| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
PARP-1 ( Ki = 1.4 nM ); PARP-2; PARP-3
In vitro activity: Rucaparib (AG014699) camsylate is a possible N-demethylation metabolite of AG14644[1]. Rucaparib (0.1, 1, 10, 100 μM; 24 hours) camsylate is cytotoxic; in Capan-1 (BRCA2 mutant) cells, its LC50 is 5 μM, while in MX-1 (BRCA1 mutant) cells, it is only 100 nM[2]. Rucaparib camsylate induces radiosensitization independent of SSB repair inhibition, as it results from downstream inhibition of NF-κB activation. Without impairing other essential inflammatory functions, rucaparib camsylate can target NF-κB that is activated by DNA damage and overcome the toxicity seen with classical NF-κB inhibitors[5]. Rucaparib camsylate inhibits PARP-1 activity in permeabilized D283Med cells by 97.1% at a concentration of 1 μM[6]. |
|---|---|
| 体外研究 (In Vitro) |
体外活性:Rucaparib 是纯化的全长人 PARP-1 的有效抑制剂,并且对 LoVo 和 SW620 细胞中的细胞 PARP 显示出更高的抑制作用。此外,Rucaparib 可检测地与其他 8 个 PARP 结构域结合,包括 PARP2、3、4、10、15、16、TNKS1 和 TNKS2。 Rucaparib 的放射增敏作用是由于下游 NF-κB 激活的抑制,并且与 SSB 修复抑制无关。 Rucaparib 可以靶向由 DNA 损伤激活的 NF-κB,并克服经典 NF-κB 抑制剂观察到的毒性,而不损害其他重要的炎症功能。在透化的 D283Med 细胞中,浓度为 1 μM 时,Rucaparib 将 PARP-1 活性抑制 97.1%。 激酶测定:测量 [32P]NAD+ 掺入对人全长重组 PARP-1 的抑制。使用 PhosphorImager 对掺入酸不溶性材料中的 [32P]ADP-核糖进行定量。通过非线性回归分析计算Ki。 细胞测定:在过表达人(h)ABCB1的MDCKII亲本细胞系中,rucaparib的顶部和基底外侧定向易位是相同的。用 ABCB1 抑制剂 zosuquidar 处理细胞导致顶端定向转运略有减少,这可能是由于基底外侧未识别的 rucaparib 摄取转运蛋白的特异性抑制,或内源性犬 ABCB1 的抑制。结果表明rucaparib是ABCB1的转运底物。
Rucaparib进入SW620人结肠癌细胞的过程是载体介导的,并遵循米氏动力学(Km = 8.4 ± 1.2 µM,Vmax = 469 ± 22 pmol per 10^6 cells per 10 min)。[2] Rucaparib逆浓度梯度在细胞内蓄积,暴露于400 nM药物30分钟内,细胞内稳态水平可达到细胞外浓度的10倍以上。在30分钟脉冲给药后,外排过程是双相的,初始快速流失后是极慢的外排;移除药物2小时后,细胞内浓度仍比原始细胞外水平高10倍以上。24小时内,细胞内浓度呈指数下降的半衰期约为20分钟。[2] 400 nM rucaparib处理30分钟,可在SW620、Capan-1 (BRCA2突变) 和MX-1 (BRCA1突变) 细胞中导致持续的PARP抑制(≥50%抑制至少72小时)。在Capan-1和MX-1细胞中,移除药物1周后,PARP活性仍低于基线水平。[2] Rucaparib对同源重组缺陷细胞具有细胞毒性。暴露24小时后,使集落形成减少50%的浓度(LC50)在Capan-1细胞中为5 µM,在MX-1细胞中为100 nM。[2] Rucaparib的羧酸代谢物在透化细胞中抑制PARP活性的IC50约为550 nM,但在完整细胞中不抑制PARP,表明其膜透性差。[2] |
