| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| Other Sizes |
|
| 靶点 |
Stimulator of Interferon Genes (STING; IC50 = 20 nM)
- STING (Stimulator of Interferon Genes) receptor: STING agonist-4 activates human STING (hSTING) with an EC₅₀ of 0.04 μM, and mouse STING (mSTING) with an EC₅₀ of 0.12 μM [1] |
|---|---|
| 体外研究 (In Vitro) |
STING agonist-4(化合物 2)(0.3-30 μM;2 小时)诱导 IFN-β 的剂量依赖性分泌,EC50 为 3.1 μM,并使 STING 和 IRF3 磷酸化,后者被 TBK1 抑制剂 BX795 抑制 [1]。全长 STING 与固相支持物的结合受到 STING agonist-4(化合物 2)(0.001 nM-1 μM)的抑制,其表观解离常数 (Kd) 约为 1.6 nM [1]。 STING agonist-4(化合物 2)(0-100 μM)的 EC50 为 53.9 μM,其效力是内源 STING 配体 cGAMP 的 18 倍 [1]。化合物 2,也称为 STING 激动剂 4(3 μM;4 小时)通过依赖于 STING 的机制刺激 TNF-α、IL-6 和干扰素 γ 诱导蛋白 10 (IP-10) 的产生介导 TBK1 的激活 [1]。
1. 报告细胞中STING通路激活:用STING agonist-4(0.01–1 μM)处理表达hSTING的HEK293T报告细胞,呈剂量依赖性诱导IFN-β荧光素酶活性,EC₅₀=0.04 μM;处理表达mSTING的报告细胞时,EC₅₀=0.12 μM。该激动剂对STING缺陷型细胞无激活作用 [1] 2. 原代细胞中细胞因子产生:0.1 μM STING agonist-4 处理人外周血单个核细胞(PBMC)后,IFN-β(1250 ± 150 pg/mL)和CXCL10(8500 ± 900 pg/mL)的分泌量较溶剂对照组显著增加;0.3 μM STING agonist-4 处理小鼠骨髓来源巨噬细胞(BMDM)后,IFN-β(820 ± 80 pg/mL)和CXCL10(6200 ± 700 pg/mL)分泌量升高 [1] 3. STING二聚化与磷酸化:Western blot分析显示,0.1 μM STING agonist-4 处理6小时后,hSTING-HEK293T细胞和小鼠BMDM中均出现STING二聚化,且STING磷酸化(p-STING)及下游IRF3磷酸化(p-IRF3)水平升高 [1] 4. 共培养体系中的抗肿瘤活性:将经0.1 μM STING agonist-4 处理的人PBMC与A549肺癌细胞共培养,肿瘤细胞存活率较未处理PBMC组显著降低(抑制率=45 ± 5%),该效应可被抗IFN-β中和抗体阻断 [1] |
| 体内研究 (In Vivo) |
具体来说,我们通过静电相互作用将OMV与亚铁离子固定,并加载STING激动剂-4,然后进行肿瘤靶向DSPE-PEG-FA修饰,从而实现OMV/SaFeFA的功能化。铁离子的锚定使omv具有过氧化物酶样活性,能够通过催化H2O2生成•OH来诱导细胞脂质过氧化。此外,OMV/SaFeFA表现出亚铁离子和激动剂的ph响应性释放,以及肿瘤靶向能力,实现肿瘤特异性治疗,同时最大限度地减少副作用。值得注意的是,STING通路和铁下垂的同时激活在结肠肿瘤小鼠模型中引起了强大的抗肿瘤反应,从而导致了卓越的治疗效果和延长的生存期。重要的是,在接受OMV/SaFeFA治疗的小鼠中未观察到急性毒性,强调了其在未来肿瘤治疗和临床转化中的潜力。[2]
OMV/FA在体内的肿瘤靶向能力[2] 药物输送系统的肿瘤蓄积能力对增强治疗效果和随后的抗肿瘤免疫反应至关重要。因此,我们通过装载荧光菁5 (CY5)分子并与DSPE-PEG(简称OMV/CY5- peg)或DSPE-PEG- fa(简称OMV/CY5- peg - fa)进行聚乙二醇化来研究工程OMV的生物分布。在结肠肿瘤模型中,9只MC38荷瘤C57/BL6小鼠随机分为3组。小鼠静脉注射CY5、OMV/CY5- peg和OMV/CY5- peg - fa。随后,使用IVIS系统记录注射后不同时间点(20 min、2 h、4 h、8 h、10 h和24 h)的荧光图像和信号(图3a)。如图3b和c所示,在给予游离CY5后,在肿瘤部位观察到最小的荧光信号。相反,经OMV/CY5- peg处理的小鼠在肿瘤部位表现出较强的荧光信号,表明OMV对CY5分子的靶向作用增强。值得注意的是,与游离CY5和OMV/CY5- peg处理的小鼠相比,注射OMV/CY5- peg - fa的小鼠显示出明显更高的荧光强度。24 h后处死小鼠,取肿瘤及主要脏器(心、肝、脾、肺、肾)成像。从图3d和e中可以看出,OMV/ cy5 - peg - fa处理小鼠的肿瘤荧光信号明显高于其他两组,进一步证明了DSPE-PEG-FA修饰OMV增强了OMV的肿瘤靶向性。此外,在乳腺肿瘤(4T1)小鼠模型中,基于组织成像(图S17)和肿瘤切片的CLSM图像(图S18), OMV/CY5- peg - fa也表现出优于游离CY5和OMV/CY5- peg的肿瘤靶向性。这些发现进一步证明了dspe - peg - fa修饰的OMV具有良好的肿瘤靶向能力,表明制备的OMV/SaFeFA具有肿瘤靶向治疗的潜力。 1. 小鼠肿瘤模型中的抗肿瘤疗效:对B16-F10黑色素瘤荷瘤C57BL/6小鼠和MC38结直肠癌荷瘤小鼠,静脉注射STING agonist-4(1 mg/kg,每周1次,连续3周),肿瘤体积分别减少68 ± 7%和72 ± 6%(相较于溶剂对照组)。肿瘤生长抑制伴随瘤内CD8⁺ T细胞(增加2.8倍)和NK细胞(增加1.9倍)浸润增多 [1] 2. 全身性免疫激活:小鼠静脉注射STING agonist-4(1 mg/kg)后6小时,血清IFN-β(350 ± 40 pg/mL)和CXCL10(5200 ± 600 pg/mL)水平升高,24小时恢复至基线。处理组小鼠脾细胞中,CD8⁺ T细胞和NK细胞表面激活标志物CD69的表达分别增加3.2倍和2.5倍 [1] 3. 远位效应:在双侧B16-F10肿瘤荷瘤小鼠中,对一侧肿瘤给予STING agonist-4(1 mg/kg,静脉注射)治疗,可同时抑制治疗侧和未治疗对侧肿瘤的生长(对侧肿瘤体积减少55 ± 5%),提示诱导了全身性抗肿瘤免疫 [1] |
