| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
- Trigonelline targets the Nrf2 transcription factor in pancreatic cancer cells, inhibiting its activity and downstream signaling [3]
Trigonelline (TG) is a naturally occurring alkaloid found in various edible plants, including fenugreek (Trigonella foenum-graecum), coffee, onions, peas, soybeans, cantaloupe, and corn. It exhibits multiple pharmacological activities, including antioxidant, anti-inflammatory, anti-diabetic, anti-hypercholesterolemic, and anti-carcinogenic properties. As a major component of coffee, trigonelline has been shown to protect cardiomyocytes from oxidative stress-induced apoptosis, inhibit Nrf2 activity in pancreatic cancer cells to enhance chemosensitivity, and exert differential effects on the skeletal system depending on metabolic conditions. The compound has demonstrated cardioprotective, chemosensitizing, and bone-modulating activities in various in vitro and in vivo models. |
|---|---|
| 体外研究 (In Vitro) |
此外,在 H9c2 细胞中 H2O2 诱导的氧化过程中,葫芦巴碱似乎可以调节 caspase-3 和 caspase-9 基因以及抗氧化基因 Bcl-2 和 Bcl-XL。根据流式细胞术数据,葫芦巴碱可显着降低 H2O2 诱导的胰腺 H9c2 细胞数量 [1]。
- 在过氧化氢(H₂O₂,200 μM)诱导凋亡的H9c2心肌细胞中:用葫芦巴碱(50 μM、100 μM、200 μM)预处理24小时,凋亡率分别较H₂O₂组降低28.3%、45.1%、62.7%。Western blot结果显示,抗凋亡蛋白Bcl-2表达量分别较H₂O₂组升高1.8倍、2.5倍、3.2倍,促凋亡蛋白Bax表达量分别降低至0.65倍、0.42倍、0.28倍;此外,葫芦巴碱可使H₂O₂诱导的活性氧(ROS)生成量减少35.6%-72.1%[1] - 在人胰腺癌细胞(PANC-1、MiaPaCa-2)中:葫芦巴碱(10 mM、20 mM)处理48小时,可抑制Nrf2核转位(较对照组减少40%-65%),并使Nrf2下游蛋白酶体基因(PSMA1、PSMB5、PSMD11)的mRNA表达量降低30%-50%。蛋白酶体糜蛋白酶样活性降低25%-40%,导致细胞凋亡率较对照组升高2.3-3.5倍(Annexin V/PI染色检测)[3] - 在抗菌及抗病毒实验中:葫芦巴碱对金黄色葡萄球菌( Staphylococcus aureus )和大肠杆菌( Escherichia coli )有抗菌活性,最低抑菌浓度(MIC)分别为256 μg/mL和512 μg/mL;对1型单纯疱疹病毒(HSV-1)有弱抗病毒活性,IC50为128 μg/mL[4] |
