| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
Chk1 (Ki = 0.49 nM); VEGFR2 (Ki = 8 nM); Fms (Ki = 10 nM); YES (Ki = 14 nM); Chk2 (Ki = 47 nM)
PF-477736 (PF-00477736) targets checkpoint kinase 1 (Chk1) with a Ki value of 0.13 nM and an IC50 value of 0.3 nM in recombinant kinase assays [1] PF-477736 inhibits checkpoint kinase 2 (Chk2) with an IC50 value of 1.9 nM, showing ~6.3-fold selectivity for Chk1 over Chk2 [1] PF-477736 exhibits minimal inhibition of other kinases (ATM, ATR, CDK1, Aurora A/B) with IC50 values > 1 μM [1][2] |
|---|---|
| 体外研究 (In Vitro) |
PF-477736 (128 nM) 在 CA46 和 HeLa 细胞中以剂量依赖性方式消除喜树碱诱导的 DNA 损伤检查点。 PF-477736 可有效消除吉西他滨诱导的 S 期停滞,并相应增加 HT29 细胞中的凋亡细胞群。 PF-477736 (540 nM) 在 HT29 细胞中以时间和剂量依赖性方式增强吉西他滨诱导的细胞毒性。在 MTT 测定中,PF-477736 增强了一组化疗药物对多种 p53 缺陷人类癌细胞系的生长抑制活性。向吉西他滨阻滞的细胞中添加 PF-477736 (360 nM) 会诱导 H2AX 磷酸化强度急剧增加,反映出 DNA 损伤位点附近有更多的 γ-H2AX 分子。 PF-477736 (0.5 nM) 在姜黄素存在的情况下选择性阻断 HL-60 细胞中的 p73 和 P53 磷酸化。 PF-477736 (360 nM) 抑制多西他赛诱导的组蛋白 H3 (Ser10) 和 Cdc25C (Ser216) 磷酸化,并增强 COLO205 细胞的凋亡。 PF-477736 (250 nM) 与 MK-1775 组合在 OVCAR-5 细胞中具有显着的协同细胞毒活性。 PF-477736 (250 nM) 与 MK-1775 结合会导致 OVCAR-5 细胞中 DNA 含量在 2N 至 4N 之间的细胞积累。 PF-477736 (250 nM) 与 MK-1775 结合会导致 DNA 复制结束前过早有丝分裂,受损的 DNA 会导致 OVCAR-5 细胞凋亡。激酶检测:检测在 96 孔板中于 30℃ 下进行 20 分钟,检测缓冲液含有 50 mM TRIS pH 7.5、0.4 M NaCl、4 mM PEP、0.15 mM NADH、28 单位乳酸脱氢酶/ mL、16 单位丙酮酸激酶/mL、3 mM DTT、0.125 mM Syntide-2、0.15 mM ATP 和 25 mM 氯化镁。使用 1 nM CHK1 激酶结构域启动测定。 CHK1 活性的抑制是通过测量不同浓度 PF-477736 存在下的初始速度来确定的。使用酶动力学和 Excel 软件对数据进行分析,并拟合竞争性抑制的动力学模型以获得 Ki 值。通过针对第 2 组约 100 种蛋白激酶筛选 1 μM 或 10 μM 的化合物来评估 PF-477736 的激酶选择性。细胞测定:IC50 测定测量 PF-477736 对 p53 缺陷型人类癌细胞系的抗增殖作用。将每行细胞以指数增长密度接种在 96 孔测定板的完全培养基中,并使其贴壁 16 小时。然后进行 PF-477736 的系列稀释,并将适当的对照添加到每个板中。将细胞与药物一起孵育 96 小时。孵育后,将用完全培养基稀释的MTT工作液添加至每孔中,并将细胞孵育4小时。离心并去除上清液后,将 DMSO 添加到每个孔中,并在 SpectraMax 读板器上在 540 nm 处读取板的读数。
