| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 2mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| Other Sizes |
|
| 靶点 |
LXR[1]
Cancer immunotherapy is restricted to immune resistance caused by immunosuppressive tumor microenvironment. Pyroptosis involved in antitumor immunotherapy as a new schedule is prospective to reverse immunosuppression. Herein, acidic tumor microenvironment (TME)-evoked MRC nanoparticles (MRC NPs) co-delivering immune agonist RGX-104 and photosensitizer chlorine e6 (Ce6) are reported for pyroptosis-mediated immunotherapy. RGX-104 remodels TME by transcriptional activation of ApoE to regress myeloid-derived suppressor cells' (MDSCs) activity, which neatly creates foreshadowing for intensifying pyroptosis. Considering Ce6-triggered photodynamic therapy (PDT) can strengthen oxidative stress and organelles destruction to increase immunogenicity, immunomodulatory-photodynamic MRC nanodrugs will implement an aforementioned two-pronged strategy to enhance gasdermin E (GSDME)-dependent pyroptosis. RNA-seq analysis of MRC at the cellular level is introduced to first elucidate the intimate relationship between RGX-104 acting on LXR/ApoE axis and pyroptosis, where RGX-104 provides the prerequisite for pyroptosis participating in antitumor therapy. Briefly, MRC with favorable biocompatibility tackles the obstacle of hydrophobic drugs delivery, and becomes a powerful pyroptosis inducer to reinforce immune efficacy. MRC-elicited pyroptosis in combination with anti-PD-1 blockade therapy boosts immune response in solid tumors, successfully arresting invasive metastasis and extending survival based on remarkable antitumor immunity. MRC may initiate a new window for immuno-photo pyroptosis stimulators augmenting pyroptosis-based immunotherapy.[2] Adv Healthc Mater . 2022 Nov;11(21):e2201233. |
|---|---|
| 体外研究 (In Vitro) |
肿瘤免疫治疗仅限于免疫抑制肿瘤微环境引起的免疫抵抗。焦亡参与抗肿瘤免疫治疗作为一种新的方案,有望逆转免疫抑制。本文报道了酸性肿瘤微环境(TME)诱发的MRC纳米颗粒(MRC NPs)共同递送免疫激动剂RGX-104和光敏剂氯e6 (Ce6)用于焦热介导的免疫治疗。RGX-104通过转录激活ApoE来重塑TME,从而降低髓源性抑制细胞(MDSCs)的活性,这为强化焦亡埋下了伏笔。考虑到ce6触发的光动力疗法(PDT)可以增强氧化应激和细胞器破坏以增加免疫原性,免疫调节光动力MRC纳米药物将实施上述双管齐下的策略来增强气凝胶蛋白E (GSDME)依赖性焦亡。引入细胞水平MRC的RNA-seq分析,首次阐明作用于LXR/ApoE轴的RGX-104与焦亡之间的密切关系,其中RGX-104为焦亡参与抗肿瘤治疗提供了前提条件。简而言之,MRC具有良好的生物相容性,解决了疏水药物递送的障碍,成为一种强大的焦亡诱导剂,增强免疫功效。mrc诱导的焦亡联合抗pd -1阻断治疗可增强实体瘤的免疫应答,成功阻止侵袭性转移,并基于显著的抗肿瘤免疫延长生存期。MRC可能为免疫光热亡刺激剂增加基于热亡的免疫治疗开启一个新的窗口。[2]Adv Healthc Mater . 2022 Nov;11(21):e2201233.
