| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 2mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|

| 靶点 |
CDK4 (IC50 = 10 nM); CDK6 (IC50 = 39 nM)
The target of Ribociclib (LEE011) is cyclin-dependent kinase 4 (CDK4) and cyclin-dependent kinase 6 (CDK6). It inhibits CDK4 with an IC50 of 10 nM and CDK6 with an IC50 of 39 nM [1] [2] Ribociclib (LEE011) specifically targets cyclin-dependent kinase 4 (CDK4) and cyclin-dependent kinase 6 (CDK6); the IC50 value for CDK4/cyclin D1 complex is 10 nM, and for CDK6/cyclin D3 complex is 19 nM [2] Ribociclib (LEE011) has extremely weak inhibitory effects on other CDK isoforms such as CDK1/cyclin B and CDK2/cyclin E (IC50>1000 nM), showing high target selectivity [1] |
|---|---|
| 体外研究 (In Vitro) |
LEE011 作为 CDK4/CDK6 双重抑制剂,显着抑制 17 种神经母细胞瘤细胞系中 12 种的生长,平均 IC50 为 307 nM。神经母细胞瘤细胞系的生长抑制主要是细胞抑制作用,由 G1 细胞周期停滞和细胞衰老介导。激酶测定:Ribociclib(以前也称为 LEE011、NVP-LEE011;商品名:Kisqali)是一种有效的、口服的、高度特异性的 CDK4/6(细胞周期蛋白依赖性激酶)抑制剂,IC50 分别为 10 nM 和 39 nM 。截至2017年3月,Ribociclib被FDA批准用于治疗患有一种晚期乳腺癌的绝经后妇女。 Ribociclib 通过减少磷酸化 RB 和 FOXM1 发挥作用。当用 17 种人类神经母细胞瘤细胞系进行测试时,其中 12 种对 Ribociclib 治疗敏感,平均 IC50=306±68 NM。 Ribociclib 治疗可以通过阻止细胞周期 G0-G1 来显着降低细胞生长。在 17 种人类神经母细胞瘤衍生细胞系中的 12 种中,LEE011 治疗可显着减少细胞增殖。细胞测定:根据先前的基质贴壁生长演示选择一组神经母细胞瘤细胞系,将其一式三份铺板在 Xcelligence 实时细胞电子传感系统上,并在 24 小时后用四对数剂量范围的抑制剂或二甲基亚砜 (DMSO) 对照。连续监测细胞指数约 100 小时,IC50 值确定如下:通过将细胞指数绘制为时间函数来生成生长曲线,并将其归一化为治疗时的细胞指数,基线细胞指数为1. 然后使用基线面积 1(治疗时的细胞指数)计算从治疗时到治疗后 96 小时的标准化生长曲线下的面积。将面积标准化为 DMSO 对照,并使用非线性对数抑制剂与标准化响应函数来分析所得数据。所有实验至少重复一次。
瑞博西尼(Ribociclib, LEE011)在具有完整视网膜母细胞瘤(Rb)蛋白的癌细胞中诱导G1期细胞周期阻滞。在表达功能性Rb的神经母细胞瘤细胞系中,用0.1-1 μM的瑞博西尼(Ribociclib, LEE011)处理后,通过流式细胞术检测显示G1期细胞比例显著增加。