| 规格 | 价格 | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
| 靶点 |
JAK3 (IC50 = 145 nM)
|
|---|---|
| 体外研究 (In Vitro) |
Ritlecitinib 是一种强效的 JAK3 选择性抑制剂,抑制 JAK3 激酶活性的 IC50 为 33.1 nM,但对 JAK1、JAK2 和 TYK2 没有活性 (IC50>10,000 nM)。 Ritlecitinib 的 IC50 值分别为 244、340、407 和 266 nM,抑制 IL-2、IL-4、IL-7 和 IL-15 诱导的 STAT5 磷酸化。 Ritlecitinib 的 IC50 为 355 nM,还可以防止 IL-21 诱导的 STAT3 磷酸化。根据功能评估,Ritlecitinib 在 T 细胞分化测定中抑制 Th1 和 Th17 分化(在 Th1 环境下 5 天后通过 IFNγ 测量,在 Th17 设置下 6 天后通过 IL-17 产生测量)(IC50 值:分别为 30 nM 和 167 nM) )。此外,ritlecitinib 可抑制 Th1 和 Th17 功能,如在 PF-06651600 治疗前已经历分化和休眠的细胞中抑制 IFNγ 产生 (IC50=48 nM) 和 IL-17 产生 (IC50=269 nM) 所证明的[1] 。
|
| 体内研究 (In Vivo) |
Ritlecitinib 可减少大鼠佐剂诱导的关节炎 (AIA) 模型中的爪肿胀,未结合的 EC50 为 169 nM。在实验性自身免疫性脑脊髓炎(EAE)小鼠模型中,以 30 或 100 mg/kg 治疗性给药或以 20 或 60 mg/kg 预防性给药,可显着降低疾病的严重程度。 Ritlecitinib 在这两种啮齿动物模型中治疗炎症和自身免疫性疾病的有效性表明,仅选择性抑制 JAK3 可能足以改变人类疾病[1]。
|
| 药代性质 (ADME/PK) |
吸收、分布和排泄
利特替尼的AUC0-τ和Cmax在剂量高达200 mg时呈近似剂量比例增加,并在大约第4天达到稳态。利特替尼的绝对口服生物利用度约为64%,口服给药后1小时即可达到血浆峰浓度。食物对利特替尼的全身暴露量无临床意义上的影响。同时服用高脂餐和100 mg利特替尼胶囊可使Cmax降低32%,AUCinf增加11%。临床试验期间,利特替尼的给药不受进餐影响。 利特替尼主要通过尿液和粪便排泄。放射性标记的利特替尼分别约有66%和20%经尿液和粪便排出。约 4% 的利特西替尼剂量以原形药物形式经尿液排出。 利特西替尼的预计分布容积为 1.3 L/kg。 利特西替尼的预计血药清除率为 5.6 mL/min/kg。 代谢/代谢物 利特西替尼通过细胞色素 P450 (CYP) 和谷胱甘肽-S-转移酶 (GST) 代谢。参与利特西替尼代谢的 GST 酶包括胞质 GST A1/3、M1/3/5、P1、S1、T2、Z1 和微粒体 GST 1/2/3,参与此过程的 CYP 酶包括 CYP3A、CYP2C8、CYP1A2 和 CYP2C9。没有哪一种单一途径对利特西替尼总代谢的贡献超过 25%。 生物半衰期 利特西替尼的终末半衰期为 1.3 至 2.3 小时。 |
| 毒性/毒理 (Toxicokinetics/TK) |
肝毒性
在斑秃的上市前临床试验中,接受利特西替尼治疗的受试者中有1%至3%出现血清转氨酶升高,安慰剂组也观察到类似发生率。这些升高通常较轻且短暂,超过正常值上限5倍的患者不足1%。这些升高很少导致早期停药,即使不调整剂量,通常也能自行恢复正常。在斑秃和其他自身免疫性疾病的上市前研究中,未发现与利特西替尼相关的肝脏严重不良事件或临床上明显的肝损伤。自利特西替尼获批上市并广泛应用以来,尚未有与其使用相关的肝毒性报告发表。最后,利特西替尼是一种免疫调节剂,可能导致包括乙型肝炎在内的病毒感染复发。其他JAK抑制剂也曾与罕见的乙型肝炎复发病例有关,但这些病例通常无症状且具有自限性。利特西替尼治疗HBsAg阳性或抗-HBc阳性但HBsAg阴性的患者发生乙型肝炎复发的风险尚未明确。 可能性评分:E(不太可能引起特异性临床表现明显的肝损伤,但可能是乙型肝炎复发的潜在原因)。 肝毒性 在斑秃的上市前临床试验中,1%至3%的利特西替尼治疗组受试者出现血清转氨酶升高,安慰剂组受试者也观察到类似发生率。这些升高通常程度较轻且短暂,超过正常值上限 (ULN) 5 倍以上的患者不足 1%。这些升高很少导致早期停药,即使不调整剂量,通常也能自行恢复正常。在斑秃和其他自身免疫性疾病的上市前研究中,未发现与利特西替尼相关的肝脏严重不良事件或临床上明显的肝损伤病例。自利特西替尼获批上市并广泛应用以来,尚未有已发表的与其使用相关的肝毒性报告。 最后,利特西替尼是一种免疫调节剂,可能导致包括乙型肝炎在内的病毒感染复发。其他 JAK 抑制剂也曾与罕见的乙型肝炎复发病例有关,但这些病例通常无症状且具有自限性。对于接受利特西替尼治疗的HBsAg阳性或抗-HBc阳性但HBsAg阴性的患者,乙型肝炎病毒再激活的风险尚未明确。 可能性评分:E(不太可能引起特异性临床表现明显的肝损伤,但可能是乙型肝炎病毒再激活的潜在原因)。 妊娠和哺乳期用药 ◉ 哺乳期用药概述 目前尚无关于利特西替尼在哺乳期临床应用的信息。由于存在包括恶性肿瘤在内的严重不良反应风险,生产商建议在利特西替尼治疗期间以及末次给药后14小时内停止母乳喂养。 ◉ 对母乳喂养婴儿的影响 截至修订日期,未找到相关的已发表信息。 ◉ 对哺乳和母乳的影响 截至修订日期,未找到相关的已发表信息。 蛋白结合 利特西替尼与血浆蛋白的结合率为14%。 |
| 参考文献 |
[1]. Telliez JB, et al. Discovery of a JAK3-Selective Inhibitor: Functional Differentiation of JAK3-Selective Inhibition over pan-JAK or JAK1-Selective Inhibition. ACS Chem Biol. 2016 Dec 16;11(12):3442-3451.
