| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|

| 靶点 |
α1-adrenergic receptor
α1-adrenergic receptor (α1-AR) subtypes, with highest affinity for α1A-AR (Ki = 0.034 nM) and α1D-AR (Ki = 0.23 nM), lower affinity for α1B-AR (Ki = 4.5 nM) [1] - α1A- and α1D-adrenergic receptors, which mediate smooth muscle contraction in the prostate and vasculature [2] |
|---|---|
| 体外研究 (In Vitro) |
体外活性:坦索罗辛是一种选择性 α1 受体拮抗剂,相对于血管中的 α1B 受体,它对前列腺中的 α1A 受体具有优先选择性。与对照患者相比,接受坦索罗辛治疗的患者发生急性尿潴留的风险降低了 0.30 倍。国际节制协会男性问卷各领域得分均未显示各组之间存在显着变化。坦索罗辛可推荐用于治疗 AUR 插管后的男性,并且可以减少需要重新插管的可能性。激酶测定: 细胞测定:
Tamsulosin HCl 在人前列腺膜制剂中竞争性抑制[3H]-哌唑嗪与α1-AR亚型的结合,对α1A-AR和α1D-AR的拮抗作用具有亚型选择性,优于对α1B-AR的作用[1] - Tamsulosin HCl 通过抑制PI3K/Akt/mTOR信号通路,抑制血管紧张素II诱导的小鼠主动脉平滑肌细胞(ASMCs)增殖和迁移;同时降低ASMCs中I型胶原蛋白和基质金属蛋白酶-9(MMP-9)的表达[2] |
| 体内研究 (In Vivo) |
坦索罗辛表现出高血浆蛋白结合,主要与 α1-酸性糖蛋白结合。它主要通过细胞色素 P450 (CYP) 3A4 和 CYP2D6 代谢为低丰度化合物,口服剂量的 8.7-15% 作为母体化合物经肾脏排泄。坦索罗辛的药代动力学在很大程度上不受年龄的影响,肾功能不全患者的药代动力学变化很大程度上与α1-酸性糖蛋白浓度的增加有关。肝功能损害引起的药代动力学改变也仅为中度,因此肾功能损害和轻至中度肝功能损害都不需要调整剂量。对大鼠和狗等实验动物进行坦索罗辛速释的早期研究表明,口服给药后(30-90 分钟内)坦索罗辛吸收迅速,但大鼠的绝对生物利用度仅为 7-23%,狗的绝对生物利用度仅为 30-42%。坦索罗辛 MR 在禁食人体中的绝对生物利用度约为 100%。在禁食状态下,tmax 通常为约 5 小时(报告的平均值范围为 2.9-5.6 小时),在进食状态下为约 6 小时(范围为 5.2-7.0 小时)。静脉注射放射性标记坦索罗辛并在10分钟后测量各组织中放射性标记的动物研究表明,该药物存在于各组织中,排列顺序如下:肾>肺≈心脏>颌下腺>肝≈脾≈主动脉≈输精管>前列腺>>大脑皮层,后者接近检测限。坦索罗辛可能无法通过血脑屏障。坦索罗辛在大鼠和狗体内进行广泛的肝脏代谢,分别仅以母体化合物的形式在尿液中排泄 1.2% 和 2.8%。在人类中,坦索罗辛也经历广泛的肝脏代谢,但显然比大鼠或狗的代谢程度稍小,因为口服剂量的 8.7-15% 以非代谢形式从尿液中排出。坦索罗辛从体内消除的速度因物种而异,例如坦索罗辛在大鼠和狗中的消除速度比在人类中更快。
在良性前列腺增生(BPH)模型大鼠中,Tamsulosin HCl(0.01-0.1 mg/kg,口服)通过松弛前列腺和膀胱颈平滑肌,降低尿道内压力并改善尿流率[1] - 在血管紧张素II诱导的腹主动脉瘤(AAA)ApoE-/-小鼠模型中,Tamsulosin HCl(0.1 mg/kg/天,灌胃给药28天)可抑制AAA生长(最大主动脉直径减少31.2%),并减轻主动脉壁炎症、弹性蛋白降解和平滑肌细胞丢失[2] |