| 酶活实验 |
为了及早发现任何潜在的脱靶问题,我们对化合物2 (STING激动剂-4)采用了基于亲和富集的化学蛋白质组学策略。化合物5是一种具有伯胺功能的活性类似物,被共价固定在蔗糖珠上,用于从THP1细胞裂解液中亲和捕获潜在的靶蛋白。下拉实验在没有游离化合物2的情况下进行,以从背景中描绘目标蛋白,或者在化合物2存在的浓度范围内。在不同条件下捕获的所有蛋白质都被洗脱,随后通过色氨酸同位素标记进行定量,然后进行LC-MS /MS分析,建立竞争结合曲线并确定半最大抑制(IC50)值。在这些实验中获得的IC50值代表了靶亲和力的量度,但也受到靶对头固定配体亲和力的影响。后一种效应可以通过测定靶蛋白被小珠消耗来推断,这样就可以确定表观解离常数,这在很大程度上与小珠配体无关(详见补充方法)。值得注意的是,在1000倍的窗口内,只有两种蛋白质被捕获并以剂量依赖的方式竞争,即STING和orosomucoid1 (ORM1, α -1-酸性糖蛋白1前体)。STING的平均值为1.6 nM,表明化合物2不仅在截断蛋白的人工生化检测系统中对目标蛋白具有高效能,而且对全长内源人蛋白也具有高效能。唯一鉴定出的脱靶蛋白ORM1的平均值为79 nM,选择性窗口约为40倍。ORM1是一种急性期反应物,是一种丰富的血浆蛋白,已知具有药物结合特性,已知在单核细胞中表达[1]。
释放行为试验[2] 制备OMV/SaFeFA并在不同pH值的缓冲溶液中孵育不同时间(0,1,2,4,8和12 h)。随后,样品在14,000 rpm下离心10分钟,收集上清以评估STING激动剂-4和Fe2+离子的水平。STING激动剂水平通过测量320 nm处的紫外-可见光谱吸光度来确定,而Fe2+离子水平通过菲罗啉分光光度法来评估。STING激动剂-4来自Invivochem[2] 1. STING报告基因实验:将hSTING或mSTING表达质粒与IFN-β荧光素酶报告质粒共转染HEK293T细胞,24小时后用系列浓度的STING agonist-4(0.001–10 μM)处理16小时。检测荧光素酶活性,根据剂量-反应曲线计算EC₅₀值 [1] 2. STING磷酸化实验:接种hSTING-HEK293T细胞或小鼠BMDM,用STING agonist-4(0.01–1 μM)处理6小时后裂解细胞,通过SDS-PAGE分离蛋白,采用STING、磷酸化STING(p-STING)、磷酸化IRF3(p-IRF3)及内参GAPDH抗体进行Western blot,检测通路激活情况 [1] |
| 细胞实验 |
细胞活力测定[1]
细胞类型: 人外周血单核细胞 (PBMC) 测试浓度: 0.3 μM、1 μM、3 μM、10 μM 和 30 μM 孵育时间: 2 小时 实验结果: 导致 IRF3 和 STING 磷酸化并诱导分泌干扰素-β 。 CCK-8化验[2] MC38细胞(5000个)接种于96孔板,培养过夜。接着,将PBS、OMV/FA、OMV/FeFA、OMV/SaFA和OMV/SaFeFA分别添加到细胞培养基中,OMV浓度为15µg/mL, STING激动剂-4浓度为7.5µg/mL。24 h后,使用CCK-8试剂盒评估细胞活力。[2] ROS和•OH成像[2] MC38细胞(1 × 105)接种于共聚焦培养皿中,培养过夜。然后,在细胞培养基中加入PBS、OMV/FA、OMV/FeFA、OMV/SaFA、OMV/SaFeFA、H2O2(200µM)和OMV/SaFeFA + H2O2(200µM)。OMV和STING激动剂-4浓度分别为15和7.5µg/mL。24 h后,按照制造商的方案,用DCFH-DA (ROS荧光探针)和BboxiProbe O26(•OH荧光探针)对细胞进行染色,并用CLSM成像。用Image J软件测量图像的荧光强度。 Ferroptosis研究[2] MC38细胞(1 × 105)接种于共聚焦培养皿中,培养过夜。随后,在细胞培养基中加入PBS、OMV/FA、OMV/FeFA、OMV/SaFA、OMV/SaFeFA和FeCl2(30µg/mL)。OMV和STING激动剂-4浓度分别为15和7.5µg/mL。24小时孵育后,根据制造商的方案,用Liperfluo探针对细胞进行染色,并使用CLSM成像。STING激动剂-4来自Invivochem 1. PBMC细胞因子产生实验:分离人PBMC并接种,用STING agonist-4(0.01–1 μM)处理24小时后收集培养上清,通过酶联免疫吸附试验(ELISA)定量IFN-β和CXCL10浓度;小鼠BMDM按类似方法制备,用0.03–3 μM STING agonist-4 处理后检测细胞因子 [1] 2. 肿瘤细胞-PBMC共培养实验:将A549肺癌细胞接种于96孔板,贴壁后按效应细胞:靶细胞=10:1的比例加入人PBMC,同时加入STING agonist-4(0.01–1 μM)。共培养72小时后,用细胞增殖检测试剂盒测定肿瘤细胞存活率;中和实验中向共培养体系加入抗IFN-β抗体 [1] |
| 动物实验 |
Biodistribution[2]