在多种人类实体瘤细胞系(HCT116、A549、MCF-7、PC3、SKOV3、HT29)中,PF-477736 表现出抗增殖活性,IC50 值范围为 3 nM 至 38 nM [1] - 5 nM PF-477736 可废除顺铂在 HCT116 细胞中诱导的 G2/M 检查点,24 小时后使 G2/M 期细胞积累比例从 64% 降至 21% [1] - 15 nM PF-477736 单独处理 A549 细胞仅诱导 9% 细胞凋亡,但与吉西他滨(5 nM)联合处理 72 小时后,凋亡率升高至 72% [1] - Western blot 检测显示,PF-477736 可抑制 HCT116 细胞中 Chk1 介导的 CDC25C(Ser216)和 Chk1(Ser345)磷酸化,10 nM 浓度时抑制作用最强 [1][2] - PF-477736 与 DNA 损伤剂联合使用时表现出协同抗增殖效应:在 HCT116 细胞中,与顺铂联合的协同指数(CI)= 0.29,与吉西他滨联合 CI = 0.23,与多柔比星联合 CI = 0.38,与伊立替康联合 CI = 0.41 [1][3] - 在 p53 缺陷型肿瘤细胞系(HCT116 p53⁻/⁻、MDA-MB-231)中,PF-477736 表现出增强的抗增殖活性(IC50 = 3 nM 至 10 nM),优于 p53 正常表达细胞(IC50 = 22 nM 至 38 nM)[1] - 20 nM PF-477736 可增强吉西他滨处理细胞中的 DNA 双链断裂,γ-H2AX 灶点形成较单独吉西他滨处理增加 4.3 倍 [2] - 在人类急性髓系白血病(AML)细胞系(MV4-11、HL-60、THP-1)中,PF-477736 抑制细胞增殖,IC50 值范围为 4 nM 至 18 nM [3] - 在患者来源的卵巢癌细胞中,PF-477736 抑制细胞增殖,IC50 值范围为 6 nM 至 25 nM,且与卡铂协同作用(CI = 0.35-0.48)[4] - 12 nM PF-477736 阻断羟基脲在 HL-60 细胞中诱导的 S 期检查点激活,使 S 期细胞死亡增加 52% [3] |
| 体内研究 (In Vivo) |
PF-477736 (4 mg/kg iv) 在大鼠中的终末半衰期 (T1/2) 为 2.9 小时,AUC 为 5.72 μg×hr/mL,CLp 为 11.8 mL/min/kg。 PF-477736 剂量依赖性地增强 Colo205 异种移植小鼠模型中吉西他滨最大耐受剂量的抗肿瘤活性。 PF-477736 (12 mg/kg) 可诱导 Colo205 异种移植小鼠模型中组蛋白 H3 (Ser10) 和磷酸化组蛋白 H2AX 的磷酸化增加。 PF-477736 (15 mg/kg ip) 可增强 COLO205 和 MDA-MB-231 异种移植模型中多西紫杉醇诱导的肿瘤生长抑制和肿瘤生长延迟。 PF 477736(10 mg/kg,每日一次,腹膜内注射)与 MK-1775(30 mg/kg,每日两次,口服)联合使用,可对携带 OVCAR-5 异种移植物的小鼠产生更大的肿瘤生长抑制作用。
在 HCT116 人结直肠癌异种移植模型(nu/nu 小鼠)中,PF-477736 口服给药(60 mg/kg,每日两次,连续 14 天)联合顺铂(5 mg/kg,腹腔给药,第 1、5、9 天)的肿瘤生长抑制率(TGI)达 94%,而顺铂单独处理的 TGI 为 46% [1] - 在 A549 人非小细胞肺癌(NSCLC)异种移植模型(nu/nu 小鼠)中,PF-477736 口服给药(50 mg/kg,每日两次,连续 14 天)联合吉西他滨(100 mg/kg,腹腔给药,第 1、5、9 天)的 TGI 为 91%,荷瘤小鼠中位生存期较吉西他滨单独处理延长 80% [1] - 在 MV4-11 人 AML 异种移植模型(SCID 小鼠)中,PF-477736 口服给药(30 mg/kg,每日两次,连续 21 天)联合阿糖胞苷(50 mg/kg,腹腔给药,每日一次,连续 5 天)使肿瘤负荷降低 89%,中位生存期从 30 天延长至 58 天 [3] - 在患者来源的卵巢癌异种移植模型(nu/nu 小鼠)中,PF-477736 口服给药(40 mg/kg,每日两次,连续 14 天)联合卡铂(40 mg/kg,腹腔给药,第 1 和 8 天)的 TGI 为 86%,肿瘤再生长延迟 28 天 [4] - PF-477736 与吉西他滨联合处理组的肿瘤组织中,TUNEL 阳性凋亡细胞增加(48% vs 吉西他滨单独处理组 16%),Ki-67 增殖指数降低(17% vs 吉西他滨单独处理组 63%)[1] |
