稳定的亚胺,含有游离羧基的化合物,以及含有可能螯合金属的构建块元素的化合物。表S2是用于当前筛选的文库列表,其中包括6万多种合成化合物。高通量筛选进行了两次,以尽量减少假阳性击中化合物的数量。从第一次筛选中,我们确定了大约446个阳性命中化合物,其中125个在第二次筛选中被确认(表S3和图S5)。我们后来发现,其中25种阳性化合物可以产生自荧光,它们对PH-Akt膜转运的影响实际上是背景荧光大大增强的结果(表S3) [1] 通过延时荧光成像确认阳性命中。在这项研究中,筛选了6万多种化合物,很难滴定出每种化合物的最佳浓度。以5 mg/ml的浓度保存于DMSO中。在我们的筛选中,将100 nl的化合物原液转移到50µl的测定体积中,得到500道尔顿化合物的最终浓度为20µM。这是工商银行通常使用的浓度。我们筛选试验的一个潜在问题是,由于孵育时间相对较短,转移的化合物可能无法在每个孔中均匀扩散。因此,某些正打击化合物对PH-Akt膜易位的影响可能是局部浓度非常高的结果。为了选择最有效的化合物进行进一步的表征,我们使用活细胞在35毫米板上培养进行了剂量范围实验。第二次筛选后确定的125种阳性命中化合物(图S5)是从几家公司购买的。新鲜原液用DMSO (5 mg/ml)新鲜配制。将原液直接加入培养基中,得到3种不同的终浓度(4、8、16µg/ml)。为了延时活细胞成像,将HeLa-PH-EGFP细胞镀于35mm玻璃底皿中,培养24 - 48小时。将细胞在2ml Leibovitz L15培养基中血清饥饿1至2h,然后用1ml新鲜的含所需浓度的每种化合物的无血清Leibovitz 3l15培养基替换培养基。预孵育30 min后,加入IGF1 (5 ng/mL),在40×油物镜下每5 ~ 10 min拍照。测定细胞膜与邻近细胞质的相对荧光强度。在16µg/ml或低于16µg/ml时,对PH-EGFP膜易位抑制大于75%的化合物被鉴定并指定为确认命中。55个已确认命中化合物的代表性实时图像和结构如图S6和图S7所示。[1] 圆二色性(CD) [1] 远紫外CD (260-195 nm)在氮气净化的Jasco-810分光偏光计上于25°C下进行。数据采集在1 mm石英试管中,带宽为1 nm,响应时间为2 s,扫描速度为10 nm/min,扫描4次。纯n端6his标记的重组全长人Akt1购自Millipore (www.millipore.com/catalogue/item/14-279)。采用化学合成纯化的SC79。蛋白质和配体样品在50 mM Tris pH 7.5, 150 mM NaCl中制备。Akt1与SC79在37°C下孵育30分钟。数据集一式两份。二级结构百分比的测定采用程序K2D (http://www.embl.de/~andrade/k2d.html)和K2D2 (http://www.ogic.ca/projects/k2d2/)。 |
| 细胞实验 |
将 HsSultan 或 NB4 细胞 (2.5 × 105) 接种于 24 孔板中,加入 500 μL 不含酚红的 RPMI 培养基(补充有 10% FBS)。每种化合物 (8 µg/mL) 在孵育 24 小时后添加,然后培养过夜(16-20 小时)。每个孔接受 50 微升 MTT 溶液(PBS 中 5 mg/mL)。孵育2小时后,将500 L异丙醇和0.1 M HCl直接添加到每个孔中以溶解紫色甲臜晶体。在570 nm波长处,离心去除细胞碎片后测量吸光度。