在Transwell MDSC分化实验中,将骨髓细胞与B16F10黑色素瘤细胞和GM-CSF共培养6天,在第3天添加 RGX-104(2 µM)可显著减少第6天时Gr-1highCD11b+细胞(粒细胞样MDSC)的数量,效果与不添加癌细胞组相当。 在体外,用 RGX-104(1 µM 或 2 µM)处理从荷瘤小鼠分离的MDSC,可显著降低MDSC存活率并增加裂解caspase-3+ MDSC的比例,表明其诱导了细胞凋亡。 |
| 体内研究 (In Vivo) |
当给患有可见肿瘤的小鼠口服 RGX-104(每天 100 毫克/公斤)时,可以有效抑制多种癌症类型的生长。 RGX-104 和抗 PD-1 的组合被发现比单独使用任何一种药物更有效。值得注意的是,在抗 PD-1 治疗的基础上接受 RGX-104 治疗的小鼠具有良好的耐受性,并且没有表现出明显的损伤 [1]。
口服 RGX-104(50 mg/kg/天、100 mg/kg/天 via 饲料,或80 mg/kg/天 via 腹腔注射)能显著抑制多种同源和异种移植小鼠模型的肿瘤生长,包括B16F10黑色素瘤、GL261胶质母细胞瘤、Lewis肺癌(LLC)、MC38结肠癌以及免疫缺陷小鼠体内的人SKOV3卵巢癌。 RGX-104 治疗降低了荷瘤小鼠肿瘤、脾脏和外周血中粒细胞样(G-MDSC)和单核细胞样(M-MDSC)髓源性抑制细胞的数量。 RGX-104 引起的MDSC减少与肿瘤浸润细胞毒性T淋巴细胞(CTL)的活化增强相关,表现为IFN-γ+Granzyme B+ CD8+ T细胞和PD-1+ CD8+ T细胞频率升高。 在使用gp100特异性Pmel CD8+ T细胞的过继性T细胞治疗模型中,联合使用 RGX-104 增强了抗肿瘤活性并提高了小鼠生存率。 在B16F10黑色素瘤和LLC模型中,RGX-104 与抗PD-1抗体联合治疗显示出优于任一单药的治疗效果,即使在Gvax诱导的MDSC积累背景下也是如此。 在一项涉及癌症患者的1期临床试验中,口服 RGX-104 治疗(28天周期中,每天一次,连续21天)显著降低了6名患者中5人的循环粒细胞样MDSC水平(中位减少86%)和单核细胞样MDSC水平。 患者接受 RGX-104 治疗后,循环CD8+ T细胞(尤其是PD-1+ 亚群)的活化增加,通过GITR表达进行测定。 |
| 细胞实验 |
MDSC体外增殖试验[1]
骨髓源性抑制细胞如前所述从荷瘤小鼠脾组织中分离。10万个细胞在聚l -赖氨酸包被板上四次镀。以1uM Abequolixron (RGX-104)或DMSO为载体处理3小时后,用4% PFA固定细胞15分钟,染色前用1X PBS洗涤3次。兔单克隆抗ki67抗体(1:400稀释)在4℃下过夜。细胞用Alexa Fluor 488二抗(1:200稀释,Invitrogen)在室温下孵育1小时,用DAPI反染(1:100稀释),用延长金贴载。为了分析Ki67阳性细胞的百分比,使用蔡司Axio Imager荧光显微镜在20倍放大下对每个重复的5个视野进行成像。使用CellProfiler软件进行图像分析。 MDSC黏附试验[1] 骨髓来源的抑制细胞从荷瘤小鼠脾组织中分离出来。10万个细胞分三次镀在聚l -赖氨酸包被板上。细胞用1uM Abequolixron (RGX-104)或DMSO作为载体处理2小时,以300 rpm振荡30分钟。之后,用4% PFA固定细胞15分钟,用1X PBS洗涤3次,用DAPI反染,用延长金贴壁。为了进行分析,使用蔡司Axio Imager荧光显微镜对每个重复的10个视场进行20倍放大成像。使用CellProfiler软件确定剩余细胞的数量。 体外MDSC细胞凋亡测定[1] 从WT、LXRαβ−/−、ApoE−/−或LRP8−/−小鼠中分离小鼠脾脏,匀浆形成单细胞悬液。细胞用1X ACK裂解缓冲液裂解并清除红细胞。使用髓源性抑制细胞分离试剂盒从细胞悬浮液中分离MDSCs。将分离的MDSCs涂于载玻片上,按指定浓度和时间用Abequolixron (RGX-104)或小鼠重组ApoE处理。然后用Cleaved Caspase-3抗体对样品进行染色。 将骨髓细胞与 B16F10 黑色素瘤细胞和 GM-CSF 一起培养 6 天。第 3 天,将 RGX-104 (2 μM) 添加到培养物中。第 6 天通过流式细胞术评估每 50 mL 培养液中 Gr-1high CD11b+ 细胞的平均数量[1]。 体外MDSC凋亡实验:使用特异性分离试剂盒从荷瘤小鼠脾脏分离MDSC。将分离的MDSC铺板,并用指定浓度(如1 µM, 2 µM)和时间(如3小时, 6小时)的 RGX-104 或对照处理。