[2]. Atli Thorarensen, et al. Pyrrolo[2,3-d]pyrimidinyl, pyrrolo[2,3-b]pyrazinyl and pyr-rolo[2,3-d]pyridinyl acrylamides. US20150158864A1. |
| 分子式 |
C15H19N5O
|
|---|---|
| 精确质量 |
285.158
|
| 元素分析 |
C, 63.14; H, 6.71; N, 24.54; O, 5.61
|
| CAS号 |
1792180-79-0
|
| 相关CAS号 |
Ritlecitinib;1792180-81-4; 2140301-97-7 (malonate) ; 2192215-81-7; 2489392-29-0
|
| PubChem CID |
118116220
|
| 外观&性状 |
White to light yellow solid
|
| LogP |
2.1
|
| tPSA |
73.9Ų
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
4
|
| 可旋转键数目(RBC) |
3
|
| 重原子数目 |
21
|
| 分子复杂度/Complexity |
402
|
| 定义原子立体中心数目 |
2
|
| SMILES |
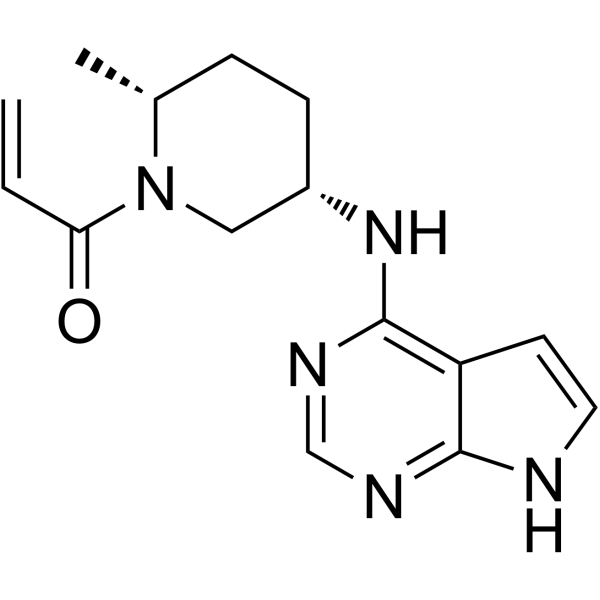
C[C@@H]1CC[C@@H](CN1C(=O)C=C)NC2=NC=NC3=C2C=CN3
|
| InChi Key |
CBRJPFGIXUFMTM-MNOVXSKESA-N
|
| InChi Code |
InChI=1S/C15H19N5O/c1-3-13(21)20-8-11(5-4-10(20)2)19-15-12-6-7-16-14(12)17-9-18-15/h3,6-7,9-11H,1,4-5,8H2,2H3,(H2,16,17,18,19)/t10-,11+/m1/s1
|
| 化学名 |
1-[(2R,5S)-2-methyl-5-(7H-pyrrolo[2,3-d]pyrimidin-4-ylamino)piperidin-1-yl]prop-2-en-1-one
|
| 别名 |
(2R,5S)-Ritlecitinib; 1792180-79-0; ent-Ritlecitinib; CHEMBL4065559; 1-((2R,5S)-5-((7H-Pyrrolo[2,3-d]pyrimidin-4-yl)amino)-2-methylpiperidin-1-yl)prop-2-en-1-one; .
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 本产品在运输和储存过程中需避光。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ~220 mg/mL (~771.01 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 5.5 mg/mL (19.28 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 55.0 mg/mL澄清的DMSO储备液加入到400 μL PEG300中,混匀;再向上述溶液中加入50 μL Tween-80,混匀;然后加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 5.5 mg/mL (19.28 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 55.0mg/mL澄清的DMSO储备液加入到900μL 20%SBE-β-CD生理盐水中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 5.5 mg/mL (19.28 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。