western blotting分析SC79诱导的Akt胞质磷酸化。[1] 将Hela细胞血清饥饿1小时,然后用IGF (100ng/ml)或SC79(4µg/ml)处理30分钟。细胞在含有250 mM蔗糖、20 mM HEPES、10 mM KCl、1.5 mM MgCl2、1 mM EDTA、1 mM EGTA并添加蛋白酶抑制剂的裂解缓冲液中裂解。25G针多次传代细胞,冰敷20分钟。此时取细胞总裂解液。细胞裂解液以100,000g离心30分钟。收集上清液作为细胞质组分。球团用裂解缓冲液洗涤,代表膜分数。用SDS-PAGE分离细胞总裂解液、胞浆和膜组分,用western blotting分析磷酸化Akt (S473)、总Akt、微管蛋白(胞浆标记物)和Orai1(膜标记物)。 MTT(3-(4,5-二甲基噻唑-2-基)-2,5-二苯基溴化四唑)细胞活力测定。[1] 将HsSultan或NB4细胞(2.5 × 105)在500 μL添加10%胎牛血清的无酚红RPMI培养基中接种于24孔板。孵育24小时后,加入每种化合物(8µg/ml),培养过夜(16-20 h)。每孔加入50微升MTT溶液(PBS中5 mg/ ml)。孵育2 h后,每孔中直接加入500 μL异丙醇和0.1 M盐酸,将紫色甲醛晶体溶解。离心清除细胞碎片后,在570nm波长处测定吸光度。 神经元细胞培养与细胞毒性[1] 原代皮层或海马神经元的培养如前所述。为了诱导兴奋毒性,用Trisbuffered对照盐(CSS)溶液(120 mM NaCl, 5.4 mM KCl, 1.8 mMCaCl2, 25 mM Tris-HCl, pH 7.4和15 mM葡萄糖)对细胞进行预洗,并用含有50µM谷氨酸的CSS处理40分钟,然后在常规培养基中恢复4小时。在谷氨酸治疗前15 min和治疗期间给予SC79(4 μg/ml)。用计算机辅助细胞计数显微镜检查谷氨酸暴露4 h后的毒性。用Hoechst (0.5 ng/ml)和碘化丙啶(1 μg/ml)染色,分别测定总细胞和死亡细胞。孵育20 min后,在360 nm荧光显微镜下观察细胞。细胞死亡以死亡总细胞数之比确定,计数1000个细胞。 |
| 动物实验 |
永久性局灶性脑缺血小鼠模型
0.04 mg/g 腹腔注射 永久性局灶性脑缺血模型[1] 永久性局灶性脑缺血的建立方法基本如前所述(11),即通过大脑中动脉闭塞(MCAO)诱导。简而言之,将体重17-25 g的小鼠(C57 Black/6)用4%异氟烷/66% N2O/30% O2麻醉,并用1.5%异氟烷维持麻醉。永久性局灶性缺血的建立方法如下:在左侧卵圆孔上方和外侧钻一个2 mm的孔,以暴露左侧大脑中动脉。使用双极电凝器永久性闭塞左侧大脑中动脉 (MCA) 近端,闭塞段位于豆纹动脉分支起始部远端 1 mm 处。在永久性 MCAO 前 5 分钟,腹腔注射 SC79(0.04 mg/g 小鼠体重)(图 4E)。在另一项实验中,额外注射 SC79(0.04 mg/g 小鼠体重,每小时一次,持续 6 小时)(图 4F)。 通过免疫组织化学评估脑内 Akt 激活情况 [1] 从心脏心尖用 PBS 灌注小鼠脑组织,并用 4% 多聚甲醛 PBS 溶液进行灌注固定。将组织切片在4%多聚甲醛溶液中于4℃下摇动固定过夜,随后依次在4℃下用10%(2小时)、15%(2小时)、20%(2小时)和25%(过夜)蔗糖PBS溶液进行低温保护。之后将切片包埋于OCT包埋剂中,并迅速用异戊烷冷冻。在低温恒温器上制备10 µm厚的冠状冰冻切片,并保存于-80℃直至使用。将冰冻切片解冻,用PBS洗涤三次,在室温下用0.1% Triton X-100/PBS溶液透化5分钟,然后在5%脱脂奶粉/3% BSA/PBS溶液中封闭60分钟。分别使用抗Akt抗体和抗磷酸化Akt(Ser473)抗体检测总Akt/PKB和磷酸化Akt/PKB。将玻片与一抗(1:200)于4℃孵育过夜,再与二抗于室温孵育2小时,采用ABC法显色。 本研究使用了28只体重220-250 g的雄性Fischer 344大鼠。它们被随机分为两组,每组14只:(1)MCAO/再灌注组,(2)SC79+MCAO/再灌注组。对于SC79+MCAO/再灌注组,分别在短暂性大脑中动脉(MCA)闭塞前10分钟、再灌注时以及再灌注后1小时,腹腔注射0.05 mg/kg的SC79(溶于5% DMSO生理盐水中)。对于MCAO/再灌注组,在每个注射时间点,均给予相同体积的溶剂。每组8只大鼠用于测定血脑屏障通透性参数,6只大鼠用于测定梗死面积。