| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 2mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| Other Sizes |
|
| 靶点 |
HIV-1 (WT)(EC50=0.068 nM);HIV-1 (MDR)(EC50=0.15 nM);HIV-1 (M184V)(EC50=3.1 nM); Human immunodeficiency virus type 1 (HIV-1) reverse transcriptase (Ki = 4.3 nM for EFdA-TP) [1]
EC50s for islatravir (MK-8591) (4'Ed2FA), a strong anti-HIV-1 agent, are 0.068 nM, 3.1 nM, and 0.15 nM for HIV-1 (WT), HIV-1 (M184V), and HIV-1 (MDR), respectively. It functions as a nucleoside reverse transcriptase inhibitor [1]. [1] Islatravir triphosphate (EFdA-TP) inhibited recombinant HIV-1 RT with Ki = 4.3 ± 0.7 nM, >1000-fold more potent than tenofovir diphosphate. EC50 against HIV-1IIIB in MT-4 cells: 0.0007 ± 0.0003 μM (vs. 0.11 μM for AZT). Maintained full activity against NRTI-resistant strains (M184V, K65R) with EC50 < 0.001 μM. [1] [2] Reduced HIVJR-CSF replication in human CD4+ T cells by 3.5 log10 at 10 nM (p < 0.001 vs. control). [2] Islatravir (2'-deoxy-4'-C-ethynyl-2-fluoroadenosine, 4′Ed2FA) is a nucleoside reverse transcriptase inhibitor (NRTI). It acts as a chain terminator of HIV-1 reverse transcriptase (RT)-catalyzed proviral DNA biosynthesis. [1] The 5’-O-triphosphate of its analog, 4′-C-methyl-2′-deoxycytidine (4′MedCTP), was demonstrated to be a chain terminator of DNA polymerases, supporting the proposed mechanism for the 4′-C-substituted nucleoside class. [1] |
|---|---|
| 体外研究 (In Vitro) |
Islatravir (MK-8591) (4'Ed2FA) 是一种强效抗 HIV-1 药物,对于 HIV-1 (WT)、HIV-1 (M184V) 和 HIV 的 EC50 分别为 0.068 nM、3.1 nM 和 0.15 nM分别为-1(MDR)。它作为核苷逆转录酶抑制剂发挥作用[1]。
