| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|
| 靶点 |
murine hepatitis virus, delayed brain tumor cell ( EC50 = 30 nM ); SARS-CoV, HAE cell ( EC50 = 74 nM ); MERS-CoV, HAE cell ( EC50 = 74 nM ); SARS-CoV-2 ( IC50 = 3.3 μM ); SARS-CoV-2 alpha ( IC50 = 4.7 μM ); SARS-CoV-2 beta ( IC50 = 32 μM ); SARS-CoV-2 gamma ( IC50 = 3.7 μM ); SARS-CoV-2 delta ( IC50 = 9.2 μM )
GS-5734 demonstrates broad-spectrum antiviral activity against other pathogenic RNA viruses in vitro and antiviral activity against multiple EBOV variants in cell-based assays (EC50=0.06-0.14 μM).[1] With an EC50 of 0.03 μM for the murine hepatitis virus in delayed brain tumor cells and 0.074 μM for SARS-CoV and MERS-CoV in HAE cells, GS-5734 functions as a broad-spectrum therapeutic to protect against CoVs.[2] • Antiviral activity against coronaviruses: Inhibited SARS-CoV replication in primary human airway epithelial (HAE) cells with EC₅₀ = 0.069 μM; reduced viral titers by >4 log₁₀ units at 1 μM after 48 hours. Similarly inhibited MERS-CoV in HAE cells (EC₅₀ = 0.074 μM) [1]. • Activity against Ebola virus: Suppressed Ebola virus (EBOV) replication in HeLa cells (EC₅₀ = 0.086 μM) and primary human macrophages (EC₅₀ = 0.17 μM) [1]. • Mechanism of action: Remdesivir triphosphate (active metabolite) acts as an adenosine triphosphate (ATP) analog, causing delayed chain termination during viral RNA synthesis. Incorporation into nascent RNA strands inhibited subsequent nucleotide additions after 3-5 bases, terminating replication [1]. • Activity against Nipah virus: Inhibited Nipah virus replication in primary human lung microvascular endothelial cells (EC₅₀ = 0.03 μM) [2]. • Cytotoxicity: CC₅₀ > 10 μM in Vero E6 cells and >20 μM in Huh7 cells, indicating high selectivity index [1] |
|---|---|
| 体外研究 (In Vitro) |
GS-5734 在体外表现出针对其他致病性 RNA 病毒的广谱抗病毒活性,并在基于细胞的检测中表现出针对多种 EBOV 变体的抗病毒活性 (EC50=0.06-0.14 μM)。 [1] GS-5734 对迟发性脑肿瘤细胞中的鼠肝炎病毒的 EC50 为 0.03 μM,对 HAE 细胞中的 SARS-CoV 和 MERS-CoV 的 EC50 为 0.074 μM,可作为一种广谱治疗剂来预防 CoV。 [2]