在另一组大鼠(SC79H + MCAO/再灌注组,n = 6)中,给予十倍剂量的SC79(0.5 mg/kg × 3),以确定梗死面积并验证高剂量SC79对神经元存活的影响。所有大鼠均通过气管插管,使用2%异氟烷/氧气混合气体进行通气,以模拟大脑中动脉闭塞。 [3] 本动物实验采用雄性、年龄匹配(6~8周龄)的C57BL/6或BALB/c小鼠(中国科学院上海生命科学研究院,上海,中国),体重16~18 g。小鼠饲养于温湿度可控的房间内,自由摄取食物和水。在腹腔注射致死剂量(0.5 mg/kg)的抗Fas Jo2激动剂抗体(C57BL/6小鼠)或致死剂量(0.4 mg/kg)前0.5小时,小鼠腹腔注射10 mg/kg的SC79或二甲基亚砜进行预处理。小鼠IgG用作Jo2的对照。致死剂量攻击后,持续监测小鼠的死亡率。除死亡率分析外,在Jo2注射后不同时间点处死小鼠。采用标准临床全自动生化分析仪(型号7020;日立,京都,日本)测定血清丙氨酸氨基转移酶(ALT)和天冬氨酸氨基转移酶(AST)水平。采集眼眶后静脉丛血样后,立即采用颈椎脱臼法处死小鼠。切除肝脏组织,切片后置于10%福尔马林溶液中4℃固定过夜,脱水,石蜡包埋,切片厚度为3 mm,并用苏木精-伊红染色进行组织学检查。取出肝组织后,立即用液氮速冻,并保存于-80℃直至分析。[4] 本研究采用雄性、年龄匹配(6-8周龄)、体重16-18 g的C57BL/6小鼠。小鼠饲养于温湿度控制的房间内,可自由获取食物和水。每组10只小鼠在腹腔注射400 mg/kg d-半乳糖胺(d-Gal)和60 µg/kg LPS(C57BL/6小鼠)前0.5小时,分别腹腔注射10 mg/kg SC79或DMSO进行预处理。PBS作为d-Gal/LPS的对照。注射d-Gal/LPS 12小时后处死小鼠。使用标准临床全自动生化分析仪测定血清丙氨酸氨基转移酶(ALT)和天冬氨酸氨基转移酶(AST)水平。采集眼眶后静脉丛血样后,立即采用颈椎脱臼法处死小鼠。切除的肝脏组织切片后,在4℃下用10%福尔马林溶液固定过夜,脱水,石蜡包埋,切片厚度为3 mm,并用苏木精-伊红染色进行组织学检查。提取肝组织后,立即用液氮速冻,并储存在−80°C直至分析。[5] |
| 参考文献 |
|
| 其他信息 |
使用Akt激活剂治疗神经系统疾病的一个注意事项是,Akt信号通路的过度激活可能诱发癌症。然而,通过增强PtdIns P3/Akt信号通路诱发癌症是一个渐进的过程,通常需要数月甚至数年。例如,在髓系特异性PTEN敲除小鼠中,我们在出生后3个月才发现肿瘤。当Akt激活剂用作谷氨酸兴奋性毒性引起的神经元死亡抑制剂时,给药时间通常仅为数天甚至数小时;因此,这种治疗不太可能导致肿瘤发生。有趣的是,最近有报道称,Akt1的激活会降低乳腺上皮细胞的迁移能力,并且Akt1能够阻止类似于转移所需事件的上皮-间质转化。另一项报告显示,在某些急性髓系白血病 (AML) 中,Akt 的激活出人意料地通过抑制 FOXO 来减少白血病细胞的生长,这表明 Akt 激活剂甚至可能用于治疗某些癌症。Akt 也是一种关键酶,参与细胞迁移、免疫细胞激活、胚胎发育、造血和间充质分化以及葡萄糖稳态等其他过程,因此 SC79 可能用于在其他生理和病理情况下调节细胞功能,例如伤口愈合、宿主防御和糖尿病中的血糖控制。例如,SC79 可能对调节 1 型和 2 型糖尿病的血糖调节反应和胰岛素敏感性具有潜在益处。GSK3β 的磷酸化和失活促进糖原合成,从而降低血糖。Akt 介导的 GLUT4 转位介导葡萄糖转运。GSK3β 和 FOXO 也参与糖异生相关基因(如 G6Pase 和 PEPCK4)的表达。在先天免疫中,已有报道指出,使用PTEN抑制剂可通过激活PI3K/Akt通路来增强中性粒细胞功能。SC79可能通过直接激活Akt发挥类似作用。此外,SC79可能还有助于预防心肌梗死,因为心肌梗死中细胞凋亡的获得性抵抗至少部分是由Akt的持续激活介导的。