| 体内研究 (In Vivo) |
在链脲佐菌素诱导的糖尿病沉积物中,三甲硫灵会降低骨矿化并倾向于恶化骨机械特性。在用链脲佐菌素和烟酰胺处理的沉积物中,曲苯乃林显着提高了骨矿物质密度(BMD),并倾向于增加松质骨的强度。葫芦巴碱对链脲佐菌素产生的 BMD 有不同的影响。诱导系统增加了链脲佐菌素治疗引起的骨质疏松改变,当链脲佐菌素和烟酰胺一起服用时,会产生肠道非高血压的积极作用[2]。
- 在链脲佐菌素诱导的糖尿病大鼠(雄性,200-250 g)中:每日口服葫芦巴碱(50 mg/kg、100 mg/kg体重),持续8周,可改善糖尿病诱导的骨骼病变。100 mg/kg组大鼠腰椎骨密度(BMD)较糖尿病对照组升高18.2%,骨小梁厚度增加22.5%,骨小梁间距减少19.8%;血清成骨细胞标志物骨钙素水平升高35.6%,破骨细胞标志物抗酒石酸酸性磷酸酶(TRAP)水平降低28.3%[2] |
| 酶活实验 |
蛋白酶体活性测定(荧光法): 裂解细胞,与蛋白酶体底物Suc-LLVY-AMC在存在或不存在蛋白酶体抑制剂MG132的情况下孵育。测量荧光以确定蛋白酶体活性。活性以蛋白含量标准化。葫芦巴碱(0.1 μM)将基础蛋白酶体活性降低39-57%,将tBHQ诱导活性降低36-72%(依细胞系而异)[3]。
Caspase-3/7活性测定: 使用商业化试剂盒按说明书测定caspase-3/7活性以评估凋亡。活性以蛋白含量标准化。葫芦巴碱增强Panc1和Colo357细胞中TRAIL和依托泊苷诱导的caspase活化[3]。 |
| 细胞实验 |
- H9c2心肌细胞凋亡实验:将H9c2细胞接种于6孔板(5×10⁵个细胞/孔),培养24小时后分为5组:对照组(无处理)、H₂O₂组(200 μM)、H₂O₂+葫芦巴碱50 μM组、H₂O₂+葫芦巴碱100 μM组、H₂O₂+葫芦巴碱200 μM组。葫芦巴碱在H₂O₂处理前24小时加入,H₂O₂作用6小时后,用Annexin V-FITC/PI双染色结合流式细胞术检测凋亡细胞;Western blot检测时,裂解细胞提取蛋白,经SDS-PAGE分离、转膜后,用Bcl-2、Bax及β-肌动蛋白(β-actin)抗体孵育检测[1]
- 胰腺癌细胞实验:将PANC-1/MiaPaCa-2细胞接种(1×10⁶个细胞/孔),用葫芦巴碱(10 mM、20 mM)处理48小时。提取细胞核蛋白,通过Western blot检测Nrf2核转位;提取总RNA,逆转录为cDNA后,用qPCR检测PSMA1、PSMB5、PSMD11的mRNA水平;采用荧光底物法检测蛋白酶体活性,Annexin V/PI染色检测细胞凋亡[3] - 抗菌实验:将金黄色葡萄球菌和大肠杆菌在LB肉汤中培养至对数期,将葫芦巴碱在肉汤中进行系列稀释(32-1024 μg/mL),接种细菌(1×10⁵ CFU/mL)后37℃孵育24小时,无可见细菌生长的最低浓度即为MIC;HSV-1实验中,Vero细胞感染HSV-1后,用葫芦巴碱(32-512 μg/mL)处理,通过空斑实验检测病毒复制[4] 细胞活力测定(EZ-CYTOX/WST法): H9c2细胞(1×10⁵细胞/孔于96孔板)用不同浓度葫芦巴碱(25-150 μM,24小时)或H₂O₂(25-125 μM,6小时)处理。保护性研究中,细胞用葫芦巴碱(25-100 μM,48小时)预处理,然后暴露于H₂O₂(100 μM,4小时)。加入WST试剂,孵育2-4小时,在450 nm处测定吸光度[1]。 流式细胞术(Annexin V-FITC/PI染色): 收集细胞,重悬于结合缓冲液,用FITC-annexin V和碘化丙啶染色,通过流式细胞术分析以量化坏死、早期凋亡和晚期凋亡。葫芦巴碱(25-100 μM)与H₂O₂单独处理相比显著减少H₂O₂诱导的坏死和凋亡[1]。 Caspase-3定量(分光光度法): 细胞裂解液与DEVD-pNA底物在37°C孵育2小时,在400 nm处测定吸光度。