• 抗冠状病毒活性: 在原代人呼吸道上皮(HAE)细胞中抑制SARS-CoV复制,EC₅₀ = 0.069 μM;1 μM浓度处理48小时后病毒滴度降低>4 log₁₀单位。在HAE细胞中同样抑制MERS-CoV(EC₅₀ = 0.074 μM)[1]。 • 抗埃博拉病毒活性: 在HeLa细胞(EC₅₀ = 0.086 μM)和原代人巨噬细胞(EC₅₀ = 0.17 μM)中抑制埃博拉病毒(EBOV)复制[1]。 • 作用机制: Remdesivir三磷酸盐(活性代谢物)作为三磷酸腺苷(ATP)类似物,在病毒RNA合成中引起延迟链终止。掺入新生RNA链后,在3-5个核苷酸下游阻止后续核苷酸添加,终止复制[1]。 • 抗尼帕病毒活性: 在原代人肺微血管内皮细胞中抑制尼帕病毒复制(EC₅₀ = 0.03 μM)[2]。 • 细胞毒性: 在Vero E6细胞中CC₅₀ > 10 μM,在Huh7细胞中CC₅₀ > 20 μM,表明高选择性指数[1] Remdesivir (GS-5734) 在 HeLa 细胞中对多种丝状病毒(埃博拉病毒 Makona 株、Kikwit 株、马尔堡病毒、本迪布焦病毒、苏丹病毒)具有广谱抗病毒活性,EC₅₀ 值在低纳摩尔至亚微摩尔水平。[1] 在 HEP-2 细胞中,对呼吸道合胞病毒(RSV)的 EC₅₀ 为 0.019 μM,CC₅₀ 为 6.0 μM。[1] 对胡宁病毒(EC₅₀ = 0.47 μM)、拉沙热病毒(EC₅₀ = 1.48 μM)和中东呼吸综合征冠状病毒(MERS-CoV,EC₅₀ = 0.34 μM)也有抑制活性,但对基孔肯雅病毒、委内瑞拉马脑炎病毒和 HIV-1 无活性(EC₅₀ > 20 μM)。[1] 在人单核细胞来源的巨噬细胞中,与 1 μM GS-5734 孵育可快速形成并持续维持细胞内 NTP,半衰期为 16 ± 1 小时。[1] |
| 体内研究 (In Vivo) |
无论何时开始治疗,施用 3 mg/kg GS-5734 均可提高生存率。病毒暴露三天后,所有接受 10 mg/kg GS-5734 治疗的动物均到达其生命阶段的末期。然而,重复给予 10 mg/kg GS-5734 剂量的动物始终表现出更强的抗病毒作用。当使用 10 mg/kg D3 方案(病毒暴露后 3 天开始)治疗时,与 EVD 相关的临床疾病体征和凝血病标志物以及终末器官病理生理学与改善相关。[1]
• 埃博拉病毒小鼠模型: 感染后1小时开始腹腔注射(25 mg/kg,每日1次,持续6天)可100%预防小鼠适应性埃博拉病毒(maEBOV)感染小鼠的死亡,而载体对照组存活率为0%。治疗后第6天血清中病毒RNA检测不到[1]。 • SARS-CoV小鼠模型: 感染后24小时开始皮下渗透泵给药(负荷剂量50 mg/kg + 维持剂量25 mg/kg/天,持续12天),使SARS-CoV感染小鼠的肺部病毒滴度降低约2 log₁₀单位,并改善肺部组织病理学[1]。 • 尼帕病毒雪貂模型: 感染前1天开始预防性给药(10 mg/kg,每日1次,持续3天),可预防体重减轻,并使咽拭子病毒RNA减少>99%[2] 在埃博拉病毒感染的猕猴模型中,感染后第3天开始,每天一次静脉注射 10 mg/kg Remdesivir (GS-5734),连续12天,可实现100%存活率,显著抑制病毒复制,并改善临床症状和病理生理指标。[1] 较低剂量(3 mg/kg)在感染后第0天或第2天开始给药,提供部分保护(存活率33–66%)。[1] |
| 酶活实验 |
体外RSV RNA合成试验[1]
使用纯化的RSV L/P复合物和RNA寡核苷酸模板(Dharmacon)在体外重建RSV聚合酶的RNA合成,该模板代表RSV前导启动子31,32,33(3′-UGCCUUUUACG-5′)的核苷酸1-14。RNA合成反应如前所述进行,不同之处在于反应混合物含有250μM三磷酸鸟苷(GTP)、10μM三磷酸尿苷(UTP)、10µM三磷酸胞苷(CTP),补充了10μCi[α-32P]CTP,并且含有10μM三磷酸腺苷(ATP)或不含ATP。在这些条件下,聚合酶能够从启动子的3位位点开始合成,但不能从1位位点开始。GS-5734的NTP代谢物在DMSO中连续稀释,并以10、30或100μM的浓度包含在每种反应混合物中,如图1f所示。在含有7M尿素的25%聚丙烯酰胺凝胶上,在Tris-牛磺酸-EDTA缓冲液中通过电泳分析RNA产物,并通过放射自显影检测放射性标记的RNA产物。 呼吸道合胞病毒A2聚合酶抑制试验[1] 转录反应在30μL反应缓冲液(50 mM三乙酸盐(pH 8.0)、120 mM乙酸钾、5%甘油、4.5 mM MgCl2、3 mM DTT、2 mM EGTA、50μg ml−1 BSA、2.5 U RNasin、20μM ATP、100μM GTP、100μM UTP、100μMCTP和1.5μCi[α-32P]ATP(3000 Ci mmol−1))中含有25μg粗制RSV-RNP复合物。选择转录测定中使用的放射性标记的核苷酸,以匹配正在评估的RSV RNP转录抑制的核苷酸类似物。 为了确定核苷酸类似物是否抑制RSV RNP转录,使用六步连续稀释法以五倍增量添加化合物。在30°C下孵育90分钟后,用350μl Qiagen RLT裂解缓冲液停止RNP反应,并使用Qiagen RNeasy 96试剂盒纯化RNA。纯化的RNA在65°C的RNA样品加载缓冲液中变性10分钟,并在含有2 M甲醛的1.2%琼脂糖/MOPS凝胶上运行。