| 体内研究 (In Vivo) |
Rucaparib 无毒,但在具有 DNA 修复蛋白能力的 D384Med 异种移植物中显着增强替莫唑胺诱导的 TGD。药代动力学研究还表明,Rucaparib在脑组织中被检测到,这表明Rucaparib具有治疗颅内恶性肿瘤的潜力。 Rucaparib 显着增强拓扑替康和替莫唑胺在 NB-1691、SH-SY-5Y 和 SKNBE (2c) 细胞中的细胞毒性。 Rucaparib 增强替莫唑胺的抗肿瘤活性,并表明 NB1691 和 SHSY5Y 异种移植物中的肿瘤完全且持续消退。
在携带Capan-1 (BRCA2突变胰腺癌) 异种移植瘤的小鼠中,单次口服rucaparib (150 mg/kg) 可抑制肿瘤PARP活性超过90%,持续长达7天。单次腹腔注射 (10 mg/kg) 也能导致长时间的抑制(24小时抑制>70%,48小时仅轻微恢复至基线约30%,随后4天无进一步恢复)。[2] 每周一次口服给予rucaparib (150 mg/kg,每周一次,持续6周) 在延缓Capan-1异种移植瘤生长方面,其效果与每日腹腔注射 (10 mg/kg,每周5天,持续6周) 相当或更优。在每周一次的方案中,十只小鼠中有三只出现了完全缓解。[2] Rucaparib对生长缓慢的MX-1 (BRCA1突变乳腺癌) 异种移植瘤没有显示出显著的抗肿瘤活性,尽管单次口服150 mg/kg后24小时,肿瘤中PARP抑制超过80%。[2] 与正常组织相比,PARP抑制在肿瘤组织中似乎更持久。肿瘤PARP活性被抑制一周,而肝脏中的活性在24小时内恢复,外周血单核细胞中的活性在给药后24小时恢复至对照的35-40%。[2] |
| 酶活实验 |
测量[ 32 P]NAD+掺入对人全长重组PARP-1的抑制程度。 PhosphorImager 用于量化整合到酸不溶性材料中的 [32P]ADP-核糖。非线性回归分析用于计算 Kiis。
PARP活性使用经验证的免疫印迹法测定。简要流程:组织(肿瘤、脑、肝脏)在等渗缓冲液中匀浆。在测定中,透化细胞(如PBMCs)或组织匀浆中的PARP活性,通过在过量NAD+ (350 µM) 存在下与双链寡核苷酸(模拟DNA断裂)共孵育而最大化激活。形成的聚腺苷二磷酸核糖聚合物通过使用抗PAR抗体进行免疫印迹定量,并以纯化的PAR标准曲线作为参照。结果表示为每10^6细胞或每毫克蛋白质的pmol PAR。[2] |
| 细胞实验 |
第二天,将细胞接种到 24 孔板(2,500-4000 个细胞/孔)后,用逐渐升高的药物浓度进行处理。 72-96 小时后,通过添加 CellTiter-Glo 试剂并利用读板器测量发光来评估细胞活力。将细胞一式三份(500-4000 个细胞/孔)接种到 6 孔板中,进行克隆存活测定。铺板后,16-18 小时后对细胞进行药物处理,并使其生长 14 天。
药物摄取与保留(放射化学测定法): 指数生长期的细胞与[¹⁴C]rucaparib和非细胞渗透性示踪剂(如[³H]蔗糖/菊粉)一起孵育,以校正细胞外液。在指定时间点,通过离心使细胞穿过硅油层进入KOH溶液,从而与培养基快速分离。通过双标记闪烁计数测定细胞沉淀中的放射性,并在校正了滞留的细胞外药物后计算细胞内药物浓度。对于保留研究,细胞用药物脉冲处理,洗涤,然后在新鲜培养基中孵育不同时间后再处理。[2] 克隆形成实验: 将指数生长期的Capan-1或MX-1细胞以低密度接种于多孔板中。贴壁后,细胞暴露于递增浓度的rucaparib中24小时,然后更换为无药培养基。让细胞形成集落10-14天,染色、计数,并计算相对于未处理对照的存活率。[2] 细胞PARP活性测定: 药物处理后(例如30分钟脉冲),细胞被洗涤、收集并冷冻保存。随后使用“酶活性实验”部分描述的基于免疫印迹的方法在透化细胞中测量PARP活性。[2] |
| 动物实验 |
CD-1裸鼠携带已建立的D283Med异种移植瘤;溶于生理盐水;1 mg/kg;每日腹腔注射1次或4次