All mouse experiments were implemented in accordance with protocols approved by the Animal Experimental Ethics Committee of the Fujian Normal University (No. IACUC-20220005). C57/BL6 and BALB/c mice (4–6 weeks) were used. Nine MC38 tumor-bearing C57/BL6 mice were randomly divided into 3 groups. Mice were i.v. injected with CY5, OMV/CY5-PEG, and, OMV/CY5-PEG-FA, with CY5 and OMV doses set at 50 µg and 60 µg, respectively. Then, the fluorescent signals of mice were recorded at different time points (20 min, 2 h, 4 h, 8 h, 10 h, and 24 h) using an IVIS system. After 24 h, mice were euthanized, and the fluorescent signals of tumors and major organs were recorded. For the 4T1 breast tumor model, nine tumor-bearing BALB/c mice were randomly allocated into 3 groups. These mice received i.v. injection of CY5, OMV/CY5-PEG, and, OMV/CY5-PEG-FA. At 24 h post-injection, mice were euthanized, and the fluorescent signals of tumors and major organs were recorded. Fluorescence images of tumor slices were captured using a CLSM. In vivo therapy[2] C57/BL6 mice received an injection of 1 × 106 MC38 cells in the right hind leg flank. Ten days later, the MC38 tumor-bearing mice were randomly divided into 5 groups (n = 4) and i.v injected with PBS, OMV/FA, OMV/SaFA, OMV/FeFA, and OMV/SaFeFA on days 0 and 4. Tumor size and body weight were measured every two days throughout the treatment period, with tumor volumes calculated using the formula: V= (length × width2)/2. On day 16, mice were euthanized, and the tumors were collected. Tumors were weighted, sliced, and analyzed for H&E, TUNEL staining, and immunohistochemical staining of IFN-γ, SLC7A11, GPX4, NCOA4, and FTH1. Serum was collected for IFN-γ detection using a commercial kit. In addition, the long-term survival rates were evaluated by the same procedures except for monitoring 58-day period. Biosafety[2] PBS, OMV/FA, OMV/FeFA, OMV/SaFA, and OMV/SaFeFA were administered via the tail vein of C57/BL6 mice. After a 16-day treatment, blood samples from C57/BL6 mice were collected for biochemical analysis. The parameters measured included blood urea nitrogen (BUN), aspartate aminotransferase (AST), alanine aminotransferase (ALT), and alkaline phosphatase (ALP), and gamma glutamyl transpeptidase (γ-GT), using assay kits from Solarbio (BC1555, BC1535, BC2145, BC1565, and BC1225). Subsequently, mice were sacrificed, and the major organs (heart, liver, spleen, lung, and kidney) were harvested and subjected to H&E staining for histological analysis. 1. Subcutaneous tumor models: C57BL/6 mice were subcutaneously inoculated with B16-F10 melanoma cells (1×10⁶ cells/mouse) or MC38 colon cancer cells (5×10⁵ cells/mouse). When tumors reached 100–150 mm³, mice were randomly divided into treatment and control groups. STING agonist-4 was administered intravenously at 1 mg/kg once weekly for 3 weeks, while the control group received vehicle. Tumor volume was measured every 2 days, and