将琼脂糖凝胶干燥,暴露于Storm磷光屏,并使用Storm磷光仪显影。 抑制人RNA聚合酶II[1] 对于25μl的反应混合物,将7.5μl 1×转录缓冲液(20 mM HEPES(pH 7.2-7.5)、100 mM KCl、0.2 mM EDTA、0.5 mM DTT、20%甘油)、3 mM MgCl2、100 ng CMV阳性或阴性对照DNA以及ATP、GTP、CTP和UTP的混合物与不同浓度(0-500μM)的抑制剂在30°C下预孵育5分钟。混合物含有5-25μM(等于Km)的竞争性33P标记ATP和400μM的GTP、UTP和CTP。通过加入3.5μl HeLa和提取物开始反应。在30°C下孵育1小时后,通过加入10.6μl蛋白酶K混合物停止聚合酶反应,该混合物含有终浓度为2.5μgμl−1蛋白酶K、5%SDS和25 mM EDTA。在37°C下孵育3-12小时后,将10μl反应混合物与10μl负载染料(98%甲酰胺、0.1%二甲苯氰基和0.1%溴酚蓝)混合,在75°C下加热5分钟,然后负载到6%聚丙烯酰胺凝胶(8M尿素)上。将凝胶在70°C下干燥45分钟,并暴露于磷光屏。使用Typhoon Trio Imager和Image Quant TL软件对全长产物363个核苷酸径流RNA进行定量。 抑制人线粒体RNA聚合酶[1] 在含有10 mM HEPES(pH 7.5)、20 mM NaCl、10 mM DTT、0.1 mg ml-1 BSA和10 mM MgCl234的缓冲液中,将20纳摩尔的POLRMT与含有POLRMT轻链启动子区和线粒体(mt)转录因子TFA(100 nM)和mtTFB2(20 nM)的20 nM模板质粒(pUC18 LSP)一起孵育。将反应混合物预温至32°C,通过加入2.5μM的天然NTPs和1.5μCi的[32P]GTP来引发反应。在32°C下孵育30分钟后,在DE81纸上发现反应并定量。 • RdRp抑制实验: 重组SARS-CoV RdRp复合物(nsp7/nsp8/nsp12)与RNA模板/引物及核苷三磷酸(NTPs)孵育。加入梯度浓度的Remdesivir三磷酸盐(RTP)。通过放射性标记核苷酸掺入定量RNA合成。RTP竞争性抑制ATP掺入,IC₅₀ = 3.65 μM [1]。 • RNA链终止实验: 使用纯化的RdRp复合物进行引物延伸反应。通过凝胶电泳分析RTP掺入RNA模板的情况。结果显示RTP在掺入位点下游3-5个核苷酸处引起终止[1] 通过体外 RNA 合成实验评估活性代谢物 NTP 对 RSV RNA 聚合酶的抑制作用。使用纯化的 RSV 核糖核蛋白复合物和 RNA 寡核苷酸模板,在含放射性标记 CTP 的反应体系中加入系列稀释的 NTP。RNA 产物通过尿素-聚丙烯酰胺凝胶电泳和放射自显影分析。NTP 通过引起链提前终止来抑制 RSV 聚合酶催化的 RNA 合成。[1] 在转录实验中测试了 NTP 对人 RNA 聚合酶 II 和线粒体 RNA 聚合酶的抑制作用,使用 HeLa 核提取物或重组 POLRMT 与转录因子。NTP 在浓度高达 500 μM 时不抑制人源聚合酶。[1] |
| 细胞实验 |
HeLa和HFF-1细胞中的EBOV检测[1]
抗病毒检测在USAMRIID的BSL-4中进行。HeLa或HFF-1细胞以每孔2000个细胞的速度接种在384孔板上。在感染前2小时,使用HP D300数字分配器以两倍稀释增量从10μM开始,将化合物的十个连续稀释液(一式三份)直接添加到细胞培养物中。使用HP D300数字分配器将每个孔中的DMSO浓度归一化为1%。将测定板转移到BSL-4套件中,用EBOV Kikwit感染HeLa细胞,感染倍数为每细胞0.5 PFU,用EBOVA Makona感染HFF-1细胞,感染复数为每细胞5 PFU。将测定板在组织培养箱中孵育48小时。如补充表1所述,在免疫染色前,通过将样品在10%福尔马林溶液中再固定48小时来终止感染。 EBOV人巨噬细胞感染试验[1] 抗病毒检测在USAMRIID的BSL-4中进行。将原代人巨噬细胞以每孔40000个细胞的速度接种在96孔板中。在感染前2小时,使用HP D300数字分配器以三倍稀释增量将八到十个连续稀释的化合物(一式三份)直接加入细胞培养物中。所有孔中DMSO的浓度均归一化为1%。将平板转移到BSL-4套件中,在100μl培养基中用每细胞1 PFU的EBOV感染细胞并孵育1小时。去除接种物,用含有稀释化合物的新鲜培养基替换培养基。感染后48小时,如补充表1所述,通过免疫染色定量病毒复制。[1] 呼吸道合胞病毒A2抗病毒试验[1] 对于抗病毒试验,化合物在源板中连续稀释三倍,使用回声声转移装置将100nl稀释的化合物转移到384孔细胞培养板上。以每毫升5×105个细胞的密度添加HEp-2细胞,然后以每毫升1×104.5个组织培养感染剂量(TCID50)的滴度添加RSV A2进行感染。添加病毒后,立即使用μFlow液体分配器将20μl病毒和细胞混合物添加到384孔细胞培养板中,并在37°C下培养4天。孵育后,让细胞在25°C下平衡30分钟。