[1] Islatravir三磷酸盐(EFdA-TP)抑制HIV-1 RT的Ki = 4.3 ± 0.7 nM,比替诺福韦二磷酸强效>1000倍。 在MT-4细胞中抗HIV-1IIIB的EC50:0.0007 ± 0.0003 μM(AZT为0.11 μM)。 对NRTI耐药株(M184V, K65R)保持完全活性(EC50 < 0.001 μM)。 [1] [2] 在10 nM浓度下使人CD4+ T细胞的HIVJR-CSF复制降低3.5 log10(p < 0.001)。 [2] Islatravir 对多种 HIV-1 毒株表现出高效力。在 MT-4 细胞中,其对野生型 HIV-1 (LAI 株) 的 EC₅₀ 为 0.068 nM,选择性指数 (CC₅₀/EC₅₀) 为 110,000。 [1] 其对多种耐药 HIV-1 突变株保持强效活性,包括 M184V (EC₅₀ = 3.1 nM) 和多药耐药 (MDR) 株 (EC₅₀ = 0.15 nM)。 [1] 该化合物对来自 7 名经深度药物治疗的艾滋病患者的 HIV-1 分离株也具有与野生型病毒相当的活性。 [1] 与其类似物相比,Islatravir 对耐药株的活性优于其 2’,3’-双脱氧 (4′Edd2FA) 和 2’,3’-双脱氢双脱氧 (4′Ed42FA) 类似物,后两者对耐药病毒的活性显著丧失。 [1] Islatravir 对多种 HIV-1 毒株表现出高效力。在 MT-4 细胞中,其对野生型 HIV-1 (LAI 株) 的 EC₅₀ 为 0.068 nM,选择性指数 (CC₅₀/EC₅₀) 为 110,000。 [1] 其对多种耐药 HIV-1 突变株保持强效活性,包括 M184V (EC₅₀ = 3.1 nM) 和多药耐药 (MDR) 株 (EC₅₀ = 0.15 nM)。 [1] 该化合物对来自 7 名经深度药物治疗的艾滋病患者的 HIV-1 分离株也具有与野生型病毒相当的活性。 [1] 与其类似物相比,Islatravir 对耐药株的活性优于其 2’,3’-双脱氧 (4′Edd2FA) 和 2’,3’-双脱氢双脱氧 (4′Ed42FA) 类似物,后两者对耐药病毒的活性显著丧失。 [1] |
| 体内研究 (In Vivo) |
Islatravir(EFdA)治疗导致大多数接受治疗的小鼠在治疗后3周内PB中的HIV-RNA降至无法检测的水平。EFdA治疗的BLT小鼠颈阴道灌洗液中的HIV-RNA水平也降至检测不到的水平,表明EFdA强烈渗透到FRT中。我们的研究结果还表明,在所有分析的组织中,HIV复制都受到了强烈的系统性抑制。特别是,我们观察到,与未经治疗的HIV感染对照小鼠相比,EFdA治疗的BLT小鼠的胃肠道和FRT中的HIV-RNA水平存在2倍以上的差异。此外,与未经治疗的HIV感染对照小鼠相比,EFdA治疗的BLT小鼠的淋巴结、肝脏、肺、脾脏中的HIV-RNA也显著降低。此外,EFdA治疗防止了PB、粘膜组织和淋巴组织中CD4+T细胞的耗竭[2]
[1] Islatravir三磷酸盐(EFdA-TP)抑制HIV-1 RT的Ki = 4.3 ± 0.7 nM,比替诺福韦二磷酸强效>1000倍。 在MT-4细胞中抗HIV-1IIIB的EC50:0.0007 ± 0.0003 μM(AZT为0.11 μM)。 对NRTI耐药株(M184V, K65R)保持完全活性(EC50 < 0.001 μM)。 [1] [2] 在10 nM浓度下使人CD4+ T细胞的HIVJR-CSF复制降低3.5 log10(p < 0.001)。 [2] 给 HIV-1 感染的人源化 BLT 小鼠口服 Islatravir (10 mg/kg/天,持续 3 周) 导致血浆病毒载量急剧下降。治疗一周后,观察到血浆 HIV-RNA 降低了 2 个对数级。三周后,大多数治疗小鼠 (6只中的4只) 的血浆 HIV-RNA 降至检测下限以下 (LOD: 750 拷贝/mL),其余两只小鼠的病毒载量分别为 1074 和 1297 拷贝/mL。 [2] Islatravir 治疗还在两周内将宫颈阴道灌洗液 (CVL) 中的 HIV-RNA 水平显著降低至不可检测水平 (LOD: 1400 拷贝/60μL),证明其能渗透到女性生殖道 (FRT) 并在此处发挥抗病毒活性。 [2] 治疗三周后的全身组织分析显示,与未治疗的对照组相比,Islatravir 治疗组小鼠的骨髓、淋巴结、脾脏、肝脏和肺中的细胞相关 HIV-RNA 水平显著更低。值得注意的是,在脾脏和淋巴结中观察到了 2-3 个对数级的差异。 [2] 在胃肠道 (GI) 和 FRT 组织中,与未治疗小鼠相比,Islatravir 治疗使细胞相关 HIV-RNA 水平降低了 >2 个对数级。治疗组小鼠 GI 道中的 HIV-DNA 水平也显著更低。 [2] Islatravir 治疗防止了 HIV 诱导的 CD4+ T 细胞耗竭。与未治疗的对照组相比,治疗组小鼠外周血、肝脏和脾脏中维持了显著更高水平的 CD4+ T 细胞。治疗组小鼠的 GI 道和 FRT 组织中的 CD4+ T 细胞水平也更高,其中在 FRT 中的差异达到统计学显著性。 [2] 给 HIV-1 感染的人源化 BLT 小鼠口服 Islatravir (10 mg/kg/天,持续 3 周) 导致血浆病毒载量急剧下降。治疗一周后,观察到血浆 HIV-RNA 降低了 2 个对数级。三周后,大多数治疗小鼠 (6只中的4只) 的血浆 HIV-RNA 降至检测下限以下 (LOD: 750 拷贝/mL),其余两只小鼠的病毒载量分别为 1074 和 1297 拷贝/mL。 [2] Islatravir 治疗还在两周内将宫颈阴道灌洗液 (CVL) 中的 HIV-RNA 水平显著降低至不可检测水平 (LOD: 1400 拷贝/60μL),证明其能渗透到女性生殖道 (FRT) 并在此处发挥抗病毒活性。 [2] 治疗三周后的全身组织分析显示,与未治疗的对照组相比,Islatravir 治疗组小鼠的骨髓、淋巴结、脾脏、肝脏和肺中的细胞相关 HIV-RNA 水平显著更低。值得注意的是,在脾脏和淋巴结中观察到了 2-3 个对数级的差异。 [2] 在胃肠道 (GI) 和 FRT 组织中,与未治疗小鼠相比,Islatravir 治疗使细胞相关 HIV-RNA 水平降低了 >2 个对数级。治疗组小鼠 GI 道中的 HIV-DNA 水平也显著更低。 [2] Islatravir 治疗防止了 HIV 诱导的 CD4+ T 细胞耗竭。与未治疗的对照组相比,治疗组小鼠外周血、肝脏和脾脏中维持了显著更高水平的 CD4+ T 细胞。治疗组小鼠的 GI 道和 FRT 组织中的 CD4+ T 细胞水平也更高,其中在 FRT 中的差异达到统计学显著性。 [2] |
| 酶活实验 |
[1] HIV-1 RT抑制实验:
重组HIV-1 RT与poly(rA)/oligo(dT)18模板-引物在缓冲液(50 mM Tris-HCl, 50 mM KCl, 5 mM MgCl2, pH 7.8)中孵育。 加入3H-dTTP ± EFdA-TP(0.1–100 nM)启动反应,37°C反应30分钟。 通过闪烁计数定量掺入的放射性。 Lineweaver-Burk作图法计算Ki值。 [1] 基于一个工作假设,提出了使用4'-C-取代-2'-脱氧核苷衍生物的想法,以解决现有获得性免疫缺陷综合征化疗(高效抗逆转录病毒疗法)的问题。随后的研究成功地证明了这一想法的有效性,并开发了2'-脱氧-4'-C-乙炔基-2-氟腺苷,这是一种核苷逆转录酶抑制剂,对所有人类免疫缺陷病毒1型(HIV-1s)都非常有效,包括多药耐药性HIV-1,并且毒性低[1]。 评估了 Islatravir-5’-O-三磷酸 (4′Ed2FATP) 对人线粒体 DNA 聚合酶 γ 的抑制作用。