SC79可通过调节eNOS抑制细胞死亡和诱导血管生成,从而在心肌缺血/再灌注损伤、心肌肥大、高血压和血管疾病中发挥广泛的心脏保护作用。[1] 提高Akt激活水平是预防神经系统疾病中进行性神经元死亡的有效临床策略。然而,由于缺乏特异性Akt激活剂,这一努力一直受到阻碍。在此,我们通过基于细胞的高通量化学遗传筛选,鉴定出一种小分子SC79,它能抑制Akt的膜转位,但矛盾的是,它却能在胞质中激活Akt。SC79特异性地结合Akt的PH结构域。与SC79结合的Akt呈现出一种有利于上游蛋白激酶磷酸化的构象。在海马神经元培养系统和缺血性脑卒中小鼠模型中,SC79在胞质中激活Akt足以重现Akt信号通路的主要细胞功能,从而增强神经元存活。因此,SC79是一种独特的特异性Akt激活剂,可用于增强各种生理和病理条件下的Akt活性。[1]
背景:BRAT1(BRCA1相关ATM激活因子1)与BRCA1、ATM和DNA-PKcs相互作用,并参与DNA损伤反应。然而,根据我们之前的研究结果,BRAT1除了参与DNA损伤反应外,还可能参与细胞生长和凋亡,这提示BRAT1可能存在尚未发现的功能。方法:我们利用RNA干扰技术敲低人BRAT1基因,构建了U2OS、Hela和MDA-MA-231三种癌细胞系的稳定BRAT1敲低株。我们检测了BRAT1敲低株与对照株的细胞生长特性以及体外/体内致瘤能力。为了验证BRAT1缺失是否会导致代谢异常,我们检测了BRAT1敲低株和对照株的糖酵解速率、ATP生成量以及丙酮酸脱氢酶(PDH)活性。 BRAT1在生长信号传导中的作用通过Akt/Erk的激活来确定,并使用Akt激活剂SC79进行验证。结果:利用BRAT1敲低的癌细胞系,我们发现BRAT1表达的缺失显著降低了体外和体内的细胞增殖和致瘤性。当BRAT1缺失时,细胞迁移也显著降低。有趣的是,在BRAT1敲低的HeLa细胞中,葡萄糖摄取和线粒体活性氧(ROS)的产生显著增加。此外,这些细胞中Akt和Erk激酶的基础活性和诱导活性均受到抑制,表明细胞生长信号级联异常。因此,用Akt激活剂处理BRAT1敲低的细胞可以改善其增殖并降低线粒体ROS浓度。[2] |
| 分子式 |
C17H17CLN2O5
|
|
|---|---|---|
| 分子量 |
364.78
|
|
| 精确质量 |
364.082
|
|
| 元素分析 |
C, 55.98; H, 4.70; Cl, 9.72; N, 7.68; O, 21.93
|
|
| CAS号 |
305834-79-1
|
|
| 相关CAS号 |
|
|
| PubChem CID |
2810830
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| 密度 |
1.3±0.1 g/cm3
|
|
| 沸点 |
524.8±50.0 °C at 760 mmHg
|
|
| 闪点 |
271.2±30.1 °C
|
|
| 蒸汽压 |
0.0±1.4 mmHg at 25°C
|
|
| 折射率 |
1.564
|
|
| LogP |
3.23
|
|
| tPSA |
112
|
|
| 氢键供体(HBD)数目 |
1
|
|
| 氢键受体(HBA)数目 |
7
|
|
| 可旋转键数目(RBC) |
7
|
|
| 重原子数目 |
25
|
|
| 分子复杂度/Complexity |
611
|
|
| 定义原子立体中心数目 |
0
|
|
| SMILES |
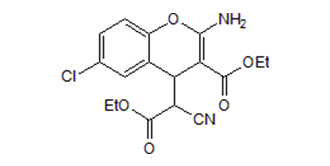
ClC1C([H])=C([H])C2=C(C=1[H])C([H])(C(C(=O)OC([H])([H])C([H])([H])[H])=C(N([H])[H])O2)C([H])(C#N)C(=O)OC([H])([H])C([H])([H])[H]
|
|
| InChi Key |