通过加入20μl CellTiter-Glo活力试剂来测定RSV诱导的细胞病变效应。在25°C下孵育10分钟后,通过使用Envision平板读数器测量发光来确定细胞存活率。 • 病毒复制抑制: 细胞(如Vero E6、HAE、Huh7)以低感染复数(MOI)感染病毒(SARS-CoV、MERS-CoV、EBOV)。用梯度稀释的Remdesivir处理48-72小时。通过空斑试验或TCID₅₀定量病毒滴度;根据剂量反应曲线计算EC₅₀ [1]。 • 细胞毒性实验: 细胞用Remdesivir处理72小时。通过ATP发光法或刃天青还原法检测细胞活力。确定CC₅₀值[1] 在多种人源细胞(包括原代巨噬细胞、HeLa、HFF-1、HMVEC-TERT 和 Huh-7 细胞)中评估 GS-5734 对埃博拉病毒的抗病毒活性。细胞与系列稀释的药物孵育 3–5 天,通过免疫荧光、空斑实验或 qRT-PCR 定量病毒复制。EC₅₀ 值范围为 0.06 至 0.14 μM。[1] 在多种人源细胞系和原代细胞中,通过 CellTiter-Glo 活性实验评估细胞毒性。GS-5734 在大多数细胞类型中的 CC₅₀ 值 >20 μM,表明细胞毒性较低。[1] |
| 动物实验 |
恒河猴(Macaca mulatta)
3 mg/kg,10 mg/kg 静脉注射 体内疗效[1] 在第0天,恒河猴(Macaca mulatta)通过肌肉注射接受1000 PFU的EBOV Kikwit(埃博拉病毒H. sapiens-tc/COD/1995/Kikwit)攻击。该病毒株来源于1995年刚果民主共和国(原扎伊尔)疫情期间采集的临床样本。攻击病毒使用培养细胞(Vero或Vero E6)从临床样本中扩增,共传代四代。使用SAS统计软件,将3-6岁的动物随机分配到实验治疗组,并按性别分层(每组雄性和雌性数量相等),同时按体重平衡分组。负责评估动物健康状况(包括安乐死评估)和实施治疗的研究人员对动物分组情况不知情。疗效研究的主要终点是病毒攻击后28天的存活率。GS-5734由吉利德科学公司配制,溶于水中,并添加12%的磺丁基醚-β-环糊精(SBE-β-CD),用盐酸将pH值调节至3.0。将制剂以约1分钟/次的静脉推注方式,经右侧或左侧大隐静脉注射给麻醉动物。所有赋形剂或GS-5734的注射体积均为2.0 ml/kg体重。动物采用肌内注射麻醉剂进行麻醉,注射液为含氯胺酮(100 mg ml⁻¹)和醋丙嗪(10 mg ml⁻¹)的混合液,剂量为0.1 ml kg⁻¹体重。[1] 每天至少观察动物两次,监测疾病症状,存活至第28天的动物被认为已获得保护。研究人员通过对临床濒死动物实施安乐死,减轻了感染动物不必要的痛苦。濒死动物安乐死的标准在研究开始前已确定,包括反应程度、体温下降和/或血清化学参数的特定改变。[35] 血清化学分析使用Vitros 350化学分析仪,凝血参数评估使用Sysmex CA-1500凝血分析仪。血液学分析使用配备多物种软件的Siemens Advia 120血液学系统进行。在计划给予 GS-5734 或载体药物并同时采集血样进行临床病理学或病毒血症分析的日期,血样在给药前立即采集。[1] • 小鼠埃博拉病毒模型:BALB/c 小鼠感染 maEBOV。从感染后 1 小时开始,每天腹腔注射瑞德西韦(25 mg/kg,溶于 10% 磺丁基醚-β-环糊精),连续 6 天。监测存活率和病毒载量。[1] • 小鼠 SARS-CoV 模型:C57BL/6 小鼠感染 SARS-CoV。从感染后 24 小时开始,通过皮下渗透泵给予瑞德西韦(负荷剂量 50 mg/kg,维持剂量 25 mg/kg/天,溶于 10% 磺丁基醚-β-环糊精),连续 12 天。评估肺部病毒滴度和组织病理学[1]。 • 雪貂尼帕病毒模型:感染尼帕病毒的雪貂。预防性治疗:瑞德西韦(10 mg/kg,溶于 10% 磺丁基醚-β-环糊精)每日肌注,连续 3 天,从感染前 1 天开始。监测临床症状、体重和病毒脱落情况[2] 查看更多药代动力学评估[1] 放射性标记组织分布[1] 六只食蟹猴单次静脉注射 [14C]GS-5734,剂量为 10 mg kg−1 (25 μCi kg−1)(缓慢推注)。分别于给药后 4 小时和 168 小时从三只动物中采集组织。将组织切取后,用生理盐水冲洗,吸干水分,称重,并置于湿冰上。组织(睾丸、附睾、眼和脑;均质化后)和血浆采用液体闪烁计数法进行分析。浓度被转换为每克样品中GS-5734的纳克当量。 体内耐药病毒毒力的评估。[2] 将10至12只10周龄的雌性BALB/c(Charles River公司)小鼠分组,用氯胺酮-赛拉嗪麻醉,并通过鼻内途径感染10⁴或10³ PFU/50 µl的表达纳米荧光素酶的野生型小鼠适应性SARS-CoV(WT SARS-CoV)或经基因工程改造携带nsp12耐药突变的SARS-MA15 NanoLuc(F480L + V557L SARS-CoV)。每日称量动物体重以监测病毒相关的体重减轻。感染后第2天和第4天,每组5至6只动物用异氟烷过量麻醉处死,并采集右下肺叶,冷冻于−80°C,直至按照先前描述的方法(38)通过噬斑试验测定滴度。