葫芦巴碱降低H₂O₂诱导的caspase-3表达[1]。 抗氧化和脂质过氧化物测定: 使用商业化试剂盒按制造商方案测定SOD活性、过氧化氢酶活性、还原型谷胱甘肽含量和MDA水平。葫芦巴碱(75-100 μM)显著增加H₂O₂处理细胞中的SOD、过氧化氢酶和GSH水平,降低MDA含量[1]。 RT-PCR和qPCR: 提取总RNA,逆转录,对caspase-3、caspase-9、Bcl-2和Bcl-XL进行PCR。以β-actin为对照。葫芦巴碱在H₂O₂诱导的氧化应激期间上调Bcl-2和Bcl-XL,下调caspase-3和caspase-9表达[1]。 ARE-荧光素酶报告基因实验: 细胞转染ARE驱动的萤火虫荧光素酶载体和海肾荧光素酶对照。葫芦巴碱处理(0.01-10 μM,16小时)后,加或不加tBHQ(50 μM,8小时),测量荧光素酶活性。葫芦巴碱在所有四种细胞系中抑制ARE驱动的荧光素酶表达,最大效果在0.1-1 μM[3]。 Western blotting(核和胞质提取物): 制备核和胞质提取物或总细胞裂解液,SDS-PAGE分离,转移至PVDF膜,用抗Nrf2、Keap1、lamin A/C、Hsp90、微管蛋白、PARP1、s5a和α5的抗体检测。葫芦巴碱降低核Nrf2蛋白水平而不影响总Nrf2表达[3]。 siRNA敲低: 使用脂质转染试剂将细胞转染对照、Nrf2、Nrf1、s5a或α5 siRNA。48小时后,用葫芦巴碱和/或tBHQ处理细胞,然后评估凋亡。Nrf2或蛋白酶体基因敲低可消除葫芦巴碱的增敏效应[3]。 |
| 动物实验 |
糖尿病大鼠骨骼疾病研究:雄性Sprague-Dawley大鼠(200-250 g)经单次腹腔注射链脲佐菌素(60 mg/kg)诱导糖尿病。确认糖尿病(血糖>16.7 mmol/L)后,将大鼠分为3组(每组n=8):糖尿病对照组(生理盐水)、葫芦巴碱 50 mg/kg组和葫芦巴碱 100 mg/kg组。葫芦巴碱溶于生理盐水,每日灌胃一次,持续8周。另设正常对照组(非糖尿病,生理盐水灌胃)。每周测量大鼠体重和血糖。研究结束时,处死大鼠;采集血清样本以检测骨钙素和抗酒石酸酸性磷酸酶(TRAP)水平,并分析腰椎样本的骨密度(双能X射线吸收法)和骨小梁参数(微型CT)[2]
|
| 药代性质 (ADME/PK) |
吸收、分布和排泄
……在兔体内静脉注射葫芦巴碱后,其浓度-时间曲线分别符合单室和双室开放模型。静脉注射葫芦巴碱后的主要参数如下:T1/2α为10.8分钟,T1/2β为44.0分钟,K21为0.044分钟-1,K10为0.026分钟-1,K12为0.017分钟-1,AUC为931.0毫克·分钟/升。结论是,葫芦巴碱在兔体内吸收速率中等,消除速率快…… 代谢/代谢物 ……葫芦巴碱(N-甲基烟酸)是烟酰胺的代谢物。 |
| 毒性/毒理 (Toxicokinetics/TK) |
毒性概述
鉴别与用途:葫芦巴碱为固体。葫芦巴碱是一种具有潜在抗糖尿病活性的生物碱,在咖啡中含量丰富。它常用于生物化学研究。人体暴露与毒性:葫芦巴碱可促进人神经母细胞瘤SK-N-SH细胞的功能性神经突生长。动物研究:葫芦巴碱在大鼠中表现出显著的中枢神经系统(CNS)兴奋活性。葫芦巴碱对链脲佐菌素诱导的代谢紊乱大鼠的骨骼系统产生不同的影响,加剧链脲佐菌素处理大鼠的骨质疏松症,而对非高血糖(烟酰胺/链脲佐菌素处理)大鼠的骨骼产生有利影响。结果表明,在某些情况下,葫芦巴碱可能损害骨骼。在大鼠中,雌激素缺乏会导致胫骨干骺端骨矿化和力学性能恶化,以及骨转换标志物升高。给予葫芦巴碱对未切除卵巢的大鼠的上述指标无影响,但会恶化切除卵巢的大鼠松质骨的矿化和力学性能。葫芦巴碱对骨骼系统的不良影响取决于雌激素水平,且仅在雌激素缺乏大鼠的松质骨中观察到。细菌诱变试验(鼠伤寒沙门氏菌TA98、YG1024和YG1029菌株)结果表明,葫芦巴碱单独使用或与大多数单一氨基酸及氨基酸混合物联合使用均具有显著的诱变活性。