| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1g |
|
||
| Other Sizes |
|

| 靶点 |
β-lactam
|
|---|---|
| 体外研究 (In Vitro) |
克拉维酸和氨苄西林表现出协同抗菌活性(针对产生 β-内酰胺酶的微生物)[2]。
Ab11 和 Ab51 菌株在 MIC 为 2–8 μg/mL 时被克拉维酸抑制[3]。 |
| 体内研究 (In Vivo) |
克拉维酸(13 mg/kg,腹腔注射)可降低鲍曼不动杆菌感染的 C57BL/6 小鼠肺炎模型的肺部细菌负荷[3]。克拉维酸(13 mg/kg,ip)在 Ab51 感染的 C57BL/6 小鼠肺炎模型中的 t1/2 为 6.69 h,AUC 为 4.03 mg·h/L[3]。
角叉菜胶 (HY-125474) 引起的爪水肿在用克拉维酸(100–300 mg/kg,腹腔注射)治疗时表现出抗炎作用[4]。 |
| 动物实验 |
药代动力学研究:免疫功能正常的C57BL/6小鼠单次腹腔注射克拉维酸(13 mg/kg)。分别于给药后5、10、15、30、60、90、120和240分钟,从每组麻醉小鼠的眶周静脉丛采集血液样本。采用肺炎克雷伯菌ATCC 29665作为指示菌,通过生物测定法测定血清药物浓度。计算药代动力学参数(Cmax、AUC、t₁/₂、T>MIC)。 [3] * **疗效研究(肺炎模型):** 将C57BL/6小鼠麻醉后,经气管内接种50 μL细菌悬液(鲍曼不动杆菌Ab11或Ab51菌株,浓度约为8.6 log₁₀ CFU/mL),该悬液与10%猪黏蛋白按1:1比例混合。接种4小时后开始治疗。小鼠随机分为三组(每组每株15只):未治疗对照组、克拉维酸组(13 mg/kg,腹腔注射)和亚胺培南组(30 mg/kg,肌肉注射)。克拉维酸的给药间隔,Ab11菌株为2.5小时,Ab51菌株为2小时,以维持T>MIC在给药间隔的40%以内。观察动物24小时。实验结束后或死亡后立即采集血液和肺组织样本进行定量培养。结果以每克肺组织中菌落形成单位 (CFU) 的对数 (log₁₀ CFU/g) 和无菌血培养的频率表示。[3]
* **毒性对照:** 另取 10 只未感染小鼠,给予相同剂量的克拉维酸,以评估其毒性。未观察到毒性。[3] |
| 药代性质 (ADME/PK) |
吸收、分布和排泄
口服克拉维酸后,在胃肠道内吸收良好。对四名受试者服用放射性标记的克拉维酸后,最低吸收率为73%,平均绝对生物利用度为64%。在一项药代动力学研究中,8名健康志愿者的平均Cmax为2.098 ± 0.441微克/毫升。该研究报告的平均Tmax为1.042 ± 0.80小时。另一项药代动力学研究报告Tmax为40-120分钟。 服用后6小时内,约40%至65%的克拉维酸以原药形式经尿液排出。克拉维酸的代谢产物主要通过尿液、粪便和呼出气体中的二氧化碳排出。克拉维酸可通过肾脏和非肾脏途径清除。约17%的放射性标记克拉维酸剂量经呼出气体排出,8%的剂量经粪便排出。一项对4名健康志愿者进行的研究表明,给予放射性标记的克拉维酸后,其分布容积为12升。克拉维酸分布于各种组织和组织间液中。在胆囊、腹部、皮肤、脂肪和肌肉组织中均检测到具有临床意义的浓度。胆汁、脓液、滑液和腹膜液中也检测到治疗浓度的克拉维酸。动物研究表明,克拉维酸可通过胎盘。一项对4名健康志愿者进行的药代动力学研究表明,给予放射性标记的克拉维酸后,其清除率为0.21升/分钟。另一项资料显示,克拉维酸的平均清除率为 12.20 升/小时/70 公斤。肾功能衰竭患者可能需要调整剂量。 代谢/代谢物 克拉维酸主要代谢生成两种代谢物:2,5-二氢-4-(2-羟乙基)-5-氧代-1H-吡咯-3-羧酸和 1-氨基-4-羟基-丁-2-酮。一项药代动力学研究发现,第一种代谢物占剂量的 15.6%,第二种代谢物占剂量的 8.8%。 生物半衰期 据报道,克拉维酸的半衰期与阿莫西林相似,持续 45-90 分钟。对 4 名健康志愿者进行的放射性标记克拉维酸研究确定其半衰期为 0.8 小时。 |
| 毒性/毒理 (Toxicokinetics/TK) |
蛋白质结合
阿莫西林的血浆蛋白结合率约为25%。 |
| 参考文献 | |
| 其他信息 |
克拉维酸是从链霉菌(Streptomyces clavuligerus)中分离得到的抗生素。它是一种自杀性β-内酰胺酶抑制剂,可抑制细菌β-内酰胺酶的活性。克拉维酸具有抗菌、抗焦虑和抑制EC 3.5.2.6(β-内酰胺酶)等多种作用。它是克拉维酸的结合物。