对5至6只动物组成的队列进行感染后7天的监测,以比较恢复动力学,之后采集肺组织样本,并按照先前样本的方法测定滴度。 恒河猴肌内注射1,000 PFU的EBOV Kikwit病毒进行攻击。 瑞德西韦 (GS-5734) 配制成 12% 磺丁基醚-β-环糊精水溶液(pH 3.0–4.0),每日一次静脉注射,连续 12 天,剂量为 3 mg/kg 或 10 mg/kg,分别于感染后第 0、2 或 3 天开始给药。监测动物的存活率、临床评分、病毒血症和临床病理学。[1] 在健康恒河猴中进行的药代动力学研究包括静脉注射 10 mg/kg GS-5734,并在 24 小时内连续采集血样进行血浆和外周血单核细胞 (PBMC) 分析。[1] 使用 [¹⁴C]GS-5734 在食蟹猴中评估组织分布,分别在给药后 4 小时和 168 小时采集组织。[1] |
| 药代性质 (ADME/PK) |
吸收、分布和排泄
瑞德西韦吸收迅速;单次30分钟静脉输注后,血浆峰浓度在0.67-0.68小时内达到(Tmax)。重复给药后,Cmax(变异系数以百分比表示)为2229 (19.2) ng/mL,AUCtau为1585 (16.6) ngh/mL。瑞德西韦代谢物[GS-441524]的测量值为:Tmax 1.51-2.00小时,Cmax 145 (19.3) ng/mL,AUCtau 2229 (18.4) ngh/mL,谷浓度Ctrough 69.2 (18.2) ng/mL。另一种代谢物 GS-704277 的测定值为:Tmax 0.75 小时,Cmax 246 (33.9) ng/mL,AUCtau 462 (31.4) ngh/mL,谷浓度 Ctrough 未测定。对食蟹猴静脉注射 10mg/kg 剂量后,药物可在 4 小时内分布至睾丸、附睾、眼睛和大脑。 瑞德西韦 74% 经尿液排出,18% 经粪便排出。回收的药物中,49% 为代谢物 [GS-441524],10% 为未代谢的母体化合物。粪便中可检测到少量(0.5%)的[GS-441524]代谢物。 关于瑞德西韦分布容积的数据尚不明确。 关于瑞德西韦清除率的数据尚不明确。 代谢/代谢物 瑞德西韦是一种磷酰胺类前药,必须在宿主细胞内代谢为三磷酸代谢物才能发挥治疗活性。据推测,瑞德西韦进入细胞后,首先经酯酶介导水解为羧酸盐形式,然后环化脱去苯氧基,最后水解环状酸酐生成可检测的丙氨酸代谢物(GS-704277)。丙氨酸代谢物随后水解生成瑞德西韦的单磷酸酯形式,该单磷酸酯形式可再次水解生成裸核苷代谢物[GS-441524],或被细胞激酶磷酸化生成活性三磷酸酯形式。 生物半衰期 单次30分钟静脉输注瑞德西韦后,其消除半衰期为1小时。在相同条件下,瑞德西韦代谢物[GS-441524]和GS-704277的消除半衰期分别为27小时和1.3小时。在非人灵长类动物中,10mg/kg静脉注射剂量的瑞德西韦血浆半衰期为0.39小时。核苷三磷酸酯代谢物在非人灵长类动物中的半衰期为14小时。核苷三磷酸代谢物在人体内半衰期约为20小时。 • 小鼠埃博拉病毒模型:BALB/c小鼠感染maEBOV。从感染后1小时开始,每天腹腔注射瑞德西韦(25 mg/kg,溶于10%磺丁基醚-β-环糊精溶液),连续6天。监测存活率和病毒载量[1]。 • 小鼠SARS-CoV模型:C57BL/6小鼠感染SARS-CoV。从感染后24小时开始,通过皮下渗透泵给予瑞德西韦(负荷剂量50 mg/kg,维持剂量25 mg/kg/天,溶于10%磺丁基醚-β-环糊精溶液),连续12天。评估肺部病毒滴度和组织病理学[1]。 • 雪貂尼帕病毒模型:雪貂感染尼帕病毒。预防性治疗:感染前1天开始,每日肌注瑞德西韦(10 mg/kg,溶于10%磺丁基醚-β-环糊精溶液),连续3天。监测临床症状、体重和病毒脱落情况[2] 在恒河猴中静脉注射10 mg/kg GS-5734后,血浆半衰期为0.39小时。该前药迅速代谢为丙氨酸中间体和母体核苷(Nuc)。[1] 外周血单核细胞(PBMC)中的细胞内NTP半衰期为14小时,并在24小时内持续高于抑制50%以上病毒所需的水平。 [1] [¹⁴C]GS-5734 在 4 小时内分布至睾丸、附睾、眼睛和大脑等放射性物质的藏匿部位,并在 168 小时后在大脑中仍可检测到。[1] |
| 毒性/毒理 (Toxicokinetics/TK) |
肝毒性
