| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
| 靶点 |
orexin receptor/OX
Suvorexant (MK-4305) acts on Orexin 1 receptor (OX1R, Ki = 0.57 nM) and Orexin 2 receptor (OX2R, Ki = 0.35 nM) [1] Suvorexant (MK-4305) is a dual antagonist of OX1R (Ki = 0.6 nM) and OX2R (Ki = 0.4 nM); it inhibits OX1R-mediated calcium mobilization with IC50 = 2.6 nM and OX2R-mediated calcium mobilization with IC50 = 1.2 nM [2] |
|---|---|
| 体外研究 (In Vitro) |
Suvorexant(也称为 MK-4305)是一种有效的双重 OX 受体拮抗剂,对于 OX1 受体和 OX2 受体的 Ki 值分别为 0.55 nM 和 0.35 nM。 Suvorexant是由默克公司开发的用于治疗失眠的药物。目前正在进行 III 期试验。 Suvorexant 的作用是关闭清醒状态,而不是诱导睡眠。作为一种双重食欲素受体 (OXR) 拮抗剂 (DORA),suvorexant (MK-4305) 已显示出治疗失眠症和睡眠障碍的前景。激酶测定:MK-4305 是 OX1 受体和 OX2 受体的有效拮抗剂,Ki 值分别为 0.55 nM 和 0.35 nM 细胞测定:体外研究表明 MK-4305 具有清晰的辅助特征(对 OX1 受体和 OX2 受体的选择性 >10000 倍) OX2R)通过 MDS Pharma 的 170 种酶、受体和离子通道脱靶筛选确定。
Suvorexant (MK-4305) 在放射性配体结合实验中对OX1R和OX2R均表现出高亲和力,Ki值处于亚纳摩尔范围。它能竞争性地将放射性标记的食欲素配体从两种受体上置换下来 [1] Suvorexant (MK-4305) 能有效抑制表达人OX1R或OX2R的细胞中食欲素-A诱导的钙内流,IC50值处于低纳摩尔范围。它对100多种其他受体、离子通道或酶无明显亲和力,显示出高选择性 [2] |
| 体内研究 (In Vivo) |
在一项小鼠体内研究中,在小鼠非活动阶段(开灯)测试了 suvorexant(25 毫克/千克),此时睡眠自然更普遍且食欲素水平通常较低。研究发现,suvorexant 在给药后的前 4 小时内选择性地增加 REM,从而显着扰乱了睡眠结构。在测试剂量下,suvorexant 仅在第一个小时内显着减少苏醒时间,而 IPSU 不影响苏醒时间。这些数据表明,与 DORA 相比,OX2R 偏好拮抗剂可能降低了扰乱 NREM/REM 结构的趋势
在大鼠中,口服给予 Suvorexant (MK-4305)(3、10、30 mg/kg)能剂量依赖性地增加活动期的总睡眠时间(TST)和非快速眼动(NREM)睡眠时间,不影响快速眼动(REM)睡眠持续时间。给药后30分钟内即可观察到促睡眠作用,且持续4-6小时 [1] 在犬中,口服 Suvorexant (MK-4305)(1、3、10 mg/kg)能显著延长活动期的总睡眠时间并减少觉醒时间。在10 mg/kg剂量下,与溶媒对照组相比,NREM睡眠增加约40% [1] 在食蟹猴中,Suvorexant (MK-4305)(0.3、1、3 mg/kg,口服)剂量依赖性地增加总睡眠时间并减少觉醒状态。它还能使药物诱导睡眠紊乱的猴子的睡眠结构恢复正常 [2] 在睡眠剥夺大鼠中,Suvorexant (MK-4305)(10 mg/kg,口服)能逆转觉醒时间的增加并恢复正常的NREM睡眠持续时间,且在药物清除后不会引起反跳性觉醒 [2] |
| 酶活实验 |
MK-4305 对 OX1 受体的 Ki 值为 0.55 nM,对 OX2 受体的 Ki 值为 0.35 nM,使其成为这两种受体的强拮抗剂。
Bioactivation测定[1] 人肝微粒体 在100 mM磷酸钾缓冲液(pH 7.4)中以1 mg/mL蛋白与10 μM测试化合物、1 mM MgCl2、1 mM EDTA、5 mM谷胱甘肽和1 mM NADPH在37℃下预孵育60分钟。用含0.15 μM拉贝他洛尔(内标)的25%乙腈终止反应。将样品涡旋混合,14000 rpm离心10分钟。每个样品的上清液转移到HPLC瓶中进行HRMS分析。 采用uplc -高分辨质谱法(HRMS)对gsh衍生加合物进行鉴定。该系统由一个Waters Acquity样品管理器和两个Waters Acquity UPLC泵组成。HRMS采用Waters Q-TOF Xevo质谱仪进行。采用Phenomenex Synergi 2.5 μm MAX-RP 100 Å柱(50 mm × 2 mm)加热至60°C实现分离。流动相为含有0.1%甲酸的水(溶剂A)和含有0.1%甲酸的乙腈(溶剂B),流速为0.5 mL/min。梯度从第一分钟的5%溶剂B开始,然后在接下来的0.5分钟内线性增加到15%溶剂B。然后在接下来的11.5分钟内将溶剂B增加到50%,然后在2分钟内进一步增加到90%。然后用95%的溶剂B洗涤1.5分钟。在每次运行结束时,在初始条件下重新平衡5分钟。质谱分析采用正离子模式电喷雾电离。ESI毛细管电压为1.5 kV,源温度为100℃,