| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
G-CSF; IL-6; IL-8; TNFα; Neurokinin-1 receptor( IC50 = 0.1 nM)
Aprepitant decreases metabolic activity with an estimated IC50 value of 20 µM. Aprepitant causes G1 cell cycle arrest and inhibition of cell growth. In Nalm-6 cells, apropitant strongly induces apoptosis, which is mediated by activating caspase-3. Pro-apoptotic p53 target gene expression and p53 accumulation are induced by aprepitant (20 µM)[2]. In a dose-dependent manner, aprepitant (1, 5, 10 µM) inhibits HIV infection in MDM from HIV-negative individuals who are depressed and those who are not. Apreciant's IC50 value is approximately 5 μM, while its IC90 value is 10 μM[4]. |
|---|---|
| 体外研究 (In Vitro) |
阿瑞吡坦会降低代谢活性,估计 IC50 值为 20 µM。阿瑞吡坦可诱导细胞生长抑制和 G1 细胞周期停滞。阿瑞匹坦显着诱导 Nalm-6 细胞凋亡,该凋亡是通过 caspase-3 激活介导的。阿瑞吡坦 (20 µM) 诱导 p53 积累和促凋亡 p53 靶基因的表达[2]。阿瑞匹坦 (1, 5, 10 µM) 以剂量依赖性方式在体外抑制抑郁和非抑郁 HIV 阴性个体的 MDM 中的 HIV 感染。阿瑞吡坦的IC90值相当于10μM,IC50值约为5μM[4]。
Aprepitant 在体外以剂量依赖的方式抑制HIV-1 Bal对人单核细胞源性巨噬细胞(MDM)的感染,ED50约为5 µM。10 µM的浓度大约相当于IC90。抑郁状态、性别、年龄或种族对这种抑制作用没有影响。在为期一周的感染性实验过程中,培养基中aprepitant浓度的测定显示其几乎没有降解。 用P物质(SP)刺激健康供体的外周血单核细胞(PBMC)可上调促炎细胞因子和趋化因子(G-CSF、IL-6、IL-8、TNFα、MIP-1α和MCP-1)的产生。 Aprepitant(10 µM)阻断了这种SP诱导的上调作用。DMSO溶剂没有效果。 用SP刺激PBMC还会导致CD4+ T细胞上PD-1表达增加,该效应可被与aprepitant共孵育所阻断。 人单核细胞暴露于SP会导致可溶性CD163(sCD163)快速、时间和剂量依赖性的释放,该释放可被aprepitant预孵育所抑制。 |
| 体内研究 (In Vivo) |
阿瑞匹坦可防止由伯氏疏螺旋体体内 NHP 感染诱导的 NK-1R 表达增加。阿瑞吡坦治疗可防止伯氏疏螺旋体诱导的 NHP 脑脊液中 CCL2 蛋白水平增加。阿瑞吡坦治疗可防止伯氏疏螺旋体诱导的 NHP 背根神经节中 CCL2 和 CXCL13 mRNA 表达增加,防止伯氏疏螺旋体诱导的 NHP 脊髓中 CCL2、CXCL13、IL-17A 和 IL-6 mRNA 表达增加NHP。阿瑞吡坦治疗可减轻伯氏疏螺旋体感染引起的星形胶质细胞活性/数量减少[1]。阿瑞吡坦(10 mg/kg,腹腔注射)显着减弱小鼠中 AMPH 和可卡因产生的 CPP 表达和运动激活。阿瑞吡坦不会诱导显着的 CPP 或条件性位置厌恶或运动激活或抑制[3]。与未治疗的对照相比,阿瑞匹坦(125 毫克/天,口服)导致血浆病毒 RNA 水平降低 1 个对数级[4]。
