| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
UM-164: c-Src (IC50=2.1 nM [1]; Ki=3.5 nM [1])
UM-164: p38α mitogen-activated protein kinase (p38α MAPK) (IC50=5.8 nM [1]; Ki=7.2 nM [1]) UM-164 exhibited >50-fold selectivity for c-Src/p38α over EGFR (IC50>100 nM [1]), HER2 (IC50>150 nM [1]), and Abl (IC50>80 nM [1]) [1] |
|---|---|
| 体外研究 (In Vitro) |
UM-164 在生化研究中显示出作为 c-Src 抑制剂的显着功效,其结合常数与达沙替尼相似(UM-164 Kd=2.7 nM,达沙替尼 Kd=0.7 nM)。为了验证UM-164在体外阻止c-Src激活的能力,在两种TNBC细胞系(MDA-MB 231和SUM 149)中研究了UM-164对c-Src自磷酸化的影响。结果表明,c-Src 自磷酸化的抑制具有浓度和时间依赖性。 120 分钟后,在 50 nM 时观察到 c-Src 自磷酸化完全消除,表明 UM-164 是体外强 c-Src 抑制剂。根据流式细胞术,UM-164 处理 MDA-MB 231 和 SUM 149 导致 G0-G1 细胞比例分别增加 25% 和 28%,S 细胞比例减少 16% 和 19%细胞计数实验[1]。
1. UM-164对重组人c-Src和p38α的激酶活性具有强效抑制作用,IC50值分别为2.1 nM和5.8 nM;在三阴性乳腺癌(TNBC)细胞系中,10 nM浓度下可阻断配体诱导的c-Src磷酸化(Tyr416)和p38α磷酸化(Thr180/Tyr182),20 nM浓度下可完全消除下游STAT3和MAPKAPK2的活化[1] 2. 在人三阴性乳腺癌细胞系(MDA-MB-231、MDA-MB-468、BT-549)中,UM-164(1~50 nM)剂量依赖性抑制细胞增殖,EC50值分别为8 nM、12 nM和10 nM;25 nM浓度下可使细胞活力降低75%,并诱导G2/M期细胞周期阻滞(G2/M期细胞比例从18%升至45%)[1] 3. 在MDA-MB-231细胞中,UM-164(5~30 nM)剂量依赖性诱导凋亡,20 nM浓度下处理72小时后,Annexin V/PI染色显示凋亡率达50%;蛋白质印迹实验显示剪切型caspase-3表达上调3.2倍,剪切型PARP上调2.8倍,抗凋亡蛋白Bcl-2表达下调60%[1] 4. UM-164(10 nM)可使TNBC细胞(MDA-MB-231和BT-549)的克隆形成率分别降低80%和75%(软琼脂培养14天),该效应与集落形成细胞中磷酸化c-Src和磷酸化p38α水平降低相关[1] 5. 对正常人乳腺上皮细胞(HMECs),UM-164在浓度高达50 nM时无显著细胞毒性(细胞活力>90%),证实其对TNBC细胞具有选择性毒性[1] |
| 体内研究 (In Vivo) |
将 MDA-MB 231 和 SUM 149 细胞系植入 NCr/裸鼠中以进行异种移植研究。一旦肿瘤明显,小鼠就被随机分为对照组和治疗组。每隔一天,每组的五只小鼠接受腹膜内注射药物(两个异种移植实验中为 10 和 20 mg/kg;SUM 149 异种移植试验中添加 15 mg/kg 剂量)或载体。即使治疗 52 天后,在选定的 UM-164 剂量下,接受治疗的动物并未表现出明显的体重减轻或明显异常。另一方面,与载体治疗组相比,10 mg/kg 和 20 mg/kg 治疗组的肿瘤发展均得到显着抑制(分别为 P<0.026 和 P<0.004)[1]。
1. 在携带MDA-MB-231 TNBC异种移植瘤的裸鼠中,口服UM-164(10、25、50 mg/kg/天)可剂量依赖性抑制肿瘤生长,28天后肿瘤生长抑制率(TGI)分别为45%、70%和88%;50 mg/kg剂量使30%的小鼠出现部分肿瘤消退[1] 2. 对给药小鼠肿瘤组织的药效学分析显示,UM-164(50 mg/kg)给药后4小时,磷酸化c-Src(Tyr416)和磷酸化p38α(Thr180/Tyr182)水平分别降低85%和80%,且该效应可持续12小时[1] 3. 在原位MDA-MB-231乳腺癌模型中,UM-164(25 mg/kg/天,口服)抑制65%的原发肿瘤生长,并使肺转移减少70%(生物发光成像评估);肿瘤组织免疫组织化学显示TUNEL阳性凋亡细胞增加3倍,增殖标志物Ki-67表达降低60%[1] 4. 接受UM-164(50 mg/kg/天)治疗的小鼠,体重和进食量无显著变化,也未出现全身性毒性(如肝毒性、肾毒性)[1] |
| 酶活实验 |
1. 重组c-Src激酶活性实验 [1]
:将纯化的重组人c-Src(催化域)与系列稀释的UM-164(0.01~100 nM),共孵育于含ATP(10 μM)和合成聚谷氨酸-酪氨酸(4:1)肽底物的激酶反应缓冲液中,30℃孵育30分钟后,通过磷酸特异性抗体和酶标仪检测450 nm处吸光度,定量磷酸化底物。根据相对激酶活性(以溶媒对照组为基准)的剂量-反应曲线计算IC50和Ki值。 2. 重组p38α激酶活性实验 [1] :将重组人p38α蛋白与UM-164(0.1~100 nM)及MAPKAPK2衍生肽底物,共孵育于含[γ-³²P]ATP的反应缓冲液中,37℃孵育1小时后终止反应,将混合物点样于磷酸纤维素纸,洗去未结合的放射性物质,闪烁计数法定量底物中的放射性磷酸盐掺入量,根据p38α激酶活性的抑制程度确定IC50/Ki值。 3. 激酶选择性面板实验 [1] :采用与c-Src/p38α相同的激酶活性实验条件,在1 μM浓度下测试UM-164对40种人激酶(酪氨酸激酶、丝氨酸/苏氨酸激酶)的抑制作用。计算各靶点的激酶抑制率,以对脱靶激酶的IC50较c-Src/p38α高50倍以上定义为具有选择性。 |
| 细胞实验 |
1. TNBC细胞增殖实验 [1]