在人体志愿者研究中,接受瑞德西韦治疗7至14天后,血清转氨酶水平出现轻微升高(低于正常值上限的5倍),但未见其他肝损伤证据。在针对COVID-19住院患者的瑞德西韦对照试验中,接受瑞德西韦治疗的患者血清ALT升高率与安慰剂组相似或更低。然而,在大多数非对照研究和病例系列中,10%至50%接受瑞德西韦治疗的患者在开始治疗后1至5天内出现短暂的轻度至中度血清ALT和AST升高,而血清胆红素或碱性磷酸酶水平未发生变化。在一些临床试验中,高达9%的患者报告ALT和AST升高超过正常值上限的5倍,但这些异常在停药后恢复正常,且未出现临床上明显的肝损伤。随着瑞德西韦在新冠肺炎治疗中的广泛应用,虽然偶有报道出现ALT显著升高并伴有黄疸,但这些病例大多发生于病情危重、伴有多器官衰竭或脓毒症的患者,或接受过其他潜在肝毒性药物(如静脉注射胺碘酮)治疗的患者(病例2)。更复杂的是,血清转氨酶升高在有症状的SARS-CoV-2感染中很常见(病例1),高达60%的患者会出现这种情况,并且在重症患者以及具有新冠肺炎重症已知危险因素(如男性、高龄、高体重指数和糖尿病)的患者中更为常见。因此,血清转氨酶升高在瑞德西韦治疗期间很常见,但通常无症状,可完全逆转,且与黄疸无关。随着这种抗病毒药物在非重症或危重症患者中的广泛应用以及疗程的延长,肝毒性特征可能会更加明显。 可能性评分:D(可能是临床上明显的肝损伤的罕见原因)。 妊娠和哺乳期影响 ◉ 哺乳期用药概述 来自 5 名患者的信息表明,瑞德西韦及其活性代谢物在乳汁中的浓度非常低。此外,瑞德西韦口服吸收不良,其代谢物口服吸收也仅为部分吸收,因此婴儿不太可能从乳汁中吸收具有临床意义的药物剂量。新生儿接受静脉注射瑞德西韦治疗埃博拉病毒和 COVID-19 后,未出现严重不良反应,并且该药物已获得 FDA 批准用于至少 28 天大且体重 3 公斤的婴儿。通过母乳接触该药物的婴儿也未报告任何不良反应。根据这些信息,接受瑞德西韦治疗的母亲无需停止哺乳,但在获得更多数据之前,哺乳期间应密切监测婴儿情况。静脉输注后最常见的不良反应包括转氨酶和胆红素水平升高以及其他肝酶升高、腹泻、皮疹、肾功能损害和低血压。 ◉ 对母乳喂养婴儿的影响 根据药物警戒报告,生产商报告称,有11名母乳喂养的婴儿通过母乳接触到瑞德西韦。报告显示,母乳喂养的婴儿不会因接触瑞德西韦及其代谢物而受到不良影响。 ◉ 对哺乳和母乳的影响 截至修订日期,未找到相关的已发表信息。 ◈ 什么是瑞德西韦? 瑞德西韦是一种抗病毒药物,获准用于治疗导致 COVID-19 的 SARS-CoV-2 病毒。瑞德西韦也可用于治疗埃博拉病毒感染。瑞德西韦的商品名为 Veklury®。有关 COVID-19 的更多信息,请参阅 MotherToBaby 的情况说明书:https://mothertobaby.org/fact-sheets/covid-19/。有时,当人们发现自己怀孕时,她们会考虑改变服药方式,甚至完全停止服药。但是,在改变服药方式之前,务必咨询您的医疗保健提供者。您的医护人员可以与您讨论治疗您病情的好处以及怀孕期间未治疗疾病的风险。 ◈ 我正在服用瑞德西韦。它会使我更难怀孕吗? 目前尚无研究表明瑞德西韦是否会使怀孕更难。 ◈ 服用瑞德西韦会增加流产的风险吗? 任何妊娠都可能发生流产。目前尚无研究表明瑞德西韦会增加流产的风险。 ◈ 服用瑞德西韦会增加胎儿出生缺陷的风险吗? 每次妊娠都有3-5%的胎儿出生缺陷风险。这被称为背景风险。根据已审查的研究,尚不清楚瑞德西韦是否会在背景风险的基础上增加胎儿出生缺陷的风险。动物研究未显示会增加胎儿出生缺陷的风险。目前尚无关于孕期使用瑞德西韦导致出生缺陷风险的人体研究。 ◈ 孕期服用瑞德西韦是否会增加其他妊娠相关问题的风险? 根据70例在孕中期和孕晚期接受瑞德西韦治疗新冠病毒感染者的报告,早产(孕37周前分娩)、低出生体重(出生体重低于5磅8盎司[2500克])和剖腹产的风险较高。然而,这些患者本身也患有严重的新冠病毒感染。包括早产在内的妊娠相关问题也与孕期新冠病毒感染有关。根据这些报告,目前尚不清楚这些结果是新冠肺炎本身、药物治疗还是两者共同作用的结果。一项研究调查了39名因新冠肺炎感染而接受瑞德西韦治疗的孕妇,并将她们与56名因新冠肺炎感染而未接受瑞德西韦治疗的孕妇进行了比较。该研究表明,两组孕妇的早产率相似。这表明,增加早产风险的可能是新冠肺炎本身,而非药物。 ◈ 孕期服用瑞德西韦是否会影响孩子未来的行为或学习能力? 目前尚无研究探讨瑞德西韦是否会导致长期的行为或学习问题。有报道称,一些新生儿在确诊埃博拉病毒和新冠肺炎后直接接受了瑞德西韦治疗。这些婴儿未报告对瑞德西韦有严重不良反应。据报道,一名曾接受瑞德西韦治疗埃博拉病毒的儿童在一岁时体重和发育均正常。 ◈ 服用瑞德西韦期间哺乳: 根据一份病例报告,母乳中的瑞德西韦浓度似乎非常低。口服瑞德西韦的吸收率也很低。这意味着哺乳婴儿不太可能从母乳中吸收大量药物。有报告称,两名新生儿在出生后接受了瑞德西韦治疗埃博拉病毒和新冠病毒感染,但未出现任何药物反应。由于关于哺乳期使用瑞德西韦的信息非常有限,如果在哺乳期使用瑞德西韦,医护人员可能会密切观察婴儿的肝肾功能、血压以及是否出现腹泻或皮疹等情况。如果您怀疑婴儿出现任何反应或症状,请联系孩子的医护人员。如果一个人想母乳喂养,但由于感染新冠病毒后身体不适而无法进行,可以与医护人员或哺乳顾问合作,帮助建立或维持母乳供应,以便在康复后进行母乳喂养。请务必就所有关于母乳喂养的问题咨询您的医护人员。 ◈ 如果男性服用瑞德西韦,是否会影响生育能力(使伴侣怀孕的能力)或增加出生缺陷的风险? 目前尚无研究探讨瑞德西韦是否会影响生育能力。一般来说,父亲或精子捐赠者接触瑞德西韦不太可能增加怀孕的风险。更多信息,请参阅 MotherToBaby 网站上的“父亲暴露”情况说明书,网址为 https://mothertobaby.org/fact-sheets/paternal-exposures-pregnancy/。 