在猕猴莱姆神经疏螺旋体病模型中,口服 阿瑞匹坦(平均剂量 28 ± 6 mg/kg/天)显著减轻了伯氏疏螺旋体感染诱导的神经炎症反应。 阿瑞匹坦 治疗阻止了感染后2周大脑皮层中NK-1R mRNA和蛋白表达的增加。 在一个实验系列中,阿瑞匹坦 治疗显著减轻了感染后2周脑脊液中CCL2蛋白水平的升高。 阿瑞匹坦 治疗显著减轻了中枢神经系统特定组织中炎症介质mRNA表达的增加:在背根神经节中,降低了感染后2周和4周的CXCL13和CCL2 mRNA表达;在颈段脊髓和硬脑膜中,降低了感染后2周的CXCL13、CCL2和IL-17A mRNA表达;在颈段脊髓中,降低了感染后4周的IL-6 mRNA表达。 通过免疫荧光检测,阿瑞匹坦 治疗减轻了感染后2周和4周额叶皮层中星形胶质细胞标志物胶质纤维酸性蛋白表达的减少。 在研究期间,未记录到任何动物出现可归因于 阿瑞匹坦 治疗的不良事件。[1] |
| 细胞实验 |
Appetitant 对 Nalm-6 细胞代谢活性的抑制作用通过活细胞对噻唑蓝四唑溴化物 (MTT) 的摄取来测量。以每孔 5000 个细胞的密度,将细胞接种到 96 孔板上。以 5、10、15、20 和 30 µM 阿瑞吡坦处理 24-36-48 小时后,将细胞与 100 µL MTT (0.5 mg/mL) 一起在 37°C 下孵育 3 小时。对照组由未经处理的细胞组成。用 100 μL DMSO 溶解沉淀的甲臜后,使用 ELISA 读数器在 578 nm 波长处测量光密度。
MDM中的HIV感染性实验: 从人血样本中分离单核细胞,并通过培养7天将其分化为巨噬细胞(MDM)。在感染HIV-1 Bal前,MDM用aprepitant(1、5、10 µM)或溶剂(0.001% DMSO)预孵育2小时。过夜孵育后,洗去未结合的病毒。含有aprepitant的培养基每周更换两次。感染后第7天,提取细胞RNA,并使用实时RT-PCR定量HIV gag mRNA的表达。 PBMC中细胞因子产生实验: 新鲜分离的人PBMC培养过夜。然后用P物质(10 µM)、aprepitant(10 µM)、两者或溶剂(DMSO)处理培养物。24小时后收集上清液,使用多重磁珠panel检测法测量细胞因子浓度。 PBMC中PD-1表达实验: PBMC用P物质(10 µM)和/或aprepitant(10 µM)处理。使用流式细胞术每天监测CD3+CD4+细胞上PD-1的表达,持续长达8天。 单核细胞释放sCD163实验: 新鲜分离的人单核细胞用不同剂量的P物质(经或不经aprepitant预孵育)处理。在不同时间点收集上清液,并通过ELISA测量可溶性CD163水平。 |
| 动物实验 |
十五只恒河猴接受麻醉后,经脊髓大池鞘内注射1×10⁸个活螺旋体。其余五只恒河猴未感染,抽取相应量的脑脊液后,给予1 mL RPMI 1640培养基。至少一份尸检组织样本的培养结果呈阳性,表明体内已建立伯氏疏螺旋体感染。第一组动物分为两组对照组(一组接受阿瑞匹坦治疗)、两组感染组(未接受治疗)和两组感染组(接受阿瑞匹坦治疗),所有动物均接受为期两周的研究。第二组动物包括五只感染但未治疗的动物、四只感染后接受阿瑞匹坦治疗的动物和三只对照组动物(其中一只接受阿瑞匹坦治疗),所有动物均接受为期四周的研究。每天两次口服阿匹坦,平均剂量为 28 ± 6 mg/kg,药物治疗在接种前两天开始。这些剂量与所选药物在非人灵长类动物中的典型兽医治疗方案一致,4 周的研究周期避免了神经病理的发生,而神经病理通常在伯氏疏螺旋体感染后 8 周出现。
本研究采用恒河猴(Macaca mulatta)莱姆病神经系统疾病模型。 将 2.5 至 5.5 岁的动物麻醉后,通过鞘内注射将 1 × 10^8 个活的伯氏疏螺旋体(溶于 RPMI 1640 培养基)接种到枕大池。 每日口服(po)阿瑞匹坦,平均剂量为 28 ± 6 mg/kg/天。 药物治疗在细菌接种前 2 天开始,持续 2 周或 4 周直至安乐死。 对照组包括未感染的动物(部分动物接受治疗)。 阿瑞匹坦)、感染但未治疗的动物,以及用阿瑞匹坦治疗的感染动物。 尸检时,采集包括脑皮层、背根神经节、脊髓和脑脊液在内的组织,用于后续分析(RT-PCR、免疫印迹、多重ELISA、免疫荧光)。[1] |