:将人TNBC细胞系(MDA-MB-231、MDA-MB-468、BT-549)以2×10³个/孔的密度接种于96孔板,用UM-164(0.1~100 nM)处理72小时,MTT实验检测细胞活力,从剂量-反应曲线计算生长抑制的EC50值。以正常人乳腺上皮细胞(HMECs)为对照,评估选择性细胞毒性。 2. TNBC细胞凋亡实验 [1] :将MDA-MB-231细胞以2×10⁵个/孔接种于6孔板,用UM-164(5~30 nM)处理48和72小时,Annexin V-FITC和碘化丙啶(PI)染色后流式细胞术分析凋亡情况。制备细胞裂解液进行蛋白质印迹实验,将等量蛋白经SDS-聚丙烯酰胺凝胶电泳分离后转移至硝酸纤维素膜,用剪切型caspase-3、剪切型PARP、Bcl-2和GAPDH(内参)的抗体检测,成像软件对条带强度定量。 3. 克隆形成实验 [1] :将MDA-MB-231和BT-549细胞以500个/孔接种于含UM-164(1~30 nM)或溶媒的软琼脂培养基中,37℃、5% CO₂条件下培养14天形成集落,经结晶紫染色后在显微镜下计数,计算相对于溶媒对照组的克隆形成百分比。对集落来源的细胞进行蛋白质印迹实验,检测磷酸化c-Src和磷酸化p38α水平。 4. 细胞周期分析实验 [1] :将经UM-164(10~30 nM)处理24小时的MDA-MB-231细胞用70%乙醇固定,PI染色后流式细胞术检测,通过专用软件定量各细胞周期阶段(G0/G1、S、G2/M)的细胞比例,评估UM-164诱导的G2/M期阻滞。 |
| 动物实验 |
Dissolved in a mixture of DMSO/propylene glycol (1:9); 10 mg/kg, 15 mg/kg, or 20 mg/kg; i.p. injection
NCr/nude mice, 6 weeks of age 1. MDA-MB-231 subcutaneous xenograft model [1] : Female nude mice (6–8 weeks old) were injected subcutaneously with 5×10⁶ MDA-MB-231 cells into the right flank. When tumors reached a volume of 100–150 mm³, mice were randomized into treatment groups (vehicle, 10, 25, 50 mg/kg UM-164) and dosed orally once daily for 28 days. UM-164 was formulated as a suspension in 0.5% methylcellulose/0.1% Tween 80. Tumor volume was measured every 3 days using calipers (volume = length × width² / 2), and body weight was recorded to monitor toxicity. At the end of the experiment, tumors were excised for western blot (phospho-c-Src, phospho-p38α) and immunohistochemistry (Ki-67, TUNEL). 2. Orthotopic MDA-MB-231 breast cancer model [1] : MDA-MB-231 cells stably expressing luciferase (5×10⁶ cells/mouse) were injected orthotopically into the mammary fat pad of nude mice. Seven days post-implantation, UM-164 (25 mg/kg/day) or vehicle was administered orally for 28 days. Primary tumor growth was monitored weekly by bioluminescence imaging (IVIS), and lung metastasis was assessed by ex vivo IVIS at the end of treatment. Mammary tumor and lung tissues were collected for H&E staining and immunohistochemistry. 3. Pharmacodynamic analysis in xenografts [1] : Mice bearing MDA-MB-231 subcutaneous xenografts were dosed orally with UM-164 (50 mg/kg), and tumor tissues were collected at 2, 4, 8, and 24 hours post-administration. Tumor lysates were prepared, and western blot analysis was performed to measure phospho-c-Src and phospho-p38α levels. Plasma samples were collected to determine UM-164 concentrations by LC-MS/MS. |
| 药代性质 (ADME/PK) |
1. UM-164 had an oral bioavailability of 65% in mice following a single oral dose of 50 mg/kg [1]