蛋白质结合 瑞德西韦与人血浆蛋白的结合率为 88-93.6%,而其代谢物 [GS-441524] 和 GS-704277 的结合率分别为 2% 和 1%。 • 小鼠毒性研究:每日皮下注射(50 mg/kg,持续 12 天)导致短暂性体重减轻(约 10%),但未造成死亡。生化指标(ALT、AST、BUN)均在正常范围内[1]。 • 食蟹猴研究:静脉注射(10 mg/kg)可引起肝酶(ALT)轻度升高,但未见组织病理学改变[1] 瑞德西韦(GS-5734)体外细胞毒性较低,在大多数原代人细胞和细胞系中CC₅₀值>20 μM。[1] 在恒河猴疗效研究中,未报告明显的药物相关毒性;与载体对照组相比,治疗组动物的临床病理参数有所改善。[1] |
| 参考文献 | |
| 其他信息 |
药效学
瑞德西韦是一种核苷类似物,用于抑制RNA聚合酶的活性。由于每日仅需给药一次,其作用持续时间中等。哺乳动物DNA和RNA聚合酶(包括人线粒体RNA聚合酶)对ATP的选择性远高于瑞德西韦三磷酸,因此瑞德西韦并非这些酶的显著抑制剂,这有助于其整体耐受性和安全性。尽管如此,瑞德西韦仍存在发生超敏反应(包括过敏性休克和其他输注相关反应)、转氨酶水平升高以及与羟氯喹或氯喹合用时可能降低疗效的风险。 • 瑞德西韦是一种单磷酰胺前药,旨在将核苷类似物 GS-441524 递送至细胞内,随后代谢为活性三磷酸形式 [1]。 • 对多种 RNA 病毒家族(冠状病毒科、丝状病毒科、副黏病毒科)具有广谱活性 [2]。 • 其作用机制与利巴韦林不同;作为延迟链终止剂而非诱变剂发挥作用 [1] 瑞德西韦 (GS-5734) 是首个在非人灵长类动物中显示出对埃博拉病毒暴露后具有实质性保护作用的小分子抗病毒化合物。[1] 其对丝状病毒、沙粒病毒和冠状病毒的广谱活性表明其具有治疗其他病毒感染的潜力。 [1] 在本文发表时,正在进行针对健康志愿者的临床研究,以评估其安全性和药代动力学。[1] |
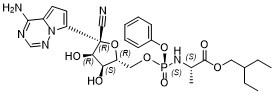
| 分子式 |
C27H35N6O8P
|
|---|---|
| 分子量 |
602.5760
|
| 精确质量 |
602.225
|
| 元素分析 |
C, 53.82; H, 5.85; N, 13.95; O, 21.24; P, 5.14
|
| CAS号 |
1809249-37-3
|
| 相关CAS号 |
1355149-45-9 [GS443902 (GS-441524 triphosphate)]; 1809249-37-3 (Remdesivir); 1191237-69-0 (GS-441524, an active metabolite of Remdesivir); 1191237-80-5 (Remdesivir O-desphosphate acetonide impurity); 1911578-74-9 (Remdesivir nucleoside monophosphate); 2250110-53-1;1911579-04-8 (GS-704277)
|
| PubChem CID |
121304016
|
| 外观&性状 |
Off-white to yellow solid powder
|
| 密度 |
1.5±0.1 g/cm3
|
| 折射率 |
1.652
|
| LogP |
2.1
|
| tPSA |
204Ų
|
| 氢键供体(HBD)数目 |
4
|
| 氢键受体(HBA)数目 |
13
|
| 可旋转键数目(RBC) |
14
|
| 重原子数目 |
42
|
| 分子复杂度/Complexity |
1010
|
| 定义原子立体中心数目 |
6
|
| SMILES |
[P@@](N([H])[C@@]([H])(C([H])([H])[H])C(=O)OC([H])([H])C([H])(C([H])([H])C([H])([H])[H])C([H])([H])C([H])([H])[H])(=O)(OC1C([H])=C([H])C([H])=C([H])C=1[H])OC([H])([H])[C@]1([H])[C@]([H])([C@]([H])([C@](C#N)(C2=C([H])C([H])=C3C(N([H])[H])=NC([H])=NN23)O1)O[H])O[H]
|
| InChi Key |
RWWYLEGWBNMMLJ-YSOARWBDSA-N
|
| InChi Code |
InChI=1S/C27H35N6O8P/c1-4-18(5-2)13-38-26(36)17(3)32-42(37,41-19-9-7-6-8-10-19)39-14-21-23(34)24(35)27(15-28,40-21)22-12-11-20-25(29)30-16-31-33(20)22/h6-12,16-18,21,23-24,34-35H,4-5,13-14H2,1-3H3,(H,32,37)(H2,29,30,31)/t17-,21+,23+,24+,27-,42-/m0/s1