| 酶活实验 |
取人前列腺组织制备膜制剂,与[3H]-哌唑嗪(放射性配体)和不同浓度的Tamsulosin HCl在25°C下孵育60分钟。通过过滤分离结合态和游离态配体,测定放射性强度以确定对α1-AR亚型的结合亲和力(Ki值)[1]
- 采用重组人α1A-、α1B-和α1D-AR表达细胞的膜制剂进行亚型特异性放射性配体结合实验,绘制竞争曲线以计算IC50值和亚型选择性比率[1] |
| 细胞实验 |
从主动脉组织中分离小鼠ASMCs,在添加胎牛血清的DMEM培养基中培养。细胞先用Tamsulosin HCl(1-10 μM)预处理1小时,再用血管紧张素II(100 nM)刺激24-48小时。通过CCK-8法检测细胞增殖,Transwell法检测细胞迁移,Western blot检测蛋白表达(PI3K、Akt、mTOR、MMP-9)[2]
- 经Tamsulosin HCl和血管紧张素II处理48小时后,采用酶联免疫吸附试验(ELISA)测定ASMCs分泌的I型胶原蛋白水平[2] |
| 动物实验 |
溶于生理盐水;0.1 和 1 μg/kg;皮下注射
雌性 Wistar 大鼠 良性前列腺增生 (BPH) 模型大鼠(丙酸睾酮注射诱导)随机分为对照组和治疗组。盐酸坦索罗辛以 0.01、0.03 和 0.1 mg/kg 的剂量每日一次口服给药,持续 2 周。在麻醉状态下测量尿流率参数和尿道内压[1] -ApoE-/- 小鼠(8-10 周龄)植入渗透泵,以 1000 ng/kg/min 的速率输送血管紧张素 II 以诱导腹主动脉瘤 (AAA)。盐酸坦索罗辛溶于生理盐水,以 0.1 mg/kg/天的剂量灌胃给药,持续 28 天。处死小鼠,收集主动脉组织进行组织病理学和分子分析[2] |
| 药代性质 (ADME/PK) |
盐酸坦索罗辛在人体内的口服生物利用度约为90%(食物对吸收无显著影响)。口服给药后0.5-1小时达到血浆峰浓度(Cmax)[1]
- 血浆蛋白结合率为99%,主要与白蛋白和α1-酸性糖蛋白结合[1] - 盐酸坦索罗辛在肝脏中通过细胞色素P450(CYP)酶代谢,主要通过CYP3A4和CYP2D6代谢为无活性代谢物[1] - 人体内的消除半衰期(t1/2)为10-14小时;约70%的剂量经尿液排出(其中10%为原药),20%经粪便排出[1] |
| 毒性/毒理 (Toxicokinetics/TK) |
人类常见不良反应包括头晕(6.3%)、体位性低血压(3.2%)、鼻炎(2.8%)和射精异常(2.1%);这些不良反应与剂量相关,通常为轻度至中度[1]
- 临床试验中未观察到明显的肝毒性或肾毒性;每日重复给药不会导致血浆浓度蓄积[1] - 动物毒性研究中,高剂量(治疗剂量的100倍)可引起低血压和心动过缓,但未报告致死性毒性[1] |
| 参考文献 | |
| 其他信息 |
盐酸坦索罗辛是由等摩尔量的坦索罗辛与氯化氢反应生成的盐酸盐。它是一种α-肾上腺素能拮抗剂和抗肿瘤药物。它含有坦索罗辛(1+)分子。它是ent-坦索罗辛盐酸盐的对映异构体。
盐酸坦索罗辛是坦索罗辛的盐酸盐,坦索罗辛是一种磺胺类衍生物,具有肾上腺素能拮抗活性。坦索罗辛选择性地与人前列腺和膀胱颈中的α1肾上腺素能受体结合并阻断其活性;阻断这些肾上腺素受体可使前列腺和膀胱颈的平滑肌松弛,从而改善尿流率。 盐酸坦索罗辛是一种磺胺类衍生物和肾上腺素能α1受体拮抗剂,用于缓解良性前列腺增生引起的尿路梗阻症状。 另见:坦索罗辛(注释已移至)。 盐酸坦索罗辛是一种选择性α1A/α1D肾上腺素能受体拮抗剂,获准用于治疗良性前列腺增生(BPH)相关的下尿路症状(LUTS)[1]。 - 其治疗作用是通过松弛前列腺间质平滑肌和膀胱颈,降低尿道阻力并改善尿流率来实现的[1]。 - 盐酸坦索罗辛通过抑制平滑肌细胞增殖/迁移、减轻炎症来减缓腹主动脉瘤(AAA)的生长。并保护主动脉壁细胞外基质[2] |
| 分子式 |