4′Ed2FATP 抑制由人线粒体 DNA 聚合酶 γ 介导的 dATP 掺入的 EC₅₀ 为 10 µM。 [1] 4′Ed2FATP 对 DNA 聚合酶 α 和 β 的 EC₅₀ 值高于 200 µM。 [1] 评估了对腺苷脱氨酶的稳定性。在使其类似物 4′EdA 在 60 分钟内完全脱氨的条件下,Islatravir 完全稳定。 [1] 评估了 Islatravir-5’-O-三磷酸 (4′Ed2FATP) 对人线粒体 DNA 聚合酶 γ 的抑制作用。4′Ed2FATP 抑制由人线粒体 DNA 聚合酶 γ 介导的 dATP 掺入的 EC₅₀ 为 10 µM。 [1] 4′Ed2FATP 对 DNA 聚合酶 α 和 β 的 EC₅₀ 值高于 200 µM。 [1] 评估了对腺苷脱氨酶的稳定性。在使其类似物 4′EdA 在 60 分钟内完全脱氨的条件下,Islatravir 完全稳定。 [1] |
| 细胞实验 |
标本采集和处理[2]
在HIV暴露前后6周内纵向(每周)收集PB和CVL样本。将PB收集在EDTA中,通过在300g下离心5分钟分离血浆用于HIV-RNA分析。用PBS重构剩余的血细胞以恢复PB样品的原始体积,并用于流式细胞术分析。通过用无菌PBS进行宫颈阴道灌洗(CVL,第0-5周)获得宫颈阴道分泌物(CVS)(每次洗涤3次,每次20μl,总体积约60μl)。为确保手术无创,使用20μl无菌过滤吸管头进行CVL,吸管头插入阴道腔不超过1-3mm。离心(300g 5分钟)后,使用无细胞上清液进行HIV-RNA分析。将颗粒重新悬浮在PBS中,用于流式细胞术分析。在HIV暴露后6周的尸检中采集骨髓(BM)、LN、人胸腺类器官(ORG)、肝脏、肺、脾脏、胃肠道(从十二指肠到直肠)和FRT(阴道、宫颈和子宫),并如前所述分离单核细胞进行HIV-RNA、HIV-DNA和流式细胞术分析。 HIV病毒载量和流式细胞术分析[2] 使用一步逆转录酶实时PCR[ABI定制TaqMan设计检测法(检测限(LOD):血浆-750拷贝/ml,CVL-1400拷贝/60μl)测量PB和CVL HIV-RNA水平。低于检测限的血浆和CVL病毒载量水平分别绘制为375拷贝/ml和700拷贝/ml。我们使用这些值计算各组的平均值。通过实时RT-PCR(HIV-RNA,LOD-1.5拷贝/105细胞和HIV-DNA,LOD为2.5拷贝/105电池)确定从组织分离的单核细胞中HIV-RNA和HIV-DNA的存在。作为从人类细胞中提取的DNA存在的对照。,所有样本均通过实时PCR检测人类γ珠蛋白DNA的存在。 [1] MT-4细胞抗病毒活性: 细胞感染HIV-1IIIB(MOI 0.01)后,用Islatravir(0.0001–10 μM)处理5天。 MTT法检测细胞活性,剂量效应曲线计算EC50。 线粒体毒性: HepG2细胞暴露于Islatravir(0.1–100 μM)14天。 实时荧光定量PCR检测mtDNA;100 μM下未观察到耗竭。 [1] 使用 MTT 法测定抗 HIV-1 活性。简述如下:用 HIV-1 (LAI 株) 感染 MT-4 细胞,并在系列稀释的药物存在下培养。孵育 5 天后,通过 MTT 还原为甲臜来测量细胞活力,并计算 EC₅₀ (半数有效浓度) 和 CC₅₀ (半数细胞毒性浓度)。 [1] 使用 MAGI (多核激活半乳糖苷酶指示剂) 法测定对耐药 HIV-1 突变株的活性。用各种 HIV-1 突变株感染 MAGI-CCR5 细胞,通过测量 β-半乳糖苷酶活性来确定 EC₅₀。 [1] 在 CEM、MT-4 和 MAGI-CCR5 细胞中研究了细胞内代谢。将细胞与 Islatravir 孵育,并随时间定量检测其单磷酸、二磷酸和三磷酸代谢物 (4′Ed2FA-MP, 4′Ed2FA-DP, 4′Ed2FATP) 的细胞内水平。