|
| 化学名 |
2-ethylbutyl (2S)-2-[[[(2R,3S,4R,5R)-5-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-5-cyano-3,4-dihydroxyoxolan-2-yl]methoxy-phenoxyphosphoryl]amino]propanoate
|
| 别名 |
GS-5734; GS 5734; Prodrug of GS441524; Prodrug of GS441524; Remdesivir; GS5734; Prodrug of GS-441524; 3QKI37EEHE; GS 5734; GS 5734 [WHO-DD]; GS-5734; GS5734; Remdesivir; REMDESIVIR [INN];
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO: ~100 mg/mL (~166 mM)
Ethanol: ~100 mg/mL |
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (4.15 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.17 mg/mL (3.60 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 21.7 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.17 mg/mL (3.60 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 配方 4 中的溶解度: 5%DMSO+ 40%PEG300+ 5%Tween 80+ 50%ddH2O: 5.0mg/ml (8.30mM) 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6595 mL | 8.2977 mL | 16.5953 mL | |
| 5 mM | 0.3319 mL | 1.6595 mL | 3.3191 mL | |
| 10 mM | 0.1660 mL | 0.8298 mL | 1.6595 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
AZD7442 for Inpatients With COVID-19 (An ACTIV-3/TICO Treatment Trial)
CTID: NCT05780437
Phase: Phase 3 Status: Completed
Date: 2024-07-30
The F476L and V553L mutations mediate resistance to GS-5734 and are associated with a fitness defect.MBio.2018 Mar 6;9(2). pii: e00221-18. |
MHV resistance mutations confer resistance and are attenuated in SARS-CoV.MBio.2018 Mar 6;9(2). pii: e00221-18. |
Antiviral activity of GS-441524 and GS-5734 and modeled therapeutic efficacy of GS-5734 against SARS-CoV and MERS-CoV in HAE cultures.
GS-5734 acts at early times postinfection to decrease viral RNA levels. |
GS-441524 and GS-5734 inhibit MHV with minimal cytotoxicity.MBio.2018 Mar 6;9(2). pii: e00221-18. |
Viruses lacking ExoN-mediated proofreading are more sensitive to GS-5734 inhibition.MBio.2018 Mar 6;9(2). pii: e00221-18. |
Two mutations in the predicted fingers domain of the nsp12 RdRp, F476L and V553L, arose after 23 passages in the presence of GS-441524, and these residues are completely conserved across CoVs.MBio.2018 Mar 6;9(2). pii: e00221-18. |