| 酶活实验 |
该实验在 96 孔板中于 30°C 下进行 20 分钟,使用 0.1 mL 测定缓冲液,其中含有 25 mM 氯化镁、0.4 M NaCl、4 mM PEP、0.15 mM NADH、28 单位乳酸脱氢酶/mL 、16 单位丙酮酸激酶/mL、3 mM DTT、0.125 mM Syntide-2、0.15 mM ATP 和 28 单位乳酸脱氢酶/mL。添加一纳米的 CHK1 激酶结构域以开始测定。通过测量 PF-477736 以不同浓度存在时的初始速度,可以确定 CHK1 活性的抑制情况。通过使用酶动力学和 Excel 软件进行分析,将竞争性抑制的动力学模型拟合到数据,从而得出 Ki 值。针对第二组大约 100 种蛋白激酶检查 1 μM 或 10 μM 的 PF-477736,可以确定该化合物的激酶选择性。
重组 Chk1/Chk2 激酶活性测定:反应体系包含重组人 Chk1/Chk2、ATP(10 μM)和荧光标记肽底物,加入系列浓度的 PF-477736(0.05 nM 至 20 nM),30°C 孵育 60 分钟。通过荧光共振能量转移(FRET)检测磷酸化底物,非线性回归计算 Ki/IC50 值 [1] - 激酶选择性面板测定:采用相同的 FRET 方法,在 1 μM 浓度下测试 PF-477736 对 50 种人类激酶的抑制作用。相对于溶媒对照组计算抑制率,对抑制率 > 20% 的激酶计算 IC50 值 [1] - Chk1 结合测定:采用表面等离子体共振(SPR)技术测量结合亲和力。PF-477736 系列稀释(0.1 nM 至 10 nM)后通过固定有 Chk1 的传感器芯片,记录结合响应信号,通过稳态分析推导解离常数(Kd)[2] |
| 细胞实验 |
使用 IC50 测定法测量 PF-477736 对具有 p53 缺陷的人类癌细胞系的抗增殖作用。将每个细胞系以指数增长的密度接种到含有完全培养基的 96 孔测定板中,并使细胞贴壁 16 小时。之后,连续稀释 PF-477736,并将适当的对照添加到每个板中。该药物在细胞中孵育九十六小时。每个孔都充满已在完全培养基中稀释的 MTT 工作液,并将细胞再孵育 4 小时。离心并去除上清液后,将 DMSO 添加到每个孔中,然后使用 SpectraMax 读板器在 540 nm 处读取平板。
抗增殖实验:癌细胞或患者来源的原代细胞接种于 96 孔板(3×103 个细胞 / 孔),用系列浓度的 PF-477736(1 nM 至 200 nM)单独或与 DNA 损伤剂联合处理 72 小时。基于四唑盐还原的比色法评估细胞活力,计算 IC50 值及协同指数 [1][3][4] - 细胞周期分析:细胞用 PF-477736(5 nM)联合顺铂(2 μM)处理 24 小时后,收集细胞,70% 乙醇固定,碘化丙啶染色,流式细胞术测定细胞周期分布 [1][2] - 凋亡实验:细胞经 PF-477736(15 nM)和 / 或吉西他滨(5 nM)处理 72 小时后,用膜联蛋白 V-FITC 和碘化丙啶染色,流式细胞术分析 [1][3] - Western blot 分析:细胞用冰浴 RIPA 缓冲液裂解,蛋白经 SDS-PAGE 分离后转移至膜上,与抗磷酸化 CDC25C(Ser216)、磷酸化 Chk1(Ser345)、γ-H2AX、剪切型半胱天冬酶 -3/7、PARP 及 β- 肌动蛋白抗体孵育。化学发光法检测信号,密度计量法定量 [1][2][4] - γ-H2AX 灶点实验:细胞用 PF-477736(20 nM)和吉西他滨(5 nM)处理 24 小时后,固定,用 γ-H2AX 抗体和 DAPI 染色,荧光显微镜观察。通过图像分析软件计数每个细胞的灶点数量 [2] - 克隆形成实验:AML 细胞用 PF-477736(3 nM 至 15 nM)处理 24 小时后,接种于甲基纤维素培养基中,14 天后计数菌落(> 50 个细胞)。相对于溶媒对照组计算克隆形成效率 [3] |