4′Ed2FATP 的细胞内半衰期 (T₁/₂) 约为 18 小时。此外,在用 0.1 µM 药物预孵育后,在去除细胞外 Islatravir 后,约 50% 的细胞在 24 小时内仍能免受 HIV-1 感染。 [1] 使用 MTT 法测定抗 HIV-1 活性。简述如下:用 HIV-1 (LAI 株) 感染 MT-4 细胞,并在系列稀释的药物存在下培养。孵育 5 天后,通过 MTT 还原为甲臜来测量细胞活力,并计算 EC₅₀ (半数有效浓度) 和 CC₅₀ (半数细胞毒性浓度)。 [1] 使用 MAGI (多核激活半乳糖苷酶指示剂) 法测定对耐药 HIV-1 突变株的活性。用各种 HIV-1 突变株感染 MAGI-CCR5 细胞,通过测量 β-半乳糖苷酶活性来确定 EC₅₀。 [1] 在 CEM、MT-4 和 MAGI-CCR5 细胞中研究了细胞内代谢。将细胞与 Islatravir 孵育,并随时间定量检测其单磷酸、二磷酸和三磷酸代谢物 (4′Ed2FA-MP, 4′Ed2FA-DP, 4′Ed2FATP) 的细胞内水平。4′Ed2FATP 的细胞内半衰期 (T₁/₂) 约为 18 小时。此外,在用 0.1 µM 药物预孵育后,在去除细胞外 Islatravir 后,约 50% 的细胞在 24 小时内仍能免受 HIV-1 感染。 [1] |
| 动物实验 |
病毒攻击和 EFdA 给药[2]
HIV-1JR-CSF 病毒株通过瞬时转染 293T 细胞制备,并使用 TZM-bl 细胞进行滴度测定,方法如前所述。HIV-1JR-CSF (30,000 TCIU) 通过尾静脉注射给药。 EFdA 用磷酸盐缓冲液 (PBS) 复溶至 1 mg/mL,并以 10 mg/kg 的剂量,每日一次,连续 3 周,通过灌胃法给予 BLT 小鼠,从 HIV 感染后 3 周开始给药。PBS (200 μL) 通过灌胃法给予(未处理的)对照组。 [1] 大鼠药代动力学: Sprague-Dawley 大鼠单次静脉注射 Islatravir (1 mg/kg,溶于生理盐水)。 给药后 0.08–24 小时通过颈动脉导管进行系列血液采样。[1] [2] BLT 小鼠疗效: 人源化小鼠经口灌胃给予Islatravir(10 mg/kg/天,溶于 0.5% 甲基纤维素),持续 7 天。 末次给药后 4 小时采集组织进行病毒载量分析。[2] 评估了小鼠的急性毒性。6 周龄 ICR 雄性小鼠分别通过口服 (po) 或静脉注射 (iv) 途径单次给予 Islatravir,剂量分别为 1、3、10、30 和 100 mg/kg。观察小鼠在给药后 7 天内的死亡率和体重变化。无论采用何种给药途径,在 100 mg/kg 以下的任何剂量下均未观察到急性毒性(0% 死亡率)。 [1] 对小鼠进行了急性毒性评估。六周龄的ICR雄性小鼠分别经口服(po)或静脉注射(iv)途径单次给予1、3、10、30和100 mg/kg剂量的伊斯拉曲韦。给药后观察小鼠的死亡率和体重变化长达7天。无论采用何种给药途径,在100 mg/kg剂量范围内均未观察到急性毒性(0%死亡率)。[1] |
| 药代性质 (ADME/PK) |
[1] 大鼠血浆半衰期:8.7 ± 1.2 小时。
EFdA-TP 在人外周血单核细胞 (PBMC) 中的细胞内半衰期:>36 小时。 小鼠口服生物利用度:82 ± 9%。[1] 伊斯拉曲韦 在细胞内表现出较高的代谢稳定性。其活性三磷酸形式 (4′Ed2FATP) 在 CEM、MT-4 和 MAGI-CCR5 细胞中的细胞内半衰期 (T₁/₂) 约为 18 小时。[1] 它在模拟胃液 (pH 1.06) 的酸性条件下稳定。在 24°C 下放置 120 分钟后仅观察到 3% 的分解,而 2',3'-二脱氧腺苷 (ddA) 在相同条件下 5 分钟内完全分解。 [1] 该化合物对腺苷脱氨酶完全稳定。[1] 伊斯拉曲韦在细胞内表现出很高的代谢稳定性。其活性三磷酸形式(4′Ed2FATP)在CEM、MT-4和MAGI-CCR5细胞中的细胞内半衰期(T₁/₂)约为18小时。[1] 它在模拟胃液(pH 1.06)的酸性条件下稳定。在24°C下放置120分钟后仅观察到3%的分解,而2',3'-二脱氧腺苷(ddA)在相同条件下5分钟内完全分解。[1] 该化合物对腺苷脱氨酶完全稳定。[1] |