| 药代性质 (ADME/PK) |
吸收、分布和排泄
阿瑞吡坦的平均绝对口服生物利用度约为 60% 至 65%。 阿瑞吡坦主要通过代谢消除;不经肾脏排泄。阿瑞吡坦可分泌至大鼠乳汁中。尚不清楚该药物是否会分泌至人乳中。 70 L 表观血浆清除率=62-90 mL/min 代谢/代谢物 阿瑞吡坦主要通过 CYP3A4 介导的代谢,少量通过 CYP1A2 和 CYP2C19 介导的代谢。在人血浆中已鉴定出约七种阿瑞匹坦代谢物,它们均保留了较弱的药理活性。 阿瑞匹坦已知的人体代谢物包括5-氧代-1,4-二氢-1,2,4-三唑-3-甲醛、1-[3,5-双(三氟甲基)苯基]乙酮、5-{[(2S,3S)-3-(4-氟苯基)-2-羟基吗啉-4-基]甲基}-2,4-二氢-1,2,4-三唑-3-酮和(2R,3S)-2-((R)-1-(3,5-双(三氟甲基)苯基)乙氧基)-3-(4-氟苯基)吗啉。 生物半衰期 9-13小时 阿瑞匹坦是一种中枢性NK-1受体拮抗剂,可穿过血脑屏障。口服给药后可穿过血脑屏障。人体正电子发射断层扫描研究支持了这一观点,该研究表明其能够以口服剂量和血浆浓度依赖的方式占据脑NK-1受体。[1] |
| 毒性/毒理 (Toxicokinetics/TK) |
肝毒性
在阿瑞匹坦的预注册临床试验中,接受治疗的患者中有6%出现血清转氨酶升高,而接受癌症化疗的对照组患者中这一比例为4.3%。这些转氨酶升高是短暂的,程度为轻度至中度,且不伴有症状或黄疸。文献中尚未发表任何令人信服的、由阿瑞匹坦或福沙匹坦引起的临床明显肝损伤病例,因此,即使确实存在,使用这两种药物导致的严重肝损伤也极其罕见。 可能性评分:E(不太可能是临床明显肝损伤的原因)。 蛋白质结合 据报道,蛋白质结合率>95%。 在这项涉及恒河猴的特定研究中,接受阿瑞匹坦治疗的动物均未记录到任何可归因于该治疗的不良事件。 该研究引用了其他研究(在人和小鼠中),这些研究均未发现使用阿瑞匹坦存在安全问题。[1] |
| 参考文献 |
|
| 其他信息 |
药效学
阿瑞匹坦是一种止吐药,属于P物质/神经激肽1 (NK1) 受体拮抗剂,与其他止吐药联合使用,适用于预防高致吐性癌症化疗初始疗程和重复疗程引起的急性及迟发性恶心呕吐。阿瑞匹坦是人P物质/神经激肽1 (NK1) 受体的选择性高亲和力拮抗剂。阿瑞匹坦对血清素 (5-HT3)、多巴胺和皮质类固醇受体几乎没有亲和力,而这些受体正是现有化疗引起的恶心呕吐 (CI NV) 疗法的靶点。 阿瑞匹坦是一种特异性的非肽类神经激肽-1受体 (NK-1R) 拮抗剂。 它是一种获得美国食品药品监督管理局 (FDA) 批准的药物,目前(与其前药福沙匹坦一起)在临床上用作化疗后止吐药。 本研究探索了阿瑞匹坦作为辅助疗法,用于限制细菌性中枢神经系统感染(特别是莱姆病)相关的神经炎症损伤的潜在用途。 其作用机制涉及拮抗P物质 (SP)/NK-1R相互作用,这种相互作用与感染期间中枢神经系统炎症反应的加剧有关。 在非人灵长类动物模型中,阿瑞匹坦(apprepitant)能减轻伯氏疏螺旋体(B. burgdorferi)引起的神经炎症,这支持对其治疗神经炎症性疾病的潜力进行进一步研究。[1] |
| 分子式 |
C23H21F7N4O3
|
|---|---|
| 分子量 |
534.426669836044
|
| 精确质量 |
534.15
|
| 元素分析 |
C, 51.69; H, 3.96; F, 24.88; N, 10.48; O, 8.98
|
| CAS号 |
170729-80-3
|
| PubChem CID |
135413536
|
| 外观&性状 |
White solid powder
|
| 密度 |
1.5±0.1 g/cm3
|
| 熔点 |
75-76 °C(lit.)