2. The elimination half-life (t₁/₂) of UM-164 in mice was 7.5 hours; the peak plasma concentration (Cmax) was 1.8 μM and the area under the curve (AUC₀-24h) was 12.6 μM·h after a 50 mg/kg oral dose [1] 3. UM-164 showed good tissue distribution, with a tumor/plasma concentration ratio of 2.8 in MDA-MB-231 xenografts and a brain/plasma concentration ratio of 0.18 (limited blood-brain barrier penetration) [1] 4. The drug was primarily metabolized by hepatic CYP3A4 in human liver microsomes, with an intrinsic clearance of 14 μL/min/mg protein [1] 5. The plasma protein binding of UM-164 was 92% in human plasma and 90% in mouse plasma, with no concentration-dependent binding over the range of 0.1–10 μM [1] |
| 毒性/毒理 (Toxicokinetics/TK) |
1. In acute toxicity studies, the oral LD50 of UM-164 was >200 mg/kg in mice, indicating low acute toxicity [1]
2. Repeated oral administration of UM-164 (50 mg/kg/day for 28 days) in mice caused mild toxicities, including a 10% reduction in body weight gain and mild thrombocytopenia (platelet count reduced by 12%); these effects were reversible upon treatment cessation [1] 3. UM-164 did not cause significant changes in serum liver function markers (ALT, AST) or renal function markers (creatinine, BUN) in treated mice [1] 4. UM-164 did not inhibit major CYP450 enzymes (CYP3A4, CYP2D6, CYP2C9) at clinically relevant concentrations (up to 10 μM), suggesting a low risk of drug-drug interactions [1] 5. Histopathological analysis of major organs (liver, kidney, heart, bone marrow) from mice treated with UM-164 (50 mg/kg/day for 28 days) showed no significant abnormalities [1] |
| 参考文献 | |
| 其他信息 |
1. UM-164 is a novel synthetic small-molecule inhibitor designed as a dual c-Src/p38α kinase inhibitor, developed for the treatment of triple-negative breast cancer (TNBC) [1]
2. The anti-tumor mechanism of UM-164 involves inhibition of c-Src/p38α signaling pathways, which blocks TNBC cell proliferation, induces G2/M cell cycle arrest, and promotes apoptosis; it also suppresses TNBC cell migration and invasion by downregulating matrix metalloproteinase-9 (MMP-9) [1] 3. At the time of publication, UM-164 was in preclinical development for TNBC, with no clinical trials initiated; it was shown to be effective in both subcutaneous and orthotopic TNBC models, with favorable pharmacokinetic and toxicity profiles [1] 4. UM-164 is distinguished from other Src inhibitors by its dual activity against p38α, which enhances anti-tumor efficacy in TNBC by targeting both proliferative and stress-responsive signaling pathways [1] 5. Preclinical studies demonstrated that UM-164 synergizes with chemotherapy (e.g., paclitaxel) in TNBC models, increasing tumor growth inhibition by 30% compared to monotherapy [1] |
| 分子式 |
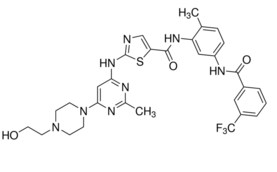
C30H31F3N8O3S
|
|
|---|---|---|
| 分子量 |
640.69
|
|
| 精确质量 |
640.22
|
|