体内抗肿瘤活性测定[1] 用于抗肿瘤研究的雌性无胸腺裸鼠(CD1 nu/nu)在特定病原体清除(SPF)条件下于隔离器中饲养和处理。我们将SW620结直肠肿瘤细胞(每只动物1 × 10⁷个细胞)皮下植入每只小鼠的一侧腹部,在肿瘤可触及时(植入后10-12天)对小鼠(每组5只)进行治疗,并使用二维游标卡尺测量监测肿瘤生长。肿瘤体积使用公式a² × b / 2计算,其中a为最小测量值,b为最大测量值。数据以中位相对肿瘤体积(RTV)表示,定义为计算的肿瘤体积除以治疗初始日(第0天)计算的肿瘤体积。因此,在第 0 天,RTV 值为 1,RTV4 值表示肿瘤体积是其初始值的四倍。 单剂量研究。[1] 我们以 200 mg/kg 的剂量口服替莫唑胺(生理盐水混悬液),单独给药或与单次腹腔注射 PARP 抑制剂联合给药,剂量分别为 0.1 [AG14447 和 MS-AG14644(相当于 0.078 mg/kg 游离 AG14644)]、1.0 和 10 mg/kg(甲磺酸盐相当于 0.79 和 7.9 mg/kg 游离 AG14451 和 AG14452 以及 0.78 和 7.8 mg/kg 游离 AG14531 和 AG14644)。对照组动物分别接受生理盐水口服和腹腔注射,或生理盐水口服联合PARP抑制剂10 mg/kg腹腔注射。 五日给药研究。[1] 我们用替莫唑胺(以生理盐水混悬液形式)口服给药,每日五次,单独给药或与以下PARP抑制剂联合腹腔注射,每日五次:0.05、0.15和0.5 mg/kg AG14447;0.15和0.5 mg/kg MS-AG14644(相当于0.12和0.39 mg/kg游离AG14644);1.5、5和15 mg/kg AG14361;以及5 mg/kg AG14452。对照组动物分别接受生理盐水口服和腹腔注射,或生理盐水口服联合较高剂量(0.5、5 或 15 mg/kg,取决于所研究的化合物)的 PARP 抑制剂腹腔注射。 组织分布[1] 我们向携带 SW620 异种移植瘤(约 10 × 10 mm)的小鼠(每组三只)腹腔注射 AG14361、AG14452 或 AG14447(10 mg/kg)。120 分钟后,在全身麻醉下,通过心脏穿刺取血,切除肿瘤并立即置于液氮中速冻。分离血浆并储存于 -20°C。采用反相高效液相色谱法(等度洗脱流动相:40%乙腈/0.1%甲酸铵,Hypersil BDS 3 μm 4.6 × 250 mm色谱柱,Waters Alliance 2690高效液相色谱仪;Waters公司,英国赫特福德郡埃尔斯特里)通过加标法测定乙腈处理血浆和匀浆肿瘤中PARP抑制剂的浓度。 异种移植瘤的建立:使用10-12周龄的雌性CD-1裸鼠。将Capan-1细胞皮下植入小鼠侧腹。将MX-1细胞植入生长因子减少的基底膜基质和培养基的1:1混合物中。 [2]药代动力学和组织分布研究:将已建立Capan-1肿瘤(约25-50 mm³)的小鼠腹腔注射(10 mg/kg)或口服(50、100或150 mg/kg)单剂量rucaparib。药物溶于无菌去离子水中。分别于给药后0.5、4、24、48、72和168小时处死小鼠(每组3只)。采集血液(用于血浆分析)、肿瘤和脑组织,速冻后保存,用于药物浓度(HPLC法)和PARP活性分析。[2]疗效研究:将携带可触及Capan-1肿瘤(≥5×5 mm)的小鼠随机分为治疗组(n=8-10)。 Rucaparib 按照不同的给药方案给药 6 周:载体对照组;10 mg/kg 腹腔注射,每日一次,每周 5 天;50 或 150 mg/kg 口服,每日一次,每周 5 天;150 mg/kg 口服,每周一次;150 mg/kg 口服,每周三次;或 150 mg/kg 口服,每 3 周一次,每日一次,连续 5 天。定期测量肿瘤大小并计算体积。监测小鼠的体重变化,如果肿瘤达到预设大小限制或出现应激迹象,则对小鼠实施安乐死。[2] |
| 药代性质 (ADME/PK) |
吸收