| 动物实验 |
Colo205异种移植小鼠模型
40 mg/kg 静脉注射 HCT116结肠癌异种移植模型:将5×10⁶个HCT116细胞皮下植入6-8周龄的雌性nu/nu小鼠体内。当肿瘤体积达到100-150 mm³时,将小鼠随机分组(每组n=8),并分别进行以下治疗:(1)口服载体(0.5%甲基纤维素+0.2%吐温80),(2)口服PF-477736(60 mg/kg),每日两次,持续14天,(3)第1、5、9天腹腔注射顺铂(5 mg/kg),(4)PF-477736+顺铂。每2天测量一次肿瘤体积和体重[1] - A549 NSCLC异种移植模型:将5×10⁶个A549细胞皮下植入6-8周龄的雌性nu/nu小鼠体内。待肿瘤体积达到100-150 mm³时,将小鼠随机分组(每组n=8),并分别接受以下治疗:(1)口服载体;(2)口服PF-477736(50 mg/kg),每日两次,持续14天;(3)腹腔注射吉西他滨(100 mg/kg),分别于第1、5、9天给药;(4)PF-477736+吉西他滨。监测肿瘤体积和生存情况[1] - MV4-11 AML异种移植模型:将1×10⁷个MV4-11细胞静脉注射到6-8周龄的雌性SCID小鼠体内。接种后7天,将小鼠随机分组(每组n=8),并分别接受以下治疗:(1)口服赋形剂;(2)口服PF-477736(30 mg/kg),每日两次,持续21天;(3)腹腔注射阿糖胞苷(50 mg/kg),每日一次,持续5天;(4)PF-477736+阿糖胞苷。记录肿瘤负荷和生存情况[3]。 - 患者来源的卵巢癌异种移植模型:将1×10⁷个患者来源的卵巢癌细胞皮下植入雌性nu/nu小鼠(6-8周龄)。肿瘤体积达到 100-150 mm³ 的病例被随机分组(每组 n=8),并分别接受以下治疗:(1)口服赋形剂;(2)口服 PF-477736(40 mg/kg),每日两次,持续 14 天;(3)腹腔注射卡铂(40 mg/kg),分别于第 1 天和第 8 天给药;(4)PF-477736 + 卡铂。记录肿瘤体积和复发情况[4] |
| 药代性质 (ADME/PK) |
在小鼠中,口服 PF-477736 (60 mg/kg) 后,其血药浓度峰值 (Cmax) 为 6.8 μM,24 小时曲线下面积 (AUC0-24h) 为 42.3 μM·h,口服生物利用度为 83% [1]
- 在小鼠中,静脉注射 PF-477736 (10 mg/kg) 后,其清除率为 6.9 mL/min/kg,分布容积 (Vss) 为 1.5 L/kg,末端半衰期 (t1/2) 为 11.4 小时 [1] - PF-477736 具有良好的水溶性(pH 7.4 时 ≥180 μM)和较高的人血浆蛋白结合率 (96%) [1] - 在大鼠中,口服 PF-477736 (40 mg/kg) 后,其血药浓度峰值 (Cmax) 为 6.8 μM 5.9 μM,AUC0-24h 为 36.7 μM·h,口服生物利用度为 79% [1] - 在犬中,口服 PF-477736 (30 mg/kg) 显示 Cmax 为 4.2 μM,AUC0-24h 为 29.8 μM·h,t1/2 为 9.8 小时 [1] |
| 毒性/毒理 (Toxicokinetics/TK) |
在小鼠重复给药口服毒性研究(28 天,20-100 mg/kg/天)中,PF-477736 的最大耐受剂量 (MTD) 为 80 mg/kg/天,剂量限制性毒性 (DLT) 为骨髓抑制(100 mg/kg/天时中性粒细胞减少 38-42%)[1]
- 小鼠口服 PF-477736(60 mg/kg/天,连续 14 天)可引起短暂性体重减轻(≤5%),停药后 4 天内恢复 [1] - 小鼠接受 PF-477736(80 mg/kg/天,连续 28 天)治疗后,肝脏、肾脏、心脏或脾脏未观察到明显的组织病理学变化 [1] - PF-477736在浓度高达20 μM时未抑制人细胞色素P450酶(CYP1A2、CYP2C9、CYP2C19、CYP2D6、CYP3A4)[1] - 在I期临床试验中,PF-477736显示出可控的毒性,最常见的不良反应为中性粒细胞减少症(41%)、血小板减少症(35%)、疲乏(29%)和恶心(26%)[3] |