western blot和qPCR分析表明,这伴随着Rb磷酸化(pRb)水平降低和E2F靶基因表达减少 [2] 在神经母细胞瘤细胞中,用瑞博西尼(Ribociclib, LEE011)(0.5 μM)长期处理7天可诱导细胞衰老,表现为衰老相关β-半乳糖苷酶(SA-β-gal)活性增加和衰老标志物(如p16INK4a)上调 [2] 该化合物具有选择性抗增殖活性,对Rb阳性细胞的 efficacy 更强,而对Rb阴性细胞的IC50显著更高(> 10 μM) [1] [2] Ribociclib (LEE011) 对神经母细胞瘤细胞系具有显著抗增殖活性:SK-N-SH细胞IC50=45 nM,IMR-32细胞IC50=38 nM,LAN-5细胞IC50=52 nM;处理48小时后诱导细胞周期停滞于G1期,G1期细胞比例从35%升至68%,伴随视网膜母细胞瘤蛋白(Rb)磷酸化水平显著下调(降低70%)[2] Ribociclib (LEE011) 可诱导神经母细胞瘤细胞衰老,100 nM浓度处理7天后,SA-β-半乳糖苷酶(衰老标志物)阳性细胞比例从8%升至56%;同时上调p21和p16蛋白表达(分别为对照组的3.2倍和2.8倍)[2] Ribociclib (LEE011) 对激素受体阳性乳腺癌细胞系MCF-7的IC50=22 nM,与来曲唑联合使用时,抗增殖活性协同增强,联合指数(CI)=0.53 [1] |
| 体内研究 (In Vivo) |
LEE011(每日 200 mg/kg,口服)显着导致携带 BE2C 或 1643 异种移植物的小鼠肿瘤生长延迟,且没有体重减轻或其他毒性迹象。
Ribociclib (LEE011)对CDK4/6的抑制导致体内肿瘤生长延迟[2] 鉴于观察到神经母细胞瘤细胞系对CDK4/6抑制的不同敏感性,我们使用神经母细胞癌细胞系衍生的异种移植物来检测体内疗效,这些异种移植物代表了体外敏感性的极端。携带BE2C、NB-1643(MYCN扩增,体外敏感)或EBC1(非扩增,体外耐药)异种移植物的CB17免疫缺陷小鼠每天用Ribociclib(LEE011)或载体对照治疗21天。这种给药策略具有良好的耐受性,因为在任何异种移植物模型中都没有观察到体重减轻或其他毒性迹象。如图5A和S6所示,在含有BE2C或1643异种移植物的小鼠中,肿瘤生长在整个21天的治疗过程中明显延迟(两者都有,p<0.0001),尽管治疗后生长恢复(数据未显示)。相比之下,正如体外数据所预期的那样,EBC1异种移植物模型中的肿瘤生长抑制效果较差(p=0.51)。通过免疫组织化学对Ki67增殖标志物的评估证实,仅在BE2C和1643异种移植物模型中增殖受损,因为从单独的BE2C或1643异种移植小鼠队列中切除的肿瘤在用Ribociclib(LEE011)治疗7天后显示出比载体对照相对较弱的染色,而在EBC1异种移植物中没有观察到Ki67染色差异(图5B)。在BE2C和1643异种移植物中,RB的磷酸化也显著减少,而在EBC1模型中仅检测到最小的减少(图5B和5C)[2]。 在神经母细胞瘤(Rb阳性)异种移植小鼠模型中,每日口服瑞博西尼(Ribociclib, LEE011) 150 mg/kg,持续21天,可显著抑制肿瘤生长,与溶媒处理对照组相比,肿瘤体积减少60-70%。处理组小鼠的肿瘤样本中pRb水平降低且SA-β-gal活性增加,证实了体内G1期阻滞和衰老诱导 [2] Ribociclib (LEE011) 以40 mg/kg剂量口服给药,每日1次,持续21天,可显著抑制裸鼠SK-N-SH神经母细胞瘤移植瘤生长,肿瘤体积抑制率达67%,肿瘤重量抑制率达63%;肿瘤组织中Rb磷酸化水平降低65%,p21蛋白表达上调 [2] Ribociclib (LEE011) 口服给药(60 mg/kg,每日1次,持续28天)联合来曲唑(1 mg/kg,每周1次),对ER阳性乳腺癌PDX模型的肿瘤生长抑制率达83%,显著高于单药组(来曲唑32%,Ribociclib 59%)[1] |
| 酶活实验 |
Ribociclib 是一种强效、口服、高选择性的 CDK4/6(细胞周期蛋白依赖性激酶)抑制剂,IC50 分别为 10 nM 和 39 nM,之前称为 LEE011、NVP-LEE011;商品名:Kisqali。 2017 年 3 月,FDA 批准 Ribociclib 用于治疗患有晚期乳腺癌的绝经后妇女。 Ribociclib 通过降低磷酸化 FOXM1 和 RB 的水平发挥作用。在测试的 17 个人类神经母细胞瘤细胞系中,有 12 个显示出对 ribofacilb 治疗的敏感性(平均 IC50=306±68 NM)。通过停止 G0-G1 细胞周期,ribociclib 治疗可能会显着降低细胞增殖率。 LEE011 治疗可显着抑制 17 种人类神经母细胞瘤来源细胞系中 12 种的细胞增殖。