mice were sacrificed at the end of treatment to collect tumors and spleen for immune cell analysis [1] 2. Abscopal effect model: C57BL/6 mice were subcutaneously inoculated with B16-F10 cells in both left and right flanks. When tumors reached 100–150 mm³, STING agonist-4 (1 mg/kg) was intravenously administered once weekly for 3 weeks. Tumor volumes of both treated and contralateral tumors were measured every 2 days [1] 3. Systemic immune activation assessment: C57BL/6 mice were intravenously injected with STING agonist-4 (1 mg/kg) or vehicle. At 6, 12, and 24 hours post-administration, blood was collected to measure serum IFN-β and CXCL10 levels by ELISA. Spleens were harvested at 24 hours, and splenocytes were analyzed by flow cytometry for CD69 expression on CD8⁺ T cells and NK cells [1] |
| 药代性质 (ADME/PK) |
1. Plasma pharmacokinetics: Intravenous administration of STING agonist-4 (1 mg/kg) to C57BL/6 mice showed a plasma half-life (t₁/₂) of 4.2 ± 0.5 hours, maximum plasma concentration (Cₘₐₓ) of 8.5 ± 1.2 μM, and area under the curve (AUC₀₋₂₄h) of 32.6 ± 3.8 μM·h [1]
2. Tissue distribution: At 2 hours post-intravenous administration (1 mg/kg), STING agonist-4 was distributed in major tissues including liver (6.8 ± 0.8 μg/g), spleen (5.2 ± 0.6 μg/g), lungs (4.5 ± 0.5 μg/g), and tumors (3.9 ± 0.4 μg/g). Low accumulation was observed in brain (0.3 ± 0.1 μg/g) [1] 3. Metabolism: In vitro liver microsome incubation showed that STING agonist-4 was metabolized slowly, with a metabolic stability half-life (t₁/₂) of 38 ± 4 minutes in human liver microsomes and 45 ± 5 minutes in mouse liver microsomes [1] |
| 毒性/毒理 (Toxicokinetics/TK) |
1. Acute toxicity: Intravenous administration of STING agonist-4 at doses up to 5 mg/kg (5-fold the therapeutic dose) to C57BL/6 mice caused no mortality within 14 days. Mice showed no significant changes in body weight, and histopathological analysis of liver and kidney revealed no obvious tissue damage [1]
2. Hematological and biochemical parameters: Mice treated with STING agonist-4 (1 mg/kg, weekly for 3 weeks) showed no significant abnormalities in white blood cell count, red blood cell count, platelet count, or liver function markers (ALT, AST) and kidney function markers (BUN, creatinine) compared to vehicle control [1] |
| 参考文献 |
[1]. Design of amidobenzimidazole STING receptor agonists with systemic activity. Nature. 2018 Dec;564(7736):439-443.
[2]. Metal ions-anchored bacterial outer membrane vesicles for enhanced ferroptosis induction and immune stimulation in targeted antitumor therapy. J Nanobiotechnology . 2024 Aug 9;22(1):474. |
| 其他信息 |