然而,另一项研究发现,在沙门氏菌平板掺入试验和小鼠淋巴瘤L5178Y TK +/-试验中,该物质不具有致突变性。 相互作用 通过注射牛GABA(A)受体α(1)和β(1)亚基的cRNA,研究了咖啡成分和咖啡提取物对非洲爪蟾卵母细胞中表达的GABA(A)受体电反应的影响。咖啡水提取物呈剂量依赖性地抑制GABA诱发的反应,而咖啡的亲脂性乙醚提取物在低剂量(0.1-0.4 μL/mL)下略微增强了该反应,但在高剂量(0.5-0.8 μL/mL)下则表现出抑制作用。茶碱以非竞争性机制抑制该反应(K(i) = 0.55 mM),而可可碱和盐酸葫芦巴碱则以竞争性方式抑制该反应,K(i) 分别为 3.8 和 13 mM……/盐酸葫芦巴碱/ 非人类毒性值 大鼠口服 LD50 5 g/kg /取自表格/ 大鼠皮下注射 LD50 5 g/kg /取自表格/ |
| 参考文献 |
|
| 其他信息 |
N-甲基烟酸酯是一种亚胺甜菜碱,是N-甲基烟酸的共轭碱,由羧基去质子化产生。它是一种植物代谢物、食品成分和人体尿液代谢物。它是一种亚胺甜菜碱和生物碱,在功能上与烟酸酯相关,是N-甲基烟酸的共轭碱。
据报道,在苋属植物(Amaranthus hybridus)、苋菜(Alternanthera paronychioides)和其他有相关数据的生物体中均发现了葫芦巴碱。 另见:胡芦巴籽(部分)。 治疗用途 /EXPL THER/ 胡芦巴籽因其独特的汤料香味和作为印度咖喱的配料而闻名。传统上,人们将薯蓣种子浸泡后用于治疗糖尿病、咳嗽和胀气,促进乳汁分泌,并具有抗炎和壮阳功效。其用途受限于其难闻的气味和苦涩的味道,但可通过在浸泡液中加入薄荷叶来改善。薯蓣的抗糖尿病特性主要归功于半乳甘露聚糖、4-羟基异亮氨酸(4-OH-Ile)、薯蓣皂苷元和葫芦巴碱。临床研究表明,这些物质通过增加胰岛素分泌(4-OH-Ile)、降低胰岛素抵抗和促进胃肠道葡萄糖重吸收(半乳甘露聚糖)以及改善β细胞再生(葫芦巴碱)发挥直接的抗糖尿病作用。除了主要功效外,该草药还能改善血脂谱(4-羟基异亮氨酸、薯蓣皂苷元),并具有肾脏保护(4-羟基异亮氨酸、葫芦巴碱)、神经保护(葫芦巴碱)和抗氧化(薯蓣皂苷元、葫芦巴碱)作用。葫芦巴碱的降血糖疗效与格列本脲治疗相当,且优于西格列汀治疗。鉴于大量证据和与标准药物疗法相比的良好疗效,胡芦巴活性成分有望成为新型降血糖药物的来源。关键词:胡芦巴,Trigonella foenum-graecum,2型糖尿病,生物活性。 /EXPL THER/ 有证据表明,传统中药胡芦巴(Trigonella foenum-graecum L.)及其成分对预防和治疗糖尿病及中枢神经系统疾病有益。胡芦巴碱是胡芦巴的主要生物碱成分,其药理活性已得到比胡芦巴其他成分更为深入的研究,尤其是在糖尿病和中枢神经系统疾病方面。胡芦巴碱具有降血糖、降血脂、神经保护、抗偏头痛、镇静、改善记忆、抗菌、抗病毒和抗肿瘤活性,并已被证实能够减轻糖尿病听觉神经病变和血小板聚集。其作用机制可能涉及β细胞再生、胰岛素分泌、葡萄糖代谢相关酶的活性、活性氧的产生、轴突延伸以及神经元兴奋性。然而,仍需进一步研究胡芦巴碱的药理活性及其确切作用机制,并将其应用于临床实践。本综述旨在为读者提供胡芦巴碱药理作用的概览,尤其是在糖尿病、糖尿病并发症和中枢神经系统疾病方面的应用。此外,鉴于其药理价值和低毒性,本文简要回顾了已报道的葫芦巴碱在实验动物模型和人体中的不良反应,并讨论了葫芦巴碱的药代动力学。 - 葫芦巴碱是一种天然生物碱,主要存在于咖啡、胡芦巴和其他植物中[2] - 葫芦巴碱在H9c2细胞中的抗凋亡机制涉及降低氧化应激(降低活性氧)和调节Bcl-2/Bax凋亡通路[1] - 在胰腺癌细胞中,葫芦巴碱通过抑制Nrf2-蛋白酶体轴使细胞对凋亡更加敏感,因为Nrf2介导的蛋白酶体激活通常会促进癌细胞存活[3] - 葫芦巴碱通过平衡成骨细胞和……改善糖尿病引起的骨骼损伤。破骨细胞活性可能通过调节葡萄糖代谢和降低骨组织中的氧化应激来发挥作用[2] - 葫芦巴碱的抗菌活性弱于传统抗生素,但它可能具有作为辅助抗菌剂的潜力[4] |
