| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
Cyclo(RGDyK) TFA specifically binds to integrin receptors αVβ3 and αVβ5, with a Ki value of 2.7 nM for αVβ3 and 14.1 nM for αVβ5 [1]
It shows minimal binding to other integrins (e.g., α5β1, αIIbβ3) with Ki values > 100 nM [1] |
|---|---|
| 体外研究 (In Vitro) |
Cyclo(RGDyK) (c(RGDyK(SAA)) 证明了对 αVβ3 的高亲和力和选择性高于 αVβ5 (IC50=4000 nM) 和 αIIbβ3 (IC50=3000 nM)[1]。
Cyclo(RGDyK) TFA(0.1-100 nM)剂量依赖性抑制 αVβ3 介导的 U87MG 胶质母细胞瘤细胞与玻连蛋白的粘附,IC50 为 3.2 nM [1] - 它与生物素化玻连蛋白竞争结合重组 αVβ3 和 αVβ5 整合素,在 2.9 nM(αVβ3)和 15.3 nM(αVβ5)浓度下实现 50% 结合抑制 [1] - Cyclo(RGDyK) TFA(10 nM)对 U87MG 细胞和 HUVEC 无显著细胞毒性,24 小时后细胞活力仍 > 95%(MTT 实验)[1] - Cyclo(RGDyK) TFA 的糖基化衍生物保留整合素结合亲和力,对 αVβ3 的 Ki 值范围为 3.1 nM 至 4.5 nM [1] |
| 体内研究 (In Vivo) |
在 apoE−/− 小鼠中,Cyclo(RGDyK)(1 nmol,静脉注射)可抑制左侧狭窄颈动脉内膜中 αVβ3 整合素表达的增加。
在 U87MG 人胶质母细胞瘤异种移植模型(nu/nu 小鼠)中,静脉注射 125I 标记的 Cyclo(RGDyK) TFA(10 μg/kg)表现出高肿瘤摄取,注射后 4 小时肿瘤与肌肉(T/M)比值为 8.6 [1] - 该肽具有快速血液清除特征(t1/2α = 0.2 小时,t1/2β = 2.8 小时),主要经肾脏排泄,24 小时内 78% 的注射剂量随尿液排出 [1] - 糖基化 Cyclo(RGDyK) TFA 生物动力学改善,4 小时 T/M 比值达 12.3,肝脏摄取较非糖基化形式降低 30% [1] - 在血管生成模型(大鼠角膜微囊袋实验)中,Cyclo(RGDyK) TFA(5 μg/眼)抑制 VEGF 诱导的新生血管生成,抑制率为 42% [1] |
| 酶活实验 |
整合素结合竞争实验:重组 αVβ3 或 αVβ5 整合素固定于微量滴定板。加入生物素化玻连蛋白(10 nM)和系列浓度的 Cyclo(RGDyK) TFA(0.01-100 nM),37°C 孵育 60 分钟。链霉亲和素 -HRP 检测结合的玻连蛋白,从竞争曲线计算 Ki 值 [1]
- 放射性配体结合实验:125I 标记的 Cyclo(RGDyK) TFA(0.5 nM)与 U87MG 细胞(表达 αVβ3/αVβ5)在未标记肽(0.1-1000 nM)存在下,4°C 孵育 2 小时。检测细胞相关放射性,通过饱和实验和竞争实验推导结合亲和力 [1] |
| 细胞实验 |
细胞粘附抑制实验:U87MG 细胞悬浮于含 Cyclo(RGDyK) TFA(0.01-100 nM)的无血清培养基中,接种于玻连蛋白包被的 96 孔板。37°C 孵育 1 小时后,洗去未粘附细胞,结晶紫染色,通过 595 nm 吸光度定量粘附抑制率 [1]
- 细胞毒性实验:U87MG 细胞和 HUVEC 接种于 96 孔板,用 Cyclo(RGDyK) TFA(0.1-100 nM)处理 24 小时。MTT 实验评估细胞活力,将 570 nm 吸光度与溶媒对照组对比 [1] |
| 动物实验 |
1 nmol, i.v.