Stimulator of interferon genes (STING) is a receptor in the endoplasmic reticulum that propagates innate immune sensing of cytosolic pathogen-derived and self DNA. The development of compounds that modulate STING has recently been the focus of intense research for the treatment of cancer and infectious diseases and as vaccine adjuvants. To our knowledge, current efforts are focused on the development of modified cyclic dinucleotides that mimic the endogenous STING ligand cGAMP; these have progressed into clinical trials in patients with solid accessible tumours amenable to intratumoral delivery. Here we report the discovery of a small molecule STING agonist that is not a cyclic dinucleotide and is systemically efficacious for treating tumours in mice. We developed a linking strategy to synergize the effect of two symmetry-related amidobenzimidazole (ABZI)-based compounds to create linked ABZIs (diABZIs) with enhanced binding to STING and cellular function. Intravenous administration of a diABZI STING agonist to immunocompetent mice with established syngeneic colon tumours elicited strong anti-tumour activity, with complete and lasting regression of tumours. Our findings represent a milestone in the rapidly growing field of immune-modifying cancer therapies.[1]
The activation of ferroptosis presents a versatile strategy for enhancing the antitumor immune responses in cancer therapy. However, developing ferroptosis inducers that combine high biocompatibility and therapeutic efficiency remains challenging. In this study, we propose a novel approach using biological nanoparticles derived from outer membrane vesicles (OMVs) of Escherichia coli for tumor treatment, aiming to activate ferroptosis and stimulate the immune responses. Specifically, we functionalize the OMVs by anchoring them with ferrous ions via electrostatic interactions and loading them with the STING agonist-4, followed by tumor-targeting DSPE-PEG-FA decoration, henceforth referred to as OMV/SaFeFA. The anchoring of ferrous ions endows the OMVs with peroxidase-like activity, capable of inducing cellular lipid peroxidation by catalyzing H2O2 to •OH. Furthermore, OMV/SaFeFA exhibits pH-responsive release of ferrous ions and the agonist, along with tumor-targeting capabilities, enabling tumor-specific therapy while minimizing side effects. Notably, the concurrent activation of the STING pathway and ferroptosis elicits robust antitumor responses in colon tumor-bearing mouse models, leading to exceptional therapeutic efficacy and prolonged survival. Importantly, no acute toxicity was observed in mice receiving OMV/SaFeFA treatments, underscoring its potential for future tumor therapy and clinical translation.