| 毒性/毒理 (Toxicokinetics/TK) |
[1] 在MT-4细胞中,CC50:>100 μM(选择性指数>140,000)。
在HepG2细胞中,100 μM浓度下未观察到线粒体DNA耗竭(而20 μM扎西他滨可导致47%的线粒体DNA耗竭)。[1] [2] 在BLT小鼠中,10 mg/kg/天的剂量下,肠道/淋巴组织未见组织病理学异常。[2] 伊斯拉曲韦在ICR小鼠中单次口服或静脉注射剂量高达100 mg/kg后,未显示急性毒性(0%死亡率,每组n=8)。 [1] 其三磷酸形式(4′Ed2FATP)对人线粒体DNA聚合酶γ的抑制EC₅₀(10 µM)高于ddATP(0.2 µM),表明其线粒体毒性潜力较低。[1] 在细胞试验中,对MT-4细胞的CC₅₀(细胞毒性浓度)为7500 nM。[1] 在ICR小鼠中,单次口服或静脉注射剂量高达100 mg/kg的伊斯拉曲韦未显示急性毒性(0%死亡率,每组n=8)。 [1] 其三磷酸形式(4′Ed2FATP)对人线粒体DNA聚合酶γ的抑制EC₅₀值(10 µM)高于ddATP(0.2 µM),表明其线粒体毒性较低。[1] 在细胞实验中,其对MT-4细胞的CC₅₀(细胞毒性浓度)为7500 nM。[1] |
| 参考文献 |
|
| 其他信息 |
伊斯拉曲韦是一种正在研究用于治疗和预防HIV感染的试验性药物。
伊斯拉曲韦属于一类称为核苷类逆转录酶转位抑制剂(NRTTIs)的HIV药物。NRTTIs通过多种不同的方法来阻断一种名为逆转录酶的HIV酶。通过阻断逆转录酶,核苷类逆转录酶抑制剂(NRTTIs)可阻止HIV病毒复制,并降低体内HIV病毒的数量。 伊斯拉曲韦可能对某些对其他抗HIV药物产生耐药性的HIV毒株有效。 伊斯拉曲韦正在临床试验NCT04233216(多拉韦林/伊斯拉曲韦(DOR/ISL)用于治疗既往接受过大量治疗的HIV-1感染者(MK-8591A-019))中进行研究。 药物适应症 预防人类免疫缺陷病毒(HIV-1)感染。 基于一项工作假设,提出了使用4'-C-取代的-2'-脱氧核苷衍生物来解决现有获得性免疫缺陷综合征化疗(高效抗逆转录病毒疗法)问题的设想。后续研究成功验证了该理念的有效性,并最终开发出核苷类逆转录酶抑制剂2'-脱氧-4'-C-乙炔基-2-氟腺苷。该抑制剂对包括多重耐药HIV-1在内的所有1型人类免疫缺陷病毒(HIV-1)均具有高效抑制作用,且毒性较低。[1] 背景:处于临床前开发阶段的核苷类逆转录酶抑制剂(NRTI)4'-乙炔基-2-氟-2'-脱氧腺苷(EFdA)与已获批准的NRTI相比,展现出更高的安全性和更强的抗病毒活性,且耐药性更低。然而,EFdA的全身抗病毒疗效尚未得到充分评估。本研究利用骨髓/肝脏/胸腺(BLT)人源化小鼠模型,探讨了EFdA治疗对HIV复制以及外周血(PB)和组织中CD4+ T细胞耗竭的全身性影响。我们重点对雌性生殖道(FRT)和胃肠道(GI)进行了全面分析,这两个部位是主要的病毒传播、复制和CD4+ T细胞耗竭部位,也是一些现有抗逆转录病毒药物疗效欠佳的部位。结果:EFdA治疗后,大多数受试小鼠外周血(PB)中的HIV-RNA水平在治疗后3周降至无法检测的水平。EFdA治疗的BLT小鼠宫颈阴道灌洗液中的HIV-RNA水平也降至无法检测的水平,表明EFdA能够有效渗透至FRT。