| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|
| 靶点 |
proNGF
LM11A-31 HCl is a selective modulator of the p75 neurotrophin receptor (p75NTR). [1][2] It does not significantly interact with other neurotrophin receptors (e.g., TrkA, TrkB) at therapeutic concentrations[2] |
|---|---|
| 体外研究 (In Vitro) |
抑制内皮细胞炎症与RhoA激酶激活:在高糖(HG)处理的人视网膜微血管内皮细胞(HRMECs)中,0.1-10 μM的LM11A-31 HCl 以剂量依赖性方式降低HG诱导的促炎细胞因子(TNF-α、IL-6)表达35%-60%(qRT-PCR/Western blot),并抑制RhoA激活(GTP-RhoA水平降低45%-70%)[1]
- 减轻内皮通透性:1 μM浓度下,该化合物通过FITC-葡聚糖渗漏实验检测,减少55%的HG诱导HRMEC单层通透性,逆转紧密连接破坏(ZO-1和闭合蛋白上调1.8-2.2倍)[1] - 促进胆碱能神经突生长:在暴露于Aβ1-42(1 μM)的原代大鼠基底前脑胆碱能神经元(BFCNs)中,0.01-1 μM的LM11A-31 HCl 剂量依赖性增加神经突长度30%-80%,减少神经突营养不良(异常分支减少50%)[2] - 抑制神经元凋亡:在Aβ1-42处理的BFCNs中,0.1 μM LM11A-31 HCl 使凋亡率从38%(仅Aβ组)降至12%(Annexin V/PI染色),下调活化型caspase-3并上调Bcl-2[2] - 低细胞毒性:浓度高达20 μM时,MTT法检测显示HRMECs、BFCNs或正常人成纤维细胞的活力无显著降低[1][2] |
| 体内研究 (In Vivo) |
LM11A-31(口服管饲;50 mg kg/天,持续 4 周)可显着减轻 proNGF 积累并保持 BRB 完整性[1]。从 6-8 月龄开始给予 LM11A-31(口服;50 或 75 mg/kg)3 个月,可预防和/或逆转中期雄性 APPL/S 小鼠的基底前脑胆碱能神经突和皮质营养不良性神经突的萎缩。 2]。动物模型:雄性C57BL/6 J小鼠[1] 剂量:50 mg kg/天 给药方式:口服灌胃;持续 4 周 结果:减少 proNGF 积累并保持 BRB 完整性。
预防糖尿病视网膜血管通透性增加:链脲佐菌素(STZ)诱导的糖尿病C57BL/6小鼠,皮下注射LM11A-31 HCl(1、5 mg/kg/天)治疗8周。5 mg/kg剂量下,视网膜血管通透性(伊文思蓝渗漏)较溶媒对照组降低62%,视网膜TNF-α/IL-6水平降低55%-60%[1] - 抑制视网膜RhoA/ROCK通路:糖尿病小鼠经5 mg/kg/天治疗后,视网膜GTP-RhoA水平降低58%,ROCK底物磷酸化MYPT1降低65%,减轻内皮紧密连接损伤[1] - 逆转AD小鼠胆碱能神经突营养不良:12月龄3xTg-AD小鼠(中晚期AD)灌胃口服LM11A-31 HCl(10 mg/kg/天)治疗2个月。基底前脑胆碱能神经突长度增加70%,营养不良神经突密度降低65%(免疫荧光染色)[2] - 改善认知功能:10 mg/kg/天治疗的AD小鼠,Morris水迷宫实验表现显著改善:逃避潜伏期缩短40%,在目标象限停留时间增加35%(相较于溶媒对照组)[2] - 减少海马区Aβ负荷:3xTg-AD小鼠经10 mg/kg/天治疗后,海马区Aβ1-42水平(ELISA)降低38%,Aβ斑块数量(免疫组织化学)减少45%[2] |
| 酶活实验 |
p75NTR竞争结合实验:将重组人p75NTR胞外域包被在微量滴定板上,生物素标记的NGF(p75NTR配体)与系列稀释的LM11A-31 HCl(0.01-50 μM)在4°C共孵育2小时。通过链霉亲和素-HRP检测结合的生物素-NGF,该化合物剂量依赖性竞争NGF与p75NTR的结合,10 μM时竞争作用最强[2]
- RhoA激酶(ROCK)活性实验:将HG处理细胞的HRMEC裂解液与ROCK底物肽及LM11A-31 HCl(0.1-10 μM)在激酶反应缓冲液中混合,加入ATP启动反应,ELISA检测磷酸化底物。5 μM浓度下,ROCK活性被抑制62%[1] |