| 参考文献 | |
| 其他信息 |
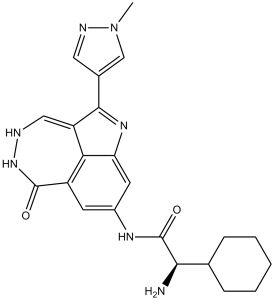
PF-00477736 是一种二氮杂环庚并吲哚类化合物,其化学名称为 8-氨基-4,5-二氢-6H-[1,2]二氮杂环庚并[4,5,6-cd]吲哚-6-酮,其 2 位被 1-甲基吡唑-4-基取代,8 位氨基与 (2R)-2-环己基甘氨酸的羧基缩合生成相应的羧酰胺。它是检查点激酶 1 (Chk 1) 的抑制剂,具有 EC 2.7.11.1(非特异性丝氨酸/苏氨酸蛋白激酶)抑制剂和抗肿瘤药物的双重活性。它是一种氨基酸酰胺,属于吡唑类和二氮杂环庚烯吲哚类化合物。
PF-00477736 已用于肿瘤治疗研究的临床试验。 CHK1 抑制剂 PF-477736 是一种专有化合物,靶向细胞周期检查点激酶 1 (chk1),具有潜在的化学增效活性。Chk1 抑制剂 PF-477736 可抑制 chk1,chk1 是一种 ATP 依赖性丝氨酸/苏氨酸激酶,是 DNA 复制监控 S/G2 检查点系统的关键组成部分。 Chk1抑制剂PF-477736通过绕过DNA损伤剂诱导的致命损伤的最后一道检查点防御,可能增强多种化疗药物对具有内在检查点缺陷的肿瘤细胞的抗肿瘤疗效。 PF-477736(PF-00477736)是一种强效且选择性的小分子Chk1抑制剂,对Chk2具有中等活性,Chk2是DNA损伤反应和细胞周期检查点的关键调节因子[1]。 PF-477736的作用机制包括阻断G2/M期和S期检查点,迫使DNA未修复的癌细胞进入有丝分裂,最终导致有丝分裂灾难和细胞凋亡[1][2][3]。 PF-477736可增强DNA靶向化疗的疗效,尤其在以下方面表现出增强的活性: p53缺陷型肿瘤依赖Chk1/Chk2介导的免疫检查点维持生存[1][4] PF-477736已进入I/II期临床试验,用于治疗晚期实体瘤(结直肠癌、肺癌、卵巢癌)和血液系统恶性肿瘤(急性髓系白血病),初步数据显示其与吉西他滨和阿糖胞苷联合使用具有抗肿瘤活性[3][4] PF-477736具有良好的药代动力学特性(口服生物利用度高、半衰期长),支持其在临床上作为口服联合疗法使用[1] |
| 分子式 |
C22H25N7O2
|
|
|---|---|---|
| 分子量 |
419.48
|
|
| 精确质量 |
419.206
|
|
| 元素分析 |
C, 62.99; H, 6.01; N, 23.37; O, 7.63
|
|
| CAS号 |
952021-60-2
|
|
| 相关CAS号 |
1175132-90-7 (HCl);1071848-28-6 952238-93-6 (?HCl);1247874-19-6 (2HCl);952021-60-2;
|
|
| PubChem CID |
135565545
|
|
| 外观&性状 |
Solid powder
|
|
| 密度 |
1.6±0.1 g/cm3
|
|
| 折射率 |
1.790
|
|
| LogP |
0.95
|
|
| tPSA |
134.48
|
|
| 氢键供体(HBD)数目 |
4
|
|
| 氢键受体(HBA)数目 |
5
|
|
| 可旋转键数目(RBC) |
4
|
|
| 重原子数目 |
31
|
|
| 分子复杂度/Complexity |
725
|
|
| 定义原子立体中心数目 |
1
|
|
| SMILES |
O=C1NN=CC2=C(C3=CN(C)N=C3)NC3C2=C1C=C(NC(=O)[C@@H](C1CCCCC1)N)C=3
|
|
| InChi Key |
NDEXUOWTGYUVGA-LJQANCHMSA-N
|
|
| InChi Code |