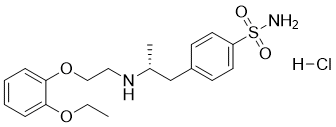
C20H28N2O5S.HCL
|
|
|---|---|---|
| 分子量 |
444.97
|
|
| 精确质量 |
444.148
|
|
| 元素分析 |
C, 55.00; H, 6.56; Cl, 8.54; N, 6.75; O, 15.42; S, 7.73
|
|
| CAS号 |
106463-17-6
|
|
| 相关CAS号 |
Tamsulosin;106133-20-4; Tamsulosin-d5 hydrochloride; Tamsulosin-d4 hydrochloride; 2518100-55-3
|
|
| PubChem CID |
5362376
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| 沸点 |
595.5ºC at 760 mmHg
|
|
| 熔点 |
228-230ºC
|
|
| 闪点 |
313.9ºC
|
|
| 蒸汽压 |
3.79E-14mmHg at 25°C
|
|
| LogP |
4.512
|
|
| tPSA |
108.26
|
|
| 氢键供体(HBD)数目 |
3
|
|
| 氢键受体(HBA)数目 |
7
|
|
| 可旋转键数目(RBC) |
11
|
|
| 重原子数目 |
29
|
|
| 分子复杂度/Complexity |
539
|
|
| 定义原子立体中心数目 |
1
|
|
| SMILES |
Cl[H].S(C1=C(C([H])=C([H])C(=C1[H])C([H])([H])[C@@]([H])(C([H])([H])[H])N([H])C([H])([H])C([H])([H])OC1=C([H])C([H])=C([H])C([H])=C1OC([H])([H])C([H])([H])[H])OC([H])([H])[H])(N([H])[H])(=O)=O
|
|
| InChi Key |
ZZIZZTHXZRDOFM-XFULWGLBSA-N
|
|
| InChi Code |
InChI=1S/C20H28N2O5S.ClH/c1-4-26-17-7-5-6-8-18(17)27-12-11-22-15(2)13-16-9-10-19(25-3)20(14-16)28(21,23)24;/h5-10,14-15,22H,4,11-13H2,1-3H3,(H2,21,23,24);1H/t15-;/m1./s1
|
|
| 化学名 |
5-[(2R)-2-[2-(2-ethoxyphenoxy)ethylamino]propyl]-2-methoxybenzenesulfonamide;hydrochloride
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|---|
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2473 mL | 11.2367 mL | 22.4734 mL | |
| 5 mM | 0.4495 mL | 2.2473 mL | 4.4947 mL | |
| 10 mM | 0.2247 mL | 1.1237 mL | 2.2473 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
Investigating Medication vs. Prostatic Urethral Lift: Assessment and Comparison of Therapies for BPH
CTID: NCT04987892
Phase: Phase 4 Status: Recruiting
Date: 2023-04-25
|
|
|