克拉维酸是一种β-内酰胺酶抑制剂,常与阿莫西林或替卡西林联合使用,通过阻止β-内酰胺酶降解抗生素,从而扩大其对敏感细菌感染的抗菌谱,以对抗抗生素耐药性。克拉维酸来源于链霉菌(Streptomyces clavuligerus)。当与阿莫西林联合使用时,克拉维酸常被称为奥格门汀(Augmentin)、阿莫西林克拉维酸钾(Co-Amoxiclav)或克拉维林(Clavulin)。 克拉维酸是一种β-内酰胺酶抑制剂。克拉维酸的作用机制是作为β-内酰胺酶抑制剂。 已有报道称,克拉维酸存在于卡特利亚链霉菌(Streptomyces cattleya)和棒状链霉菌(Streptomyces clavuligerus)中,并有相关数据。 克拉维酸是一种从链霉菌中分离得到的半合成β-内酰胺酶抑制剂。克拉维酸含有一个β-内酰胺环,能够与β-内酰胺酶活性位点或其附近区域牢固结合,从而抑制酶活性。这可以保护其他β-内酰胺类抗生素免受β-内酰胺酶的催化,从而增强其抗菌作用。该药物通常与对β-内酰胺酶敏感的抗生素(如青霉素和头孢菌素)联合使用,用于治疗由产生β-内酰胺酶的微生物引起的感染。 棒状链霉菌(Streptomyces clavuligerus)是一种由放线菌产生的β-内酰胺类抗生素。它是一种细菌β-内酰胺酶的自杀性抑制剂。单独使用时,它对大多数微生物的抗菌活性较弱,但与其他β-内酰胺类抗生素联合使用时,可防止微生物β-内酰胺酶使抗生素失活。 药物适应症 克拉维酸与其他抗生素联合使用,可预防耐药菌株的产生,并增强其抗菌治疗效果。以下疾病,当病原体产生β-内酰胺酶时,可使用阿莫西林/克拉维酸或替卡西林/克拉维酸联合治疗:由流感嗜血杆菌和卡他莫拉菌引起的急性中耳炎;由流感嗜血杆菌和卡他莫拉菌引起的鼻窦炎;由流感嗜血杆菌、金黄色葡萄球菌、克雷伯菌属和卡他莫拉菌引起的下呼吸道感染;由金黄色葡萄球菌、大肠杆菌和克雷伯菌属引起的皮肤及皮肤软组织感染;由大肠杆菌、克雷伯菌属、肠杆菌属、粘质沙雷氏菌或金黄色葡萄球菌引起的泌尿道感染;由多种细菌引起的妇科感染,包括产黑色素假单胞菌、肠杆菌属、大肠杆菌属和克雷伯菌属。金黄色葡萄球菌、表皮葡萄球菌引起的败血症;由多种细菌(包括克雷伯菌属、大肠杆菌属、金黄色葡萄球菌或假单胞菌属)引起的败血症;金黄色葡萄球菌引起的骨和关节感染;大肠杆菌、肺炎克雷伯菌或脆弱拟杆菌群引起的腹腔内感染。关于药敏性的说明 需要注意的是,本品仅适用于已确诊或高度怀疑由敏感菌引起的感染。应尽可能进行细菌培养和药敏试验,并以此作为是否使用该抗生素的依据。如果在微生物学检测中未检测到β-内酰胺酶的产生,则不应使用克拉维酸。当无法进行这些检测时,可参考局部感染模式和药敏试验结果来判断是否适合使用克拉维酸。替卡西林联合克拉维酸钾在混合感染中显示出特别的疗效,可作为经验性治疗,用于在确定致病菌药敏性之前进行治疗。替卡西林-克拉维酸钾组合可能成为一种有效的单药抗生素疗法,用于治疗通常需要多种药物联合治疗的感染。 作用机制 克拉维酸钾的结构中含有一个β-内酰胺环,该环能与β-内酰胺酶不可逆地结合,从而阻止β-内酰胺酶灭活某些β-内酰胺类抗生素,有效治疗对β-内酰胺类抗生素敏感的革兰氏阳性菌和革兰氏阴性菌感染。 |
| 精确质量 |
199.048
|
|---|---|
| CAS号 |
58001-44-8
|
| 相关CAS号 |
Potassium clavulanate cellulose;Clavulanate lithium;61177-44-4;Clavulanate potassium;61177-45-5
|
| PubChem CID |
5280980
|
| 外观&性状 |
Off-white to light yellow solid powder
|
| 密度 |
1.7±0.1 g/cm3
|
| 沸点 |
545.8±50.0 °C at 760 mmHg
|
| 熔点 |
117.5-118
117.5 - 118 °C |
| 闪点 |
283.9±30.1 °C
|
| 蒸汽压 |
0.0±3.3 mmHg at 25°C
|
| 折射率 |
1.644
|
| LogP |
-1.98
|
| tPSA |
87.07
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
5
|
| 可旋转键数目(RBC) |
2
|
| 重原子数目 |
14
|
| 分子复杂度/Complexity |
324
|
| 定义原子立体中心数目 |
2
|
| SMILES |
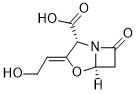
C(=C/1\[C@H](C(=O)O)N2C(=O)C[C@H]2O1)/CO
|
| InChi Key |