为测量CDK4/6抑制活性,将重组CDK4/细胞周期蛋白D1和CDK6/细胞周期蛋白D3复合物与荧光肽底物及不同浓度的瑞博西尼(Ribociclib, LEE011)共同孵育。通过测量底物的磷酸化水平评估激酶活性,IC50定义为使激酶活性降低50%所需的浓度 [1] [2] 制备重组CDK4/ cyclin D1和CDK6/ cyclin D3激酶复合物,将梯度浓度的Ribociclib (LEE011)与激酶复合物、ATP底物及生物素化特异性肽段混合,37℃孵育60分钟;加入链霉亲和素包被的微孔板结合生物素化肽段,再加入抗磷酸化肽段抗体和酶标二抗,通过比色法检测吸光度值,计算激酶活性抑制率及IC50值 [2] 采用均相时间分辨荧光(HTRF)法验证靶点特异性:将Ribociclib (LEE011)与CDK1/ cyclin B、CDK2/ cyclin E等重组复合物孵育,30℃反应45分钟后检测荧光信号,确认其对非靶点CDK的抑制活性微弱 [1] |
| 细胞实验 |
在 35 mm 平板中,细胞生长 24 小时,然后用 500 nM Ribociclib 处理 6 天。然后固定细胞,并进行过夜染色。然后,使用 Axio Observer D.1 相差显微镜对细胞进行 SA-β-gal 成像。通过计数三个不同显微镜框架中的阳性细胞数并归一化至对照,可以计算出 SA-β-gal 阳性细胞的百分比。为了评估细胞凋亡活性,用 Ribociclib 处理细胞,一式三份铺板于 96 孔板中,然后 16 小时后,在 Caspase-Glo 3/7 处理后 16 小时测量 caspase 3/7 活化。使用 SN-38 处理的细胞作为阳性对照[2]。
为进行细胞周期分析,用瑞博西尼(Ribociclib, LEE011)(0.1-1 μM)处理神经母细胞瘤细胞24-72小时。用碘化丙啶染色细胞,通过流式细胞术分析细胞周期分布,以量化G1、S和G2/M期的细胞比例 [2] 为评估衰老,用瑞博西尼(Ribociclib, LEE011)(0.5 μM)处理细胞7天,然后用比色法染色SA-β-gal活性,在显微镜下计数阳性细胞。通过western blot检测衰老标志物(如p16INK4a)的变化 [2] 为进行抗增殖实验,用0.01-10 μM的瑞博西尼(Ribociclib, LEE011)处理细胞5天。通过比色法测量细胞活力并确定IC50值 [1] [2] 神经母细胞瘤细胞接种于96孔板(5×10³个/孔),培养24小时后加入0.01-10 μM梯度浓度的Ribociclib (LEE011),继续培养72小时;采用CellTiter-Glo发光法检测细胞活力,拟合曲线计算IC50值 [2] 细胞经Ribociclib (LEE011)(100 nM)处理48小时后,收集细胞并固定,加入PI染色液室温孵育30分钟,流式细胞仪分析细胞周期分布;提取细胞总蛋白,Western blot检测Rb、磷酸化Rb、p21、p16等蛋白表达 [2] 细胞经药物处理7天后,采用SA-β-半乳糖苷酶染色试剂盒进行染色,光学显微镜下计数蓝色阳性细胞,计算衰老细胞比例 [2] MCF-7细胞接种后培养24小时,分别加入Ribociclib (LEE011)、来曲唑及两者联合用药,培养72小时后采用MTT法检测细胞存活率,计算联合指数评估协同作用 [1] |
| 动物实验 |
Mice: The xenografts derived from BE2C, NB-1643, or EBC1 cell lines are subcutaneously implanted into the right flank of CB17 SCID -/- mice. Then, for a total of 21 days, animals with engrafted tumors measuring 200–600 mm 3 are randomly assigned to receive oral treatment with 200 mg/kg Ribociclib in 0.5% methylcellulose (n = 10) or vehicle (n = 10). Throughout the course of treatment, the tumor burden is calculated on a regular basis using the formula (π/6)×d 2 , where d is the mean tumor diameter measured with a caliper.