[2] 1. STING agonist-4 is an amidobenzimidazole-derived STING receptor agonist with high selectivity for STING over other innate immune receptors (TLR4, TLR9, RIG-I) [1] 2. The antitumor mechanism of STING agonist-4 involves activation of the STING-IRF3 pathway, induction of I type interferons and proinflammatory cytokines, and enhancement of antitumor immune responses by increasing infiltration of cytotoxic T cells and NK cells into tumors [1] 3. STING agonist-4 exhibits favorable pharmacokinetic properties (long half-life, good tissue distribution) and low acute toxicity, supporting its potential for systemic antitumor therapy [1] |
| 分子式 |
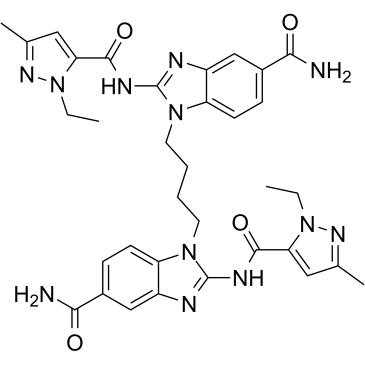
C34H38N12O4
|
|---|---|
| 分子量 |
678.7435
|
| 精确质量 |
678.313
|
| 元素分析 |
C, 60.16; H, 5.64; N, 24.76; O, 9.43
|
| CAS号 |
2138300-40-8
|
| PubChem CID |
132000066
|
| 外观&性状 |
Typically exists as white to gray solids
|
| LogP |
2
|
| tPSA |
216Ų
|
| 氢键供体(HBD)数目 |
4
|
| 氢键受体(HBA)数目 |
8
|
| 可旋转键数目(RBC) |
13
|
| 重原子数目 |
50
|
| 分子复杂度/Complexity |
1150
|
| 定义原子立体中心数目 |
0
|
| InChi Key |
ICZSAXDKFXTSGL-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C34H38N12O4/c1-5-45-27(15-19(3)41-45)31(49)39-33-37-23-17-21(29(35)47)9-11-25(23)43(33)13-7-8-14-44-26-12-10-22(30(36)48)18-24(26)38-34(44)40-32(50)28-16-20(4)42-46(28)6-2/h9-12,15-18H,5-8,13-14H2,1-4H3,(H2,35,47)(H2,36,48)(H,37,39,49)(H,38,40,50)
|
| 化学名 |
1-[4-[5-carbamoyl-2-[(2-ethyl-5-methylpyrazole-3-carbonyl)amino]benzimidazol-1-yl]butyl]-2-[(2-ethyl-5-methylpyrazole-3-carbonyl)amino]benzimidazole-5-carboxamide
|
| 别名 |
STING agonist-4; 2138300-40-8; diABZI STING agonist-2; STING agonist 2; 1,1'-(1,4-butanediyl)bis[2-[[(1-ethyl-3-methyl-1H-pyrazol-5-yl)carbonyl]amino]-1H-benzimidazole-5-carboxamide; CHEMBL4440744; STING agonist diABZI compound 2; 1-[4-[5-carbamoyl-2-[(2-ethyl-5-methylpyrazole-3-carbonyl)amino]benzimidazol-1-yl]butyl]-2-[(2-ethyl-5-methylpyrazole-3-carbonyl)amino]benzimidazole-5-carboxamide;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 请将本产品存放在密封且受保护的环境中(例如氮气保护),避免吸湿/受潮。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ~10 mg/mL (~14.73 mM)
H2O : < 0.1 mg/mL |
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 1 mg/mL (1.47 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 悬浮液;超声助溶。
例如,若需制备1 mL的工作液,可将100 μL 10.0 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: 1 mg/mL (1.47 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 悬浊液; 超声助溶。 例如,若需制备1 mL的工作液,可将 100 μL 10.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.4733 mL | 7.3666 mL | 14.7332 mL | |
| 5 mM | 0.2947 mL | 1.4733 mL | 2.9466 mL | |
| 10 mM | 0.1473 mL | 0.7367 mL | 1.4733 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。