HZZVJAQRINQKSD-PBFISZAISA-N
|
| InChi Code |
InChI=1S/C8H9NO5/c10-2-1-4-7(8(12)13)9-5(11)3-6(9)14-4/h1,6-7,10H,2-3H2,(H,12,13)/b4-1-/t6-,7-/m1/s1
|
| 化学名 |
(2R,3Z,5R)-3-(2-hydroxyethylidene)-7-oxo-4-oxa-1-azabicyclo[3.2.0]heptane-2-carboxylic acid
|
| 别名 |
Clavulanate; Acide clavulanique; Acido clavulanico; Clavulansaeure; Antibiotic MM 14151; acidum clavulanicum;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: (1). 本产品在运输和储存过程中需避光。 (2). 请将本产品存放在密封且受保护的环境中(例如氮气保护),避免吸湿/受潮。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ~13.89 mg/mL (~69.74 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (12.55 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (12.55 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: 10% DMSO+90% (20% SBE-β-CD in Saline): ≥ 2.5 mg/mL (12.55 mM) 配方 4 中的溶解度: 12.5 mg/mL (62.76 mM) in PBS (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶 (<60°C). 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT02563769 | COMPLETEDWITH RESULTS | Drug: Clavulanic acid Drug: Intravenous cocaine Drug: Placebo |
Cocaine Abuse Cocaine Addiction Cocaine Dependence Cocaine-Related Disorders |
Temple University | 2016-10-24 | Phase 1 |
| NCT00603317 | COMPLETED | Drug: Firstly : Amoxicillin-Clavulanic acid and secondly : Placebo Drug: Firstly : Placebo and secondly : Amoxicillin-Clavulanic acid |
Atrial Fibrillation Deep Venous Thrombosis Oral Anticoagulation Pulmonary Embolism |
Assistance Publique - Hôpitaux de Paris | 2008-03 | Phase 4 |
| NCT05562349 | ACTIVE, NOT RECRUITING | Drug: Clavulanic Acid Only Product Drug: Placebo |
Cocaine Dependence | Temple University | 2023-05-03 | Phase 2 |
| NCT04411914 | COMPLETEDWITH RESULTS | Drug: Clavulanic Acid Other: Placebo |
Cocaine Dependence | Temple University | 2020-09-01 | Phase 1 |
| NCT01772238 | COMPLETED | Drug: 400 mg Amoxicillin + 57 mg Clavulanic Acid/ 5 ml Drug: 400 mg Amoxicillin + 57 mg Clavulanic Acid/ 5 ml |
Infections, Respiratory Tract | GlaxoSmithKline | 2011-03-22 | Phase 1 |
|
|
|
|