DXVKFBGVVRSOLI-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C17H17ClN2O5/c1-3-23-16(21)11(8-19)13-10-7-9(18)5-6-12(10)25-15(20)14(13)17(22)24-4-2/h5-7,11,13H,3-4,20H2,1-2H3
|
|
| 化学名 |
ethyl 2-amino-6-chloro-4-(1-cyano-2-ethoxy-2-oxoethyl)-4H-chromene-3-carboxylate
|
|
| 别名 |
SC79; SC-79; 305834-79-1; SC79; ethyl 2-amino-6-chloro-4-(1-cyano-2-ethoxy-2-oxoethyl)-4H-chromene-3-carboxylate; SC 79; SC-79; 4H-1-Benzopyran-4-acetic acid, 2-amino-6-chloro-alpha-cyano-3-(ethoxycarbonyl)-, ethyl ester; 2-amino-6-chloro-alpha-cyano-3-(ethoxycarbonyl)-4h-1-benzopyran-4-acetic acid ethyl ester; MFCD02681303;
SC 79
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 该产品在溶液状态不稳定,请现配现用。 |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO: 72 mg/mL (~197.4 mM)
Water: <1 mg/mL Ethanol: 72 mg/mL (~197.4 mM) |
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 5 mg/mL (13.71 mM) in 5% DMSO + 40% PEG300 + 5% Tween80 + 50% Saline (这些助溶剂从左到右依次添加,逐一添加), 悬浮液;超声助溶。
*生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (6.85 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.5 mg/mL (6.85 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 配方 4 中的溶解度: ≥ 2.5 mg/mL (6.85 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL 澄清 DMSO 储备液加入900 μL 玉米油中,混合均匀。 配方 5 中的溶解度: 2% DMSO+corn oil: 5mg/mL 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7414 mL | 13.7069 mL | 27.4138 mL | |
| 5 mM | 0.5483 mL | 2.7414 mL | 5.4828 mL | |
| 10 mM | 0.2741 mL | 1.3707 mL | 2.7414 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
Loss of BRAT1 leads to inhibition of Akt activity and Akt activation by SC79 partially restores BRAT1 knockdown cells. BMC Cancer. 2014 Jul 29;14:548. doi: 10.1186/1471-2407-14-548. |
 |
 |