| 细胞实验 |
HRMEC炎症与通透性实验:HRMECs以2×105个细胞/孔接种到6孔板,血清饥饿24小时。1小时LM11A-31 HCl(0.1-10 μM)预处理后,暴露于HG(30 mM)48小时。qRT-PCR/Western blot检测细胞因子水平(TNF-α、IL-6);Transwell实验结合FITC-葡聚糖评估单层通透性[1]
- 胆碱能神经突生长实验:原代大鼠BFCNs接种到多聚L-赖氨酸包被的盖玻片,经LM11A-31 HCl(0.01-1 μM)+ Aβ1-42(1 μM)处理72小时。免疫荧光染色(ChAT抗体)结合图像分析,量化神经突长度和分支[2] - 神经元凋亡实验:BFCNs经LM11A-31 HCl(0.01-1 μM)+ Aβ1-42(1 μM)处理48小时,Annexin V-FITC/PI染色后流式细胞术量化凋亡细胞。Western blot检测活化型caspase-3和Bcl-2水平[2] - RhoA激活实验:LM11A-31 HCl(0.1-10 μM)处理后,裂解HG诱导的HRMECs,用RhoA-GTP亲和珠下拉GTP结合型RhoA,Western blot检测并通过密度分析法量化条带强度[1] |
| 动物实验 |
雄性C57BL/6J小鼠
50 mg/kg/天 灌胃;持续4周 STZ诱导糖尿病视网膜病变模型:将雄性C57BL/6小鼠(8周龄,20-25 g)腹腔注射STZ(50 mg/kg/天,连续5天)以诱导糖尿病。STZ注射后1周,将血糖>16.7 mmol/L的小鼠随机分组(每组n=8):1)溶剂对照组(0.9%生理盐水);2)LM11A-31 HCl组(1 mg/kg/天,皮下注射);3)LM11A-31 HCl组(5 mg/kg/天,皮下注射)。治疗持续8周。分析了视网膜血管通透性(伊文思蓝法)、炎症细胞因子和 RhoA/ROCK 信号通路[1] - 3xTg-AD 小鼠模型:将 12 月龄的 3xTg-AD 小鼠(雄性,25-30 g)和非转基因对照小鼠随机分组(每组 n=10):1) 非转基因小鼠 + 载体;2) 3xTg-AD 小鼠 + 载体;3) 3xTg-AD 小鼠 + LM11A-31 HCl(10 mg/kg/天,灌胃)。该化合物溶于 0.5% 羧甲基纤维素钠 (CMC) 溶液中。治疗持续 2 个月。评估了认知功能(莫里斯水迷宫)、胆碱能神经突形态和Aβ负荷[2] - 急性毒性试验:ICR小鼠(20-25 g)单次皮下注射LM11A-31 HCl(100-1000 mg/kg)或口服LM11A-31 HCl(200-2000 mg/kg)。观察小鼠14天,记录死亡率和异常行为;每3天记录一次体重[2] |
| 药代性质 (ADME/PK) |
口服吸收:大鼠单次口服 30 mg/kg 剂量后的口服生物利用度为 52%。口服给药后 2 小时达到血浆峰浓度 (Cmax) 3.8 μg/mL[2]
- 脑和视网膜渗透:小鼠口服给药 (10 mg/kg) 后,给药后 4 小时脑/血浆浓度比为 0.35,视网膜/血浆浓度比为 0.42,证实了中枢神经系统和眼部组织的渗透[1][2] - 半衰期:末端消除半衰期 (t1/2) 在血浆中为 7.2 小时,在脑中为 8.5 小时,在视网膜中为 9.1 小时[2] - 分布:广泛分布于外周组织(肝脏、脾脏、肾脏)和靶组织(脑、视网膜),组织/血浆比为 1.1-1.6[1][2] - 代谢:代谢极少;母体化合物占血浆中循环药物相关物质的78%[2] |
| 毒性/毒理 (Toxicokinetics/TK) |
急性毒性:小鼠单次皮下注射剂量高达 1000 mg/kg 或口服剂量高达 2000 mg/kg 均未导致死亡。皮下注射剂量 ≥500 mg/kg 时观察到轻微的短暂镇静,48 小时内消退[2]
- 亚慢性毒性:小鼠每日皮下/口服 5-10 mg/kg,持续 8-12 周,体重、血液学参数(红细胞、白细胞、血小板)或肝肾功能(ALT、AST、BUN、肌酐)均未见显著变化。主要器官未见组织病理学损伤[1][2] - 体外细胞毒性:HRMEC、BFCN 和正常人成纤维细胞的 CC50 > 20 μM[1][2] - 无脱靶毒性:治疗剂量下不影响 TrkA/TrkB 受体信号传导或正常神经元功能[2] |