然后固定细胞并用抗裂解caspase-3抗体染色,以量化凋亡细胞。 体外MDSC分化实验:从小鼠股骨分离骨髓细胞,培养于孔板底部。将B16F10黑色素瘤细胞置于Transwell插入物中。培养物中添加GM-CSF。第3天,加入 RGX-104(2 µM)或对照。第6天,收集基底室细胞,用CD11b和Gr-1染色,并通过流式细胞术分析以量化MDSC群体。 T细胞抑制实验:从初始小鼠分离CD8+ T细胞,用增殖染料标记,并与从对照或 RGX-104 处理的荷瘤小鼠分离的MDSC在CD3/CD28激活珠和IL-2存在下共培养。24小时后,通过流式细胞术评估T细胞活化(IFN-γ产生)和增殖(染料稀释)。 [1] |
| 动物实验 |
动物/疾病模型:将1×10⁶个SKOV3卵巢癌细胞注射到NOD SCID或RAG小鼠体内[1]。
剂量:100 mg/kg。 给药途径:每日口服,持续约60天。 实验结果:显著抑制肿瘤生长和进展。 小鼠[1]将B16F10癌细胞皮下注射到C57BL/6小鼠体内。当肿瘤体积生长至 5-10 mm³ 后,小鼠分别饲喂对照饲料、添加 GW3965 (100 mg/kg) 的饲料或添加 RGX-104 (100 mg/kg) 的饲料[1]。RGX-104 的给药方式有两种:一种是通过配制药物饲料(100 mg/kg/天或 50 mg/kg/天)给药;另一种是通过腹腔注射(80 mg/kg/天)给药,药物悬浮液由玉米油和乙醇(2.5% 体积比)组成,具体剂量见各图所示。对照组分别饲喂普通饲料(Purina 5001)或由玉米油和乙醇(2.5% 体积比)组成的悬浮液。在实验过程中,于指定日期使用游标卡尺测量肿瘤体积。为进行生存分析,当肿瘤总负荷接近 IACUC 指南规定的标准(肿瘤体积超过 1500 mm³)时,对小鼠实施安乐死。在相关实验中,于肿瘤注射后第3、6和9天腹腔注射抗PD-1单克隆抗体(克隆RMP1-14)或同型对照抗体,剂量为10mg/kg。Gvax的制备方法如前所述,并在实验期间按指示高频(每3天)给药。[1] 在大多数肿瘤生长研究中,将癌细胞悬浮于PBS中,与Matrigel按1:1比例混合,并皮下注射到6-8周龄小鼠的侧腹部。一旦肿瘤达到特定体积(例如,5-10 mm³、30-40 mm³),将小鼠随机分组。 RGX-104通过药物配方饲料口服给药,剂量为50 mg/kg/天或100 mg/kg/天。在某些实验中,药物以玉米油和2.5%(体积比)乙醇的混合液为载体,通过腹腔注射给药(80 mg/kg/天)。 对照组接受普通饲料或玉米油/乙醇混合液,均通过腹腔注射给药。 定期使用游标卡尺测量肿瘤尺寸(长度和宽度),并计算体积。 对于与抗PD-1抗体的联合治疗,在指定日期(例如,肿瘤注射后第3、6和9天)以10 mg/kg的剂量腹腔注射抗体。 对于过继性T细胞治疗实验,从Pmel-1 TCR转基因小鼠中分离CD8+ T细胞,并通过眼眶后注射转移到荷瘤受体小鼠体内,受体小鼠同时接受gp100肽疫苗。 |
| 毒性/毒理 (Toxicokinetics/TK) |
在 I 期临床试验中,前六名可评估患者对 RGX-104 的耐受性良好,未观察到剂量限制性毒性。治疗的前两周内,关键血液学参数(白细胞总数、中性粒细胞绝对计数、淋巴细胞绝对计数和单核细胞绝对计数)未发生显著变化,这些变化在 4 周的治疗周期内逐渐恢复正常。
|
| 参考文献 | |
| 其他信息 |
阿贝奎利松是一种口服生物利用度高的核受体肝X受体β(LXRβ;NR1H2;LXR-b)激动剂,具有潜在的免疫调节和抗肿瘤活性。口服后,阿贝奎利松选择性地靶向并结合LXRβ,从而激活LXRβ介导的信号通路,导致某些抑癌基因的转录和某些促癌基因的下调。这尤其能激活肿瘤细胞和某些免疫细胞中抑癌蛋白载脂蛋白E(ApoE)的表达。进而激活先天免疫系统,导致肿瘤微环境中免疫抑制性髓源性抑制细胞(MDSC)、肿瘤细胞和内皮细胞的耗竭。这逆转了肿瘤的免疫逃逸,增强了抗肿瘤免疫反应,并抑制了肿瘤细胞的增殖。 LXRβ是氧固醇受体家族的成员,属于核受体转录因子家族,在胆固醇转运、葡萄糖代谢和炎症反应调节中发挥关键作用;LXRβ的激活可抑制多种肿瘤细胞类型的侵袭、血管生成、肿瘤进展和转移。在人类癌症中,随着肿瘤的生长、侵袭和转移,ApoE蛋白的表达会逐渐沉默;ApoE的沉默与癌症患者生存期的缩短相关。LXR-ApoE通路调节肿瘤逃避免疫系统和募集血管的能力。