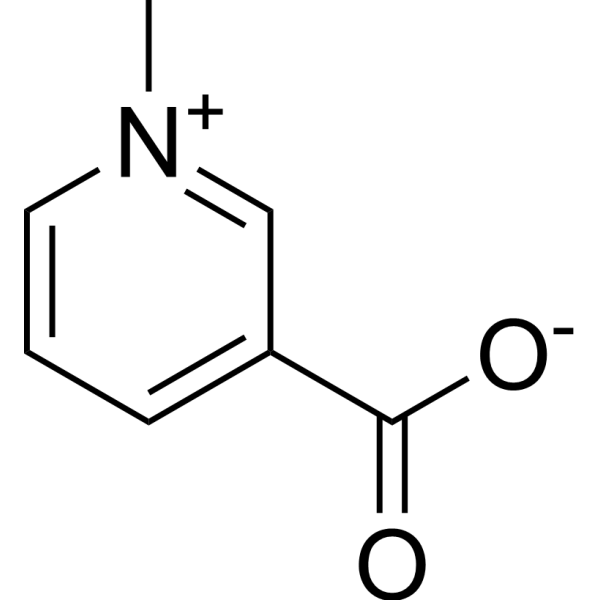
| 分子式 |
C7H7NO2
|
|---|---|
| 分子量 |
137.14
|
| 精确质量 |
137.047
|
| 元素分析 |
C, 61.31; H, 5.15; N, 10.21; O, 23.33
|
| CAS号 |
535-83-1
|
| 相关CAS号 |
Trigonelline chloride;6138-41-6; Trigonelline;535-83-1;Trigonelline-d3 chloride; 60388-20-7;
|
| PubChem CID |
5570
|
| 外观&性状 |
White to light yellow solid powder
|
| 密度 |
1.2528 (rough estimate)
|
| 沸点 |
251.96°C (rough estimate)
|
| 熔点 |
260ºC (dec.)
|
| 折射率 |
1.554
|
| LogP |
-3.91
|
| tPSA |
44.01
|
| 氢键供体(HBD)数目 |
0
|
| 氢键受体(HBA)数目 |
2
|
| 可旋转键数目(RBC) |
0
|
| 重原子数目 |
10
|
| 分子复杂度/Complexity |
130
|
| 定义原子立体中心数目 |
0
|
| SMILES |
C[N+]1=CC=CC(=C1)C(=O)[O-]
|
| InChi Key |
WWNNZCOKKKDOPX-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C7H7NO2/c1-8-4-2-3-6(5-8)7(9)10/h2-5H,1H3
|
| 化学名 |
1-methylpyridin-1-ium-3-carboxylate
|
| 别名 |
Trigonelline; 535-83-1; Gynesine; Caffearine; N-Methylnicotinate;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 本产品在运输和储存过程中需避光。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ~7.1 mg/mL (~52.1 mM)
H2O : ~100 mg/mL (~576.0 mM) |
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 0.71 mg/mL (5.18 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 7.1 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 0.71 mg/mL (5.18 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 7.1mg/mL澄清的DMSO储备液加入到900μL 20%SBE-β-CD生理盐水中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 0.71 mg/mL (5.18 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 7.2918 mL | 36.4591 mL | 72.9182 mL | |
| 5 mM | 1.4584 mL | 7.2918 mL | 14.5836 mL | |
| 10 mM | 0.7292 mL | 3.6459 mL | 7.2918 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。