| 参考文献 |
|
| 其他信息 |
背景:LM11A-31 HCl 是一种合成的小分子 p75NTR 配体,用于治疗神经退行性疾病和神经血管疾病[1][2]
- 作用机制:作为 p75NTR 调节剂,可:1) 抑制血管内皮细胞中的促炎信号(NF-κB 通路)和 RhoA/ROCK 激活(糖尿病视网膜病变); 2) 通过PI3K/Akt和MAPK等下游通路,促进胆碱能神经突生长,减少Aβ诱导的细胞凋亡,并调节AD模型中Aβ的清除[1][2] - 治疗适应症:拟用于治疗糖尿病视网膜病变(预防血管通透性和炎症)和阿尔茨海默病(逆转胆碱能神经突营养不良并改善认知功能)[1][2] - 主要优势:可穿透中枢神经系统和眼部组织(对靶组织至关重要),全身毒性低,且对p75NTR具有选择性(避免Trk受体相关的副作用)[1][2] - 制剂:已开发为口服和皮下制剂,可溶于水溶液(例如生理盐水、0.5% CMC),方便给药[1][2] |
| 分子式 |
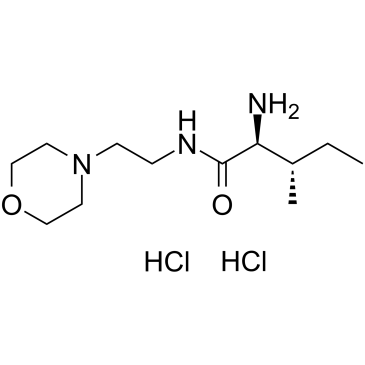
C12H25N3O2
|
|---|---|
| 分子量 |
316.267
|
| 精确质量 |
243.195
|
| 元素分析 |
C, 45.57; H, 8.61; Cl, 22.42; N, 13.29; O, 10.12
|
| CAS号 |
1243259-19-9
|
| 相关CAS号 |
(Rac)-LM11A-31 dihydrochloride; 1214672-15-7
|
| PubChem CID |
18604758
|
| 外观&性状 |
White to off-white solid powder
|
| LogP |
1.286
|
| tPSA |
71.08
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
4
|
| 可旋转键数目(RBC) |
6
|
| 重原子数目 |
17
|
| 分子复杂度/Complexity |
230
|
| 定义原子立体中心数目 |
2
|
| SMILES |
CCC(C)C(C(=O)NCCN1CCOCC1)N.Cl.Cl
|
| InChi Key |
LLIHJRRZJDEKLB-ULEGLUPFSA-N
|
| InChi Code |
InChI=1S/C12H25N3O2.2ClH/c1-3-10(2)11(13)12(16)14-4-5-15-6-8-17-9-7-15;;/h10-11H,3-9,13H2,1-2H3,(H,14,16);2*1H/t10-,11-;;/m0../s1
|
| 化学名 |
(2S,3S)-2-amino-3-methyl-N-(2-morpholin-4-ylethyl)pentanamide;dihydrochloride
|
| 别名 |
LM11A-31 HCl; LM11A-31 hydrochloride; LM11A-31; LM11A 31; LM11A31; LM 11A-31; LM 11A31; LM-11A-31;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 请将本产品存放在密封且受保护的环境中(例如氮气保护),避免吸湿/受潮。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
H2O: ~100 mg/mL (~316.2 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 100 mg/mL (316.19 mM) in PBS (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶。
请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1619 mL | 15.8093 mL | 31.6186 mL | |
| 5 mM | 0.6324 mL | 3.1619 mL | 6.3237 mL | |
| 10 mM | 0.3162 mL | 1.5809 mL | 3.1619 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
|
|
|
|
|