在每日两次、每次 240 mg 至 840 mg 的剂量范围内,鲁卡帕尼呈线性药代动力学特征。在批准的推荐剂量下,平均(变异系数 [CV])稳态鲁卡帕尼 Cmax 为 1940 ng/mL (54%),AUC0-12h 为 16900 h× ng/mL (54%)。平均 AUC 累积比率为 3.5 至 6.2 倍。在批准的推荐剂量下,稳态 Tmax 的中位数为 1.9 小时,范围为 0 至 5.98 小时。平均绝对生物利用度为 36%,范围为 30% 至 45%。高脂餐使 Cmax 和 AUC0-24h 分别增加 20% 和 38%,Tmax 延迟 2.5 小时。 消除途径 单次口服放射性标记的鲁卡帕尼后,未代谢的鲁卡帕尼占放射性总量的 64%。尿液和粪便中分别占放射性总量的 45% 和 95%。 分布容积 平均表观分布容积(变异系数)为 2300 L (21%)。 清除率 稳态下平均表观总清除率(变异系数)为 44.2 L/h (45%)。 代谢/代谢物 体外研究表明,鲁卡帕尼主要由 CYP2D6 代谢,其次由 CYP1A2 和 CYP3A4 代谢。除 CYP 介导的氧化外,鲁卡帕尼还会发生 N-去甲基化、N-甲基化和葡萄糖醛酸化。在一项研究中,研究人员在血浆、尿液和粪便中鉴定出了七种鲁卡帕尼代谢物。 生物半衰期 平均(变异系数)末端消除半衰期为26(39%)小时。 鲁卡帕尼在小鼠体内具有口服生物利用度。单次口服给药(50-150 mg/kg)后,母体药物可在血浆中检测到长达4-48小时,具体时间取决于剂量。[2] 与血浆相比,鲁卡帕尼在肿瘤组织中的浓度显著更高,且滞留时间更长。给药后4小时,肿瘤中的药物浓度比同期血浆浓度高出10倍,并且在口服给药后,该药物在肿瘤中可持续检测到长达3天。[2] 药物进入大脑的摄取有限,脑组织中的药物浓度≤血浆浓度的10%。 [2] 在血浆和肿瘤中均检测到一种羧酸代谢物,其在血浆中的浓度与母体药物相当或更高,但在肿瘤中的浓度较低。该代谢物具有较弱的PARP抑制活性(在无细胞测定中IC50约为550 nM),但不抑制完整细胞中的PARP。[2] 小鼠的口服生物利用度与人体相似,且从血浆中迅速消除。观察到剂量与药物浓度呈线性关系。[2] |
| 毒性/毒理 (Toxicokinetics/TK) |
肝毒性
在鲁卡帕尼的大型临床试验中,常规肝功能检查异常较为常见;74%的患者出现血清ALT升高,其中13%的患者ALT值超过正常值上限(ULN)的5倍。尽管临床试验中治疗期间血清酶升高较为常见,但未见出现伴有黄疸的肝炎或肝功能衰竭的报告。在鲁卡帕尼获批并更广泛应用后,尚未有已发表的临床上明显的肝损伤病例报告。因此,鲁卡帕尼是血清酶升高的常见原因,但尚未发现与显著肝毒性相关。 可能性评分:E(未经证实但怀疑是临床上明显的肝损伤的原因)。 妊娠和哺乳期影响 ◉哺乳期用药概述 目前尚无关于鲁卡帕尼在哺乳期临床应用的信息。制造商建议在接受 rucaparib 治疗期间以及末次给药后 2 周内停止母乳喂养。 ◉ 对母乳喂养婴儿的影响 截至修订日期,未找到相关的已发表信息。 ◉ 对泌乳和母乳的影响 截至修订日期,未找到相关的已发表信息。 蛋白结合 体外实验表明,rucaparib 与人血浆蛋白的结合率为 70%。Rucaparib 优先分布于红细胞,血血浆浓度比为 1.8。 在携带 Capan-1 或 MX-1 异种移植瘤的小鼠的疗效研究中,采用不同给药方案(每日或每周口服剂量高达 150 mg/kg)的 rucaparib 治疗未引起任何显著的体重减轻(最低体重减轻 ≤4%)。 [2] 该手稿提到,单药卢卡帕尼(rucaparib)以50 mg/kg的剂量,每5天重复给药一次,持续6个月,对小鼠无毒性。与替莫唑胺联合用药时,最大耐受剂量较低。[2] 临床数据(引用但并非来自本研究)表明,单药PARP抑制剂的耐受剂量在联合化疗研究中可引起3/4级骨髓抑制。单药PARP抑制剂曾报道有神经系统副作用(例如嗜睡)。在小鼠中观察到的卢卡帕尼脑渗透性较低,可能降低此类认知/神经系统副作用的发生率。[2] |
| 参考文献 | |
| 其他信息 |
鲁卡帕尼樟脑磺酸盐是鲁卡帕尼与一摩尔当量的(1S,4R)-樟脑磺酸反应制得的樟脑磺酸盐。它是聚(ADP-核糖)聚合酶的抑制剂,用于治疗晚期卵巢癌和携带致病性生殖系或体细胞BRCA突变的卵巢癌。它是一种EC 2.4.2.30(NAD(+) ADP-核糖基转移酶)抑制剂。它是一种樟脑磺酸盐和氮杂环庚并吲哚类化合物。它包含一个(S)-樟脑磺酸盐和一个rucaparib(1+)。