|
| 折射率 |
1.564
|
| LogP |
4.23
|
| tPSA |
83.24
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
12
|
| 可旋转键数目(RBC) |
6
|
| 重原子数目 |
37
|
| 分子复杂度/Complexity |
810
|
| 定义原子立体中心数目 |
3
|
| SMILES |
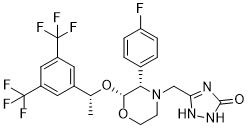
O=C1NC(CN2[C@H]([C@@H](O[C@@H](C3=CC(C(F)(F)F)=CC(C(F)(F)F)=C3)C)OCC2)C4=CC=C(F)C=C4)=NN1
|
| InChi Key |
ATALOFNDEOCMKK-OITMNORJSA-N
|
| InChi Code |
InChI=1S/C23H21F7N4O3/c1-12(14-8-15(22(25,26)27)10-16(9-14)23(28,29)30)37-20-19(13-2-4-17(24)5-3-13)34(6-7-36-20)11-18-31-21(35)33-32-18/h2-5,8-10,12,19-20H,6-7,11H2,1H3,(H2,31,32,33,35)/t12-,19+,20-/m1/s1
|
| 化学名 |
3-[[(2R,3S)-2-[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)morpholin-4-yl]methyl]-1,4-dihydro-1,2,4-triazol-5-one
|
| 别名 |
L754030; L-754030; L 754030; MK0869; MK 0869; MK-0869; ONO7436; ONO-7436; ONO 7436; Emend; Aponvie; Cinvanti; L-754030; Aprepitant; trade name: Emend
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 本产品在运输和储存过程中需避光。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO: ~107 mg/mL (~200.2 mM)
Ethanol: ~15 mg/mL |
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (4.68 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (4.68 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL 澄清 DMSO 储备液加入到 900 μL 玉米油中并混合均匀。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8712 mL | 9.3558 mL | 18.7115 mL | |
| 5 mM | 0.3742 mL | 1.8712 mL | 3.7423 mL | |
| 10 mM | 0.1871 mL | 0.9356 mL | 1.8712 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
Comparison of Adding EMEND to PONV/PDNV Treatment Regimen
CTID: NCT01186029
Phase: N/A Status: Withdrawn
Date: 2024-08-30
Aprepitant treatment preventsB. burgdorferi-induced increases in CCL2, CXCL13, IL-17A, and IL-6 mRNA expression in the spinal cord of NHPs.J Neuroinflammation.2017 Feb 15;14(1):37. |
|---|
Aprepitant treatment preventsB. burgdorferi-induced increases in CCL2 and CXCL13 mRNA expression in the dorsal root ganglia of NHPs. Rhesus macaques were uninfected (n = 5 animals) or infected intrathecally withB. burgdorferi(Bb, 1 × 108bacteria;n = 15), and were untreated (n = 7) or treated with aprepitant (Ap,n = 8) for 2 (a) or 4 (b,c) weeks.The level of expression of mRNA encoding CXCL13 (a,b) and CCL2 (c) in the dorsal root ganglia was determined by qPCR. Data is expressed as the mean ± SD and asterisks indicate statistically significant differences between the untreated and treated groups (p J Neuroinflammation.2017 Feb 15;14(1):37. |
Aprepitant treatment preventsB. burgdorferi-induced increases in CCL2 protein levels in the CSF of NHPs.Rhesus macaques were uninfected (n = 5 animals) or infected intrathecally withB. burgdorferi(Bb, 1 × 108bacteria;n = 15) and were untreated (n = 7) or treated with aprepitant (Ap,n = 8) for 2 (a) or 4 (b) weeks.
Aprepitant treatment attenuatesB. burgdorferiinfection-induced reductions in astrocyte activity/numbers. Rhesus macaques were uninfected orx infected intrathecally withB. burgdorferi(Bb, 1 × 108bacteria) and were untreated or treated with aprepitant (Ap) for 2 or 4weeks prior to euthanasia.J Neuroinflammation.2017 Feb 15;14(1):37. |