我们的结果还表明,在所有分析的组织中,HIV复制均受到强烈的系统性抑制。尤其值得注意的是,与未经治疗的HIV感染对照小鼠相比,EFdA治疗的BLT小鼠胃肠道和FRT中的HIV-RNA水平降低了2个数量级以上。此外,与未经治疗的HIV感染对照小鼠相比,EFdA治疗的BLT小鼠淋巴结、肝脏、肺和脾脏中的HIV-RNA水平也显著降低。此外,EFdA治疗阻止了外周血、黏膜组织和淋巴组织中CD4+ T细胞的耗竭。结论:我们的研究结果表明,EFdA在控制病毒复制和保护CD4+ T细胞方面非常有效,尤其是在胃肠道和生殖道中。因此,EFdA是抗逆转录病毒治疗方案中一个极具潜力的候选药物。[2] [1] 伊斯拉曲韦可掺入病毒DNA中,不引起链终止,但会导致延迟终止。对HIV-1 M/O组、HIV-2和SIV均有效。[1] [2] 在肠道相关淋巴组织中可达到高浓度(高于EC90的10倍)。 [2] 伊斯拉曲韦的设计基于一个工作假设,旨在克服现有高效抗逆转录病毒疗法(HAART)的局限性,特别是针对耐药性HIV的出现。该设计涉及在2'-脱氧核苷骨架上进行4'-C取代,保留3'-OH基团以模拟天然底物,但使其不参与链延长反应,从而起到链终止剂的作用。[1] 4'-C-乙炔基取代基有助于提高代谢稳定性(抵抗酸性和酶促降解)并由于其亲脂性的增加而有利于细胞穿透。[1] 腺嘌呤碱基2位上的氟化赋予其抵抗腺苷脱氨酶的稳定性。 [1] 由于其作用机制和对耐药毒株的活性,伊斯拉曲韦也被认为是一种潜在的乙型肝炎病毒(HBV)感染治疗候选药物,因为HBV在复制过程中也利用逆转录酶,并且已知其与HIV核苷类逆转录酶抑制剂(NRTIs)存在交叉耐药性。[1] 伊斯拉曲韦的设计基于一个工作假设,旨在克服现有高效抗逆转录病毒疗法(HAART)的局限性,特别是针对耐药HIV的出现。该设计涉及在2'-脱氧核苷骨架上进行4'-C取代,保留3'-OH基团以模拟天然底物,但使其不参与链延长反应,从而起到链终止剂的作用。[1] 4'-C-乙炔基取代基有助于提高代谢稳定性(抵抗酸性和酶促降解)并由于其亲脂性的增加而有利于细胞渗透。 [1] 腺嘌呤碱基2位上的氟化赋予其抵抗腺苷脱氨酶的稳定性。[1] 由于其作用机制和对耐药毒株的活性,伊斯拉曲韦也被认为是一种潜在的乙型肝炎病毒(HBV)感染治疗候选药物,因为HBV在复制过程中也利用逆转录酶,并且已知其与HIV核苷类逆转录酶抑制剂(NRTIs)存在交叉耐药性。[1] |
| 分子式 |
C12H12FN5O3
|
|---|---|
| 分子量 |
293.253785133362
|
| 精确质量 |
293.09
|
| 元素分析 |
C, 49.15; H, 4.12; F, 6.48; N, 23.88; O, 16.37
|
| CAS号 |
865363-93-5
|
| 相关CAS号 |
EFdA-TP;950913-56-1; 2408129-39-3 (hydrate)
|
| PubChem CID |
6483431
|
| 外观&性状 |
White to off-white solid powder
|
| LogP |
-0.6
|
| tPSA |
119
|
| 氢键供体(HBD)数目 |
3
|
| 氢键受体(HBA)数目 |
8
|
| 可旋转键数目(RBC) |
3
|
| 重原子数目 |
21
|
| 分子复杂度/Complexity |
459
|
| 定义原子立体中心数目 |
3
|
| SMILES |
C#C[C@]1([C@H](C[C@@H](O1)N2C=NC3=C(N=C(N=C32)F)N)O)CO
|
| InChi Key |
IKKXOSBHLYMWAE-QRPMWFLTSA-N
|
| InChi Code |