Rucaparib樟脑磺酸盐是rucaparib的樟脑磺酸盐形式,rucaparib是一种口服生物利用度高的三环吲哚类化合物,也是聚(ADP-核糖)聚合酶(PARP)1 (PARP1)、2 (PARP2)和3 (PARP3)的抑制剂,具有潜在的化疗/放疗增敏和抗肿瘤活性。给药后,rucaparib选择性地与PARP1、2和3结合,并抑制PARP介导的DNA修复。这会增强DNA链断裂的积累,促进基因组不稳定,并诱导细胞周期阻滞和凋亡。这可能增强DNA损伤剂的细胞毒性,并逆转肿瘤细胞对化疗和放疗的耐药性。 PARP 是由单链 DNA 断裂激活的酶,催化核蛋白的翻译后 ADP 核糖基化修饰,从而诱导信号传导并募集其他蛋白质修复受损 DNA。PARP 介导的修复通路在 DNA 修复中发挥关键作用,并在多种癌细胞类型中失调。 另见:Rucaparib(具有活性部分)。 药物适应症 治疗输卵管癌、治疗卵巢癌、治疗原发性腹膜癌、治疗前列腺恶性肿瘤 Rucaparib (AG-014699) 是首个进入癌症治疗临床试验的 PARP 抑制剂。 [2] 短期药物暴露后观察到的持续性PARP抑制归因于载体介导的细胞摄取、逆浓度梯度积累以及与酶的亲和力结合(Ki ~1.4 nM),而非PARP在DNA上的“捕获”。[2] 肿瘤PARP的长期抑制和滞留支持在临床上探索间歇给药方案(例如,每周一次),这可能在维持疗效的同时,潜在地降低毒性或提高用药便利性。[2] 本研究中MX-1异种移植瘤缺乏疗效,尽管体外试验显示其对药物敏感,这可能与该模型中肿瘤生长缓慢或肿瘤中较高的基础PARP活性有关。[2] |
| 分子式 |
C29H34FN3O5S
|
|---|---|
| 分子量 |
555.67
|
| 精确质量 |
555.22
|
| 元素分析 |
555.670
Elemental Analysis:
|
| CAS号 |
1859053-21-6
|
| 相关CAS号 |
459868-92-9 (phosphate); 1859053-21-6 (Rucaparib camsylate); 283173-50-2
|
| PubChem CID |
121490161
|
| 外观&性状 |
Light yellow to gray solid powder
|
| tPSA |
137Ų
|
| 氢键供体(HBD)数目 |
4
|
| 氢键受体(HBA)数目 |
7
|
| 可旋转键数目(RBC) |
5
|
| 重原子数目 |
39
|
| 分子复杂度/Complexity |
869
|
| 定义原子立体中心数目 |
2
|
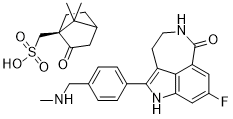
| SMILES |
S(C([H])([H])[C@@]12C(C([H])([H])[C@@]([H])(C([H])([H])C1([H])[H])C2(C([H])([H])[H])C([H])([H])[H])=O)(=O)(=O)O[H].FC1=C([H])C2C(N([H])C([H])([H])C([H])([H])C3=C(C4C([H])=C([H])C(C([H])([H])N([H])C([H])([H])[H])=C([H])C=4[H])N([H])C(=C1[H])C3=2)=O
|
| InChi Key |
INBJJAFXHQQSRW-STOWLHSFSA-N
|
| InChi Code |
InChI=1S/C19H18FN3O.C10H16O4S/c1-21-10-11-2-4-12(5-3-11)18-14-6-7-22-19(24)15-8-13(20)9-16(23-18)17(14)15;1-9(2)7-3-4-10(9,8(11)5-7)6-15(12,13)14/h2-5,8-9,21,23H,6-7,10H2,1H3,(H,22,24);7H,3-6H2,1-2H3,(H,12,13,14)/t;7-,10-/m.1/s1
|
| 化学名 |
[(1S,4R)-7,7-dimethyl-2-oxo-1-bicyclo[2.2.1]heptanyl]methanesulfonic acid;6-fluoro-2-[4-(methylaminomethyl)phenyl]-3,10-diazatricyclo[6.4.1.04,13]trideca-1,4,6,8(13)-tetraen-9-one
|
| 别名 |
AG014699 camsylate; PF-01367338; AG 014699; PF 01367338 camsylate; AG-014699; PF01367338; AG-14447 camsylate; AG 14447; Rucaparib monocamsylate; Rucaparib (monocamsylate); Rucaparib (Camsylate); rucaparib camphorsulfonate; CO-338; PF-1367338-BW; AG14447 camsylate; Trade name: Rubraca