RGX-104是一种在研的LXRβ激动剂,目前正在进行一项针对晚期实体瘤或淋巴瘤患者的多中心I期a/b临床试验(NCT02922764)。 其抗肿瘤和免疫调节作用是通过激活LXR/ApoE轴实现的。 LXR激活可转录上调ApoE,进而通过LRP8受体促进MDSC凋亡,从而降低先天免疫抑制并增强细胞毒性T细胞活性。 该研究提出RGX-104作为一种首创的靶向MDSC并逆转癌症免疫逃逸的治疗策略。 |
| 分子式 |
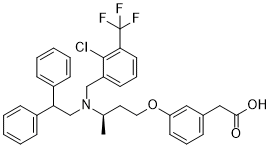
C34H33CLF3NO3
|
|
|---|---|---|
| 分子量 |
596.078939199448
|
|
| 精确质量 |
595.21
|
|
| 元素分析 |
C, 68.51; H, 5.58; Cl, 5.95; F, 9.56; N, 2.35; O, 8.05
|
|
| CAS号 |
610318-54-2
|
|
| 相关CAS号 |
RGX-104 hydrochloride;610318-03-1
|
|
| PubChem CID |
10218693
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| LogP |
6.3
|
|
| tPSA |
49.8Ų
|
|
| 氢键供体(HBD)数目 |
1
|
|
| 氢键受体(HBA)数目 |
7
|
|
| 可旋转键数目(RBC) |
13
|
|
| 重原子数目 |
42
|
|
| 分子复杂度/Complexity |
783
|
|
| 定义原子立体中心数目 |
1
|
|
| SMILES |
C[C@H](CCOC1=CC=CC(=C1)CC(=O)O)N(CC2=C(C(=CC=C2)C(F)(F)F)Cl)CC(C3=CC=CC=C3)C4=CC=CC=C4
|
|
| InChi Key |
ZLJZDYOBXVOTSA-XMMPIXPASA-N
|
|
| InChi Code |
InChI=1S/C34H33ClF3NO3/c1-24(18-19-42-29-16-8-10-25(20-29)21-32(40)41)39(22-28-15-9-17-31(33(28)35)34(36,37)38)23-30(26-11-4-2-5-12-26)27-13-6-3-7-14-27/h2-17,20,24,30H,18-19,21-23H2,1H3,(H,40,41)/t24-/m1/s1
|
|
| 化学名 |
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 本产品在运输和储存过程中需避光。 |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.17 mg/mL (3.64 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 21.7 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: 2.08 mg/mL (3.49 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 悬浊液; 超声助溶。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 0.83 mg/mL (1.39 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6776 mL | 8.3881 mL | 16.7763 mL | |
| 5 mM | 0.3355 mL | 1.6776 mL | 3.3553 mL | |
| 10 mM | 0.1678 mL | 0.8388 mL | 1.6776 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
LXR Agonist Treatment Robustly Suppresses Tumor Growth and Progression across a Broad Set of Mouse and Human Tumors.Cell.2018 Feb 8;172(4):825-840.e18. |
|---|
 LXR Agonism Reduces Tumor-Infiltrating and Systemic Myeloid-Derived Suppressor Cells.Cell.2018 Feb 8;172(4):825-840.e18. |
|