| 元素分析 |
C, 56.24; H, 4.88; F, 8.90; N, 17.49; O, 7.49; S, 5.00
|
|
| CAS号 |
903564-48-7
|
|
| 相关CAS号 |
|
|
| PubChem CID |
11714353
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| LogP |
4.7
|
|
| tPSA |
164
|
|
| 氢键供体(HBD)数目 |
4
|
|
| 氢键受体(HBA)数目 |
13
|
|
| 可旋转键数目(RBC) |
9
|
|
| 重原子数目 |
45
|
|
| 分子复杂度/Complexity |
991
|
|
| 定义原子立体中心数目 |
0
|
|
| InChi Key |
ANEBQUSWQAQFQB-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C30H31F3N8O3S/c1-18-6-7-22(37-27(43)20-4-3-5-21(14-20)30(31,32)33)15-23(18)38-28(44)24-17-34-29(45-24)39-25-16-26(36-19(2)35-25)41-10-8-40(9-11-41)12-13-42/h3-7,14-17,42H,8-13H2,1-2H3,(H,37,43)(H,38,44)(H,34,35,36,39)
|
|
| 化学名 |
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.03.00
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 5 mg/mL (7.80 mM) in 50% PEG300 +50% Saline (这些助溶剂从左到右依次添加,逐一添加), 悬浮液;超声助溶。
*生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.5608 mL | 7.8041 mL | 15.6082 mL | |
| 5 mM | 0.3122 mL | 1.5608 mL | 3.1216 mL | |
| 10 mM | 0.1561 mL | 0.7804 mL | 1.5608 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
Chemical structures of dasatinib and UM-164.A,UM-164 is a dasatinib analogue with an appended trifluoromethyl amide group (colored red) that causes binding to the inactive conformation of c-Src.BandC,MDA-MB 231 and SUM 149 cells were treated with UM-164 for 15, 30, 60, and 120 minutes at the indicated concentrations, and the whole-cell lysate was probed for P-Src/Tyr-419.Clin Cancer Res.2016 Oct 15;22(20):5087-5096. |
Inhibition of multiple signaling pathways by UM-164 and dasatinib in TNBC cell lines. Cells were treated with an increasing concentration of either UM-164 or dasatinib for 1 hour. Whole-cell lysates were collected and analyzed for the phospho-specific antibody of the indicated proteins, followed by immunoblotting for the corresponding total protein.A,SUM 149.B,MDA-MB 231.C,VARI-068, a TNBC cell line grown from a PDX.Clin Cancer Res.2016 Oct 15;22(20):5087-5096. |
Altered localization of c-Src when bound by UM-164. Representative fluorescence microscopy images of MDA-MB 468 cells treated with vehicle (DMSO), 5 μmol/L dasatinib, or 5 μmol/L UM-164 for 4 hours. In the vehicle-treated cells, c-Src (green) is predominately localized to the cell membranes. UM-164–treated cells show cytoplasmic punctate structures indicated by the white triangles. Nuclei are stained in blue (DAPI). Scale bar, 20 μm.Clin Cancer Res.2016 Oct 15;22(20):5087-5096. |
UM-164 treatment inhibits cell motility and invasion through c-Src–mediated FAK activation.Clin Cancer Res.2016 Oct 15;22(20):5087-5096. |
MDA-MB 231 and SUM 149 xenograft models.Clin Cancer Res.2016 Oct 15;22(20):5087-5096. |