InChI=1S/C12H12FN5O3/c1-2-12(4-19)6(20)3-7(21-12)18-5-15-8-9(14)16-11(13)17-10(8)18/h1,5-7,19-20H,3-4H2,(H2,14,16,17)/t6-,7+,12+/m0/s1
|
| 化学名 |
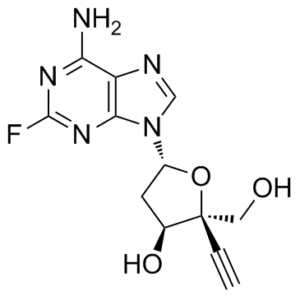
(2R,3S,5R)-5-(6-amino-2-fluoro-9H-purin-9-yl)-2-ethynyl-2-(hydroxymethyl)tetrahydrofuran-3-ol
|
| 别名 |
MK-8591; 4′-ethynyl-2-fluoro-2′-deoxyadenosine; EFdA; Islatravir; 865363-93-5; 4'-Ethynyl-2-Fluoro-2'-Deoxyadenosine; MK-8591; Islatravir [USAN]; (2R,3S,5R)-5-(6-amino-2-fluoro-9H-purin-9-yl)-2-ethynyl-2-(hydroxymethyl)tetrahydrofuran-3-ol; ISLATRAVIR ANHYDROUS; ISL; MK8591; MK 8591
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ~100 mg/mL (~341.01 mM )
H2O : ~3.57 mg/mL (~12.17 mM) |
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.08 mg/mL (7.09 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 20.8 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.08 mg/mL (7.09 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.08 mg/mL (7.09 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 配方 4 中的溶解度: ≥ 1.1 mg/mL (3.75 mM) (饱和度未知) in 5% DMSO + 40% PEG300 + 5% Tween80 + 50% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 5 中的溶解度: ≥ 1.1 mg/mL (3.75 mM) (饱和度未知) in 5% DMSO + 95% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 配方 6 中的溶解度: 10% DMSO+40% PEG300+5% Tween-80+45% Saline: ≥ 2.08 mg/mL (7.09 mM) 配方 7 中的溶解度: 1.35 mg/mL (4.60 mM) in PBS (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶. 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.4101 mL | 17.0503 mL | 34.1006 mL | |
| 5 mM | 0.6820 mL | 3.4101 mL | 6.8201 mL | |
| 10 mM | 0.3410 mL | 1.7050 mL | 3.4101 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。