InChI=1S/C22H25N7O2/c1-29-11-13(9-25-29)20-16-10-24-28-21(30)15-7-14(8-17(27-20)18(15)16)26-22(31)19(23)12-5-3-2-4-6-12/h7-12,19,27H,2-6,23H2,1H3,(H,26,31)(H,28,30)/t19-/m1/s1
|
|
| 化学名 |
(2R)-2-amino-2-cyclohexyl-N-[2-(1-methylpyrazol-4-yl)-9-oxo-3,10,11-triazatricyclo[6.4.1.04,13]trideca-1,4,6,8(13),11-pentaen-6-yl]acetamide
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|---|
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3839 mL | 11.9195 mL | 23.8390 mL | |
| 5 mM | 0.4768 mL | 2.3839 mL | 4.7678 mL | |
| 10 mM | 0.2384 mL | 1.1920 mL | 2.3839 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT00437203 | Terminated | Drug: PF-00477736 Drug: gemcitabine |
Neoplasms | Pfizer | December 2006 | Phase 1 |
PF-00477736 abrogates the camptothecin-induced DNA damage checkpoint in a dose-dependent manner.Mol Cancer Ther.2008 Aug;7(8):2394-404. |
|---|
PF-00477736 effectively abrogates the gemcitabine-induced S-phase arrest with a corresponding increase in apoptotic cell populations in the combination treatment compared with the gemcitabine treatment alone.Mol Cancer Ther.2008 Aug;7(8):2394-404. |
A,PF-00477736 enhances gemcitabine-induced cytotoxicity in a time- and dose-dependent manner in HT29 cells as determined by cell survival assay. |
A,PF-00477736 potentiates the antiproliferative effect of gemcitabine.B,in vitrocytotoxicity of PF-00477736 in selected cell lines with different DNA-damaging agents.Mol Cancer Ther.2008 Aug;7(8):2394-404. |
|---|
A,in vitroeffects of gemcitabine ± PF-00477736 on the modulation of proteins involved in the G2DNA damage checkpoint pathway.B,gemcitabine + PF-00477736 combinationin vitroleads to increased DNA damage. |
 A,PF-00477736 potentiation of gemcitabine in human colon Colo205 xenograft model.B,summary of PF-00477736 potentiation of gemcitabine in human colon xenograft models.Mol Cancer Ther.2008 Aug;7(8):2394-404 |