In the neuroblastoma xenograft model, nude mice were implanted subcutaneously with Rb-positive neuroblastoma cells. Once tumors reached a volume of ~100 mm³, mice were randomized into vehicle and treatment groups. Ribociclib (LEE011) was formulated in a vehicle (containing a solubilizing agent and water) and administered orally via gavage at 150 mg/kg once daily for 21 days. Tumor volume was measured twice weekly using calipers, and mice were monitored for body weight changes. At the end of the study, tumors were harvested for histopathological and molecular analyses [2] Female nude mice (6-8 weeks old) were subcutaneously inoculated with SK-N-SH cell suspension (2×10⁶ cells/mouse) on the right back. Drug administration started when the tumor volume reached 100-150 mm³; Ribociclib (LEE011) was dissolved in normal saline containing 0.5% hydroxypropyl methylcellulose and 0.1% Tween 80, and administered orally at a dose of 40 mg/kg once daily for 21 days; tumor volume and mouse weight were measured every 3 days, and tumors were excised and weighed at the end of the experiment to detect the expression of related proteins [2] ER-positive breast cancer PDX model mice (6-8 weeks old) were divided into control group, letrozole monotherapy group, Ribociclib (LEE011) monotherapy group, and combination therapy group; letrozole was administered orally at 1 mg/kg once weekly, and Ribociclib (LEE011) was administered orally at 60 mg/kg once daily for 28 days; the tumor growth inhibition rate was calculated at the end of the experiment, and the phosphorylation level of Rb in tumor tissues was detected [1] |
| 药代性质 (ADME/PK) |
吸收、分布和排泄
瑞博西尼是一种口服生物利用度高、选择性强的 CDK4/6 激酶抑制剂,其抑制 IC50 浓度在低纳摩尔范围内。口服给药后,瑞博西尼迅速吸收,中位达峰时间 (Tmax) 为 1 至 5 小时。由于药物蓄积,血浆浓度从第 1 周期第 1 天到第 1 周期第 18/21 天增加了约 2 至 3 倍,根据每日重复给药后的谷浓度,大约在第 8 天达到稳态。剂量比例分析表明,瑞博西尼的暴露量随剂量增加而增加,在 50–1200 mg/天的剂量范围内,Cmax 和曲线下面积 (AUC) 的增加略高于剂量比例。 生物半衰期 32.6 小时 |
| 毒性/毒理 (Toxicokinetics/TK) |
肝毒性
在大型临床试验中,不良事件较为常见,导致45%的患者剂量减少,7%的患者停药。在注册前临床试验中,利博西尼组46%的受试者出现ALT升高,而对照组为36%;ALT升高超过正常值上限5倍的患者比例分别为10%和1%。在一项研究中,1%的受试者出现临床表现明显的肝损伤并伴有黄疸,但所有患者均康复。肝损伤发生在3至5个疗程后,表现为无症状的血清ALT升高,随后出现症状和黄疸。虽然肝脏组织学检查有时显示自身免疫性肝炎样特征,但未发现免疫过敏和自身免疫特征。恢复缓慢(3至5个月),但最终完全恢复。重新开始使用利博西尼会导致更快速、更严重的复发。因此,关于瑞博西尼的经验有限,但它似乎能够引起严重的肝损伤。 可能性评分:C(可能导致临床上明显的肝损伤)。 妊娠和哺乳期影响 ◉ 哺乳期用药概述 目前尚无关于瑞博西尼在哺乳期临床应用的信息。由于瑞博西尼的蛋白结合率为70%,因此具有临床意义的药物剂量可能会进入母乳。制造商建议在接受瑞博西尼治疗期间以及末次给药后至少3周内停止母乳喂养。 ◉ 对母乳喂养婴儿的影响 截至修订日期,未找到相关的已发表信息。 ◉ 对泌乳和母乳的影响 截至修订日期,未找到相关的已发表信息。 在体外实验中,当浓度≤1 μM时,瑞博西尼 (LEE011) 对正常人成纤维细胞 (NHDF) 的存活率无显著影响(存活率≥90%)[2]。 当以60 mg/kg的剂量口服瑞博西尼 (LEE011)(每日一次,持续28天)时,PDX模型小鼠未观察到明显的体重减轻(体重变化≤±7%),且血清ALT、AST、BUN和Cr水平未发生变化。与对照组相比,统计学上存在显著差异[1] 瑞博西尼(LEE011)的人血浆蛋白结合率为93%±2%[2] |
| 参考文献 | |
| 其他信息 |
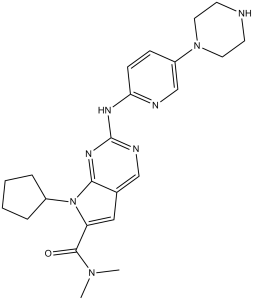
瑞博西尼属于哌嗪类和吡啶类化合物。
瑞博西尼是一种选择性细胞周期蛋白依赖性激酶抑制剂,这类药物通过抑制两种名为细胞周期蛋白依赖性激酶4和6 (CDK4/6) 的蛋白质来减缓癌症的进展。这些蛋白质过度激活时,会导致癌细胞生长和分裂过快。更精准地靶向CDK4/6可能有助于防止癌细胞继续不受控制地复制。瑞博西尼于2017年3月获得美国FDA批准,商品名为Kisqali。 瑞博西尼是一种激酶抑制剂。瑞博西尼的作用机制是作为激酶抑制剂和细胞色素P450 3A抑制剂。 瑞博西尼是一种独特的细胞周期蛋白依赖性激酶抑制剂,与芳香化酶抑制剂联合用于治疗绝经后转移性乳腺癌患者。瑞博西尼治疗期间血清转氨酶升高发生率中等,部分患者会出现临床上明显的肝损伤。 瑞博西尼是一种口服的细胞周期蛋白依赖性激酶(CDK)抑制剂,靶向细胞周期蛋白D1/CDK4和细胞周期蛋白D3/CDK6通路,具有潜在的抗肿瘤活性。瑞博西尼特异性抑制CDK4和CDK6,从而抑制视网膜母细胞瘤(Rb)蛋白的磷酸化。抑制Rb磷酸化可阻止CDK介导的G1-S期转换,从而使细胞周期停滞在G1期,抑制DNA合成并抑制癌细胞生长。某些癌症中可见的CDK4/6过度表达会导致细胞周期失调。 另见:琥珀酸瑞博西尼(活性成分)。 药物适应症 Kisqali(瑞博西尼)是一种选择性细胞周期蛋白依赖性激酶抑制剂,这类药物通过抑制两种名为细胞周期蛋白依赖性激酶4和6(CDK4/6)的蛋白质来帮助减缓癌症进展。这些蛋白质过度激活时,会导致癌细胞生长和分裂过快。更精准地靶向 CDK4/6 可能有助于防止癌细胞不受控制地持续复制。 Kisqali 适用于治疗激素受体 (HR) 阳性、人表皮生长因子受体 2 (HER2) 阴性的局部晚期或转移性乳腺癌女性患者,需与芳香化酶抑制剂或氟维司群联合作为初始内分泌治疗,或用于既往接受过内分泌治疗的女性患者。对于绝经前或围绝经期女性患者,内分泌治疗应与促黄体生成素释放激素 (LHRH) 激动剂联合使用。 神经母细胞瘤的治疗 作用机制 抑制细胞周期蛋白依赖性激酶 4 和 6 (CDK4/6) 可能对特定组织类型的致癌过程提供保护作用。例如,基因敲除小鼠研究表明,CDK4并非正常乳腺组织发育所必需,但却是Ras诱导的乳腺肿瘤生长所必需的,这提示存在一个潜在的低毒性治疗窗口。据报道,Ribociclib是一种选择性极高的CDK4/6抑制剂,并在多个临床前模型中表现出剂量依赖性的抗肿瘤活性。它通过将细胞阻滞在G1期检查点来抑制肿瘤细胞的生长,从而阻止肿瘤细胞增殖。 Ribociclib (LEE011)是一种选择性CDK4/6抑制剂,可阻断细胞周期蛋白D-CDK4/6-Rb通路,该通路是细胞周期从G1期到S期进程的关键调控因子。其活性依赖于功能性Rb蛋白,因此对Rb信号通路完整的癌症(例如乳腺癌、神经母细胞瘤)有效。它旨在诱导细胞周期阻滞和衰老,从而抑制癌细胞增殖[1][2] Ribociclib (LEE011)是一种高选择性口服活性 CDK4/6 抑制剂,它通过抑制 CDK4/6 与细胞周期蛋白 D 的结合、阻断 Rb 蛋白磷酸化以及将细胞周期阻滞在 G1 期来抑制肿瘤细胞增殖并诱导衰老[1] Ribociclib (LEE011)对 Rb 蛋白阳性肿瘤细胞更敏感,对 Rb 阴性细胞系(例如 SK-N-AS)的 IC50>1000 nM,表明 Rb 状态可作为其疗效的预测标志物[2] Ribociclib (LEE011)已获批用于治疗激素受体阳性、人表皮生长因子受体 2 阳性的癌症。用于治疗HER2阴性晚期或转移性乳腺癌,通常与芳香化酶抑制剂联合使用[1] |
| 分子式 |
C23H30N8O
|
|---|---|
| 分子量 |
434.54
|
| 精确质量 |
434.254
|
| 元素分析 |
C, 63.57; H, 6.96; N, 25.79; O, 3.68
|
| CAS号 |
1211441-98-3
|
| 相关CAS号 |
Ribociclib hydrochloride;1211443-80-9;Ribociclib-d6 hydrochloride;Ribociclib succinate;1374639-75-4;Ribociclib succinate hydrate;1374639-79-8;Ribociclib-d6;1328934-40-2;Ribociclib-d8;2167898-24-8
|
| PubChem CID |
44631912
|
| 外观&性状 |
Yellow solid powder
|
| 密度 |
1.4±0.1 g/cm3
|
| 沸点 |
730.8±70.0 °C at 760 mmHg
|
| 闪点 |
395.8±35.7 °C
|
| 蒸汽压 |
0.0±2.4 mmHg at 25°C
|
| 折射率 |
1.723
|
| LogP |
-0.74
|
| tPSA |
91.21
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
7
|
| 可旋转键数目(RBC) |
5
|
| 重原子数目 |
32
|
| 分子复杂度/Complexity |
636
|
| 定义原子立体中心数目 |
0
|
| SMILES |
N1(CCNCC1)C1C=NC(NC2N=C3N(C(C(N(C)C)=O)=CC3=CN=2)C2CCCC2)=CC=1
|
| InChi Key |
RHXHGRAEPCAFML-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C23H30N8O/c1-29(2)22(32)19-13-16-14-26-23(28-21(16)31(19)17-5-3-4-6-17)27-20-8-7-18(15-25-20)30-11-9-24-10-12-30/h7-8,13-15,17,24H,3-6,9-12H2,1-2H3,(H,25,26,27,28)
|
| 化学名 |
7-cyclopentyl-N,N-dimethyl-2-[(5-piperazin-1-ylpyridin-2-yl)amino]pyrrolo[2,3-d]pyrimidine-6-carboxamide
|
| 别名 |
LEE 011; Ribociclib; LEE011; LEE-011; trade name: Kisqali; Ribociclib (LEE011); LEE 011; 7-cyclopentyl-N,N-dimethyl-2-((5-(piperazin-1-yl)pyridin-2-yl)amino)-7H-pyrrolo[2,3-d]pyrimidine-6-carboxamide;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 1 mg/mL (2.30 mM) (饱和度未知) in 5% DMSO + 40% PEG300 + 5% Tween80 + 50% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
*生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 1 mg/mL (2.30 mM) (饱和度未知) in 5% DMSO + 95% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 0.89 mg/mL (2.05 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 配方 4 中的溶解度: ≥ 0.89 mg/mL (2.05 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将100 μL 8.9 mg/mL 澄清 DMSO 储备液加入 900 μL 20% SBE-β-CD 生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 配方 5 中的溶解度: ≥ 0.89 mg/mL (2.05 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将100 μL 8.9 mg/mL 澄清 DMSO 储备液加入900 μL 玉米油中,混合均匀。 配方 6 中的溶解度: 5% DMSO+40% PEG 300+5%Tween80+ 50%ddH2O: 1.1mg/ml 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3013 mL | 11.5064 mL | 23.0128 mL | |
| 5 mM | 0.4603 mL | 2.3013 mL | 4.6026 mL | |
| 10 mM | 0.2301 mL | 1.1506 mL | 2.3013 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
First-in-Human Study of STX-478 as Monotherapy and in Combination With Other Antineoplastic Agents in Participants With Advanced Solid Tumors
CTID: NCT05768139
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2024-11-25
 Pharmacologic inhibition of CDK4/6 suppresses neuroblastoma growthin vitro.(A)The growth of 12 of 17 neuroblastoma cell lines was significantly impaired in response to CDK4/6 inhibition with LEE011 (mean IC50= 306 ± 68 nM, sensitive lines only). Data are plotted (and tabulated) as the best fit IC50per log(inhibitor) vs. normalized response analysis (GraphPad); upper and lower bars represent 95 % confidence levels.(B)Dose-dependent decreases in pRBS780accompany growth suppression in sensitive lines and are indicative of on- target activity.Clin Cancer Res.2013 Nov 15;19(22):6173-82. |
|---|
 Growth suppression via CDK4/6 inhibition is mediated by cell cycle arrest and senescence. Neuroblastoma cell lines with demonstrated sensitivity or resistance to LEE011 were analyzed for cell cycle arrest and senescence associated β-galactosidase (SA-β-gal) activity.(A)A significant G1arrest accompanied by reductions in the fraction of cells in S phase and G2/M was observed in sensitive lines only.(B)Representative cell cycle histograms of a sensitive and resistant cell line.(C)Down-regulation of FOXM1 mRNA and(D)protein was observed in sensitive lines and was associated with(E)the induction of a senescent phenotype.Clin Cancer Res.2013 Nov 15;19(22):6173-82. |
 Inhibition of CDK4/6 suppresses neuroblastoma growthin vivo.(A)Mice with subcutaneously implanted xenografts were treated daily with 200 mg/kg LEE011 or with a vehicle for 21 days. In two of three neuroblastoma xenograft models, treatment with LEE011 significantly reduced tumor burden in comparison to vehicle, as determined by linear mixed effects analysis (BE2C, p<0.0001; 1643, p <0.0001; EBC1 p = 0.51).(B)The reduction in tumor proliferation observed in sensitive lines was confirmed by Ki67 staining of resected xenografts, and inhibition of CDK4/6 activity was confirmed by(C)immunohistochemical staining and western blot for pRBS780.Clin Cancer Res.2013 Nov 15;19(22):6173-82. |