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 请将本产品存放在密封且受保护的环境中,避免吸湿/受潮。 |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO: ≥ 100 mg/mL
Water: N/A Ethanol: N/A |
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.08 mg/mL (3.74 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 20.8 mg/mL澄清DMSO储备液加入400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.08 mg/mL (3.74 mM) (饱和度未知) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 20.8 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.08 mg/mL (3.74 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7996 mL | 8.9981 mL | 17.9963 mL | |
| 5 mM | 0.3599 mL | 1.7996 mL | 3.5993 mL | |
| 10 mM | 0.1800 mL | 0.8998 mL | 1.7996 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT03442556 | Active Recruiting |
Drug: Rucaparib Camsylate Drug: Rucaparib |
ATM Gene Mutation PSA Progression |
University of Washington | August 24, 2018 | Phase 2 |
| NCT03552471 | Active Recruiting |
Drug: Rucaparib Camsylate Other: Pharmacokinetic Study |
BRCA1 Gene Mutation BRCA2 Gene Mutation |
Ohio State University Comprehensive Cancer Center |
July 12, 2018 | Phase 1 |
| NCT04455750 | Active Recruiting |
Drug: Rucaparib camsylate Drug: Enzalutamide |
Stage IV Prostate Cancer AJCC v8 Stage IVA Prostate Cancer AJCC v8 |
Alliance for Clinical Trials in Oncology |
February 19, 2021 | Phase 3 |
| NCT03521037 | Active Recruiting |
Drug: Rucaparib camsylate | Neoplasms | pharmaand GmbH | February 27, 2018 | Phase 1 |
| NCT02986100 | Completed | Drug: C-14 labeled Rucaparib Drug: Rucaparib |
Solid Tumor | pharmaand GmbH | November 2016 | Phase 1 |
AG-014699 inhibits Single strand break (SSB) repair to a similar extent regardless of cellular NF-κB status.Oncogene, 2012, 31(2), 251-264. |
|---|