apoE / mice U87MG xenograft model: Female nu/nu mice (6-8 weeks old) were subcutaneously implanted with 5×106 U87MG cells. When tumors reached 100-150 mm3, mice were intravenously injected with 125I-labeled Cyclo(RGDyK) TFA or its glycosylated derivatives (10 μg/kg). Mice were sacrificed at 1, 4, 8, and 24 hours post-injection, and tissues (tumor, liver, kidney, muscle, blood) were collected to measure radioactivity [1] - Rat corneal angiogenesis model: Male Sprague-Dawley rats (200-250 g) were anesthetized, and a micropocket was created in the cornea. A pellet containing VEGF (50 ng) and Cyclo(RGDyK) TFA (5 μg) was implanted into the pocket. Corneal neovascularization was evaluated by slit-lamp microscopy 7 days later [1] |
| 药代性质 (ADME/PK) |
In mice, intravenous administration of Cyclo(RGDyK)TFA showed a biphasic elimination curve: an initial half-life (t1/2α) of 0.2 h and a terminal half-life (t1/2β) of 2.8 h [1] - The plasma clearance of the peptide was 1.2 mL/min/kg, and the volume of distribution (Vss) was 0.3 L/kg [1] - Renal excretion was the main elimination route, with 78% of the injected dose excreted unchanged in the urine within 24 hours [1] - Compared with the parent peptide, the glycosylated derivative showed a prolonged circulation time (t1/2β = 4.5 h) and a decreased renal clearance (0.8 mL/min/kg) [1]
|
| 毒性/毒理 (Toxicokinetics/TK) |
In mice, doses up to 1 mg/kg (intravenous) of cyclic (RGDyK) TFA did not show acute toxicity, and no significant weight loss or histopathological abnormalities of the liver, kidneys or heart were observed.[1] In rats treated with cyclic (RGDyK) TFA (0.1–1 mg/kg intravenous), no adverse effects on platelet aggregation or coagulation were observed.[1] The human plasma protein binding rate of cyclic (RGDyK) TFA at a concentration of 10 nM was 32%.[1]
|
| 参考文献 | |
| 其他信息 |
Cyclo(RGDyK)TFA is a cyclic peptide containing the RGD sequence, exhibiting high selectivity for αVβ3 and αVβ5 integrins, which are overexpressed in tumor cells and angiogenic endothelial cells [1]. Its main application is as a tracer for tumor targeting and angiogenesis imaging, utilizing its specific integrin-mediated binding and favorable biokinetic properties [1]. Glycosylation of Cyclo(RGDyK)TFA can improve its in vivo stability, reduce non-specific tissue uptake, and enhance tumor targeting efficiency [1]. The mechanism of action of this peptide involves competitive binding to αVβ3/αVβ5 integrins, thereby inhibiting integrin-mediated cell adhesion and angiogenesis without significant cytotoxicity [1].
|
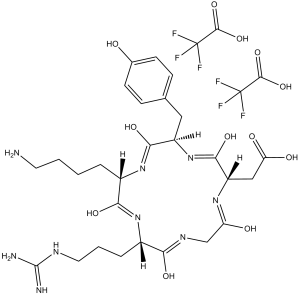
| 分子式 |
C31H43F6N9O12
|
|
|---|---|---|
| 分子量 |
847.716648340225
|
|
| 精确质量 |
847.293
|
|
| CAS号 |
250612-42-1
|
|
| 相关CAS号 |
Cyclo(RGDyK);217099-14-4
|
|
| PubChem CID |
129896716
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| tPSA |
368
|
|
| 氢键供体(HBD)数目 |
12
|
|
| 氢键受体(HBA)数目 |
20
|
|
| 可旋转键数目(RBC) |
12
|
|
| 重原子数目 |
58
|
|
| 分子复杂度/Complexity |
1130
|
|
| 定义原子立体中心数目 |
4
|
|
| SMILES |
FC(C(=O)O)(F)F.FC(C(=O)O)(F)F.O=C1C(CCCCN)NC(C(CC2C=CC(=CC=2)O)NC(C(CC(=O)O)NC(CNC(C(CCC/N=C(\N)/N)N1)=O)=O)=O)=O
|
|
| InChi Key |
CDDUWKKOPQABPG-TVSMIREGSA-N
|
|
| InChi Code |
InChI=1S/C27H41N9O8.2C2HF3O2/c28-10-2-1-4-18-24(42)34-17(5-3-11-31-27(29)30)23(41)32-14-21(38)33-20(13-22(39)40)26(44)36-19(25(43)35-18)12-15-6-8-16(37)9-7-15;2*3-2(4,5)1(6)7/h6-9,17-20,37H,1-5,10-14,28H2,(H,32,41)(H,33,38)(H,34,42)(H,35,43)(H,36,44)(H,39,40)(H4,29,30,31);2*(H,6,7)/t17-,18-,19+,20-;;/m0../s1
|
|
| 化学名 |
2-[(2S,5R,8S,11S)-8-(4-aminobutyl)-11-[3-(diaminomethylideneamino)propyl]-5-[(4-hydroxyphenyl)methyl]-3,6,9,12,15-pentaoxo-1,4,7,10,13-pentazacyclopentadec-2-yl]acetic acid;2,2,2-trifluoroacetic acid
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 请将本产品存放在密封且受保护的环境中,避免吸湿/受潮。 |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.17 mg/mL (2.56 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 21.7 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.17 mg/mL (2.56 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 21.7 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 View More
配方 3 中的溶解度: 2.17 mg/mL (2.56 mM) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.1796 mL | 5.8982 mL | 11.7963 mL | |
| 5 mM | 0.2359 mL | 1.1796 mL | 2.3593 mL | |
| 10 mM | 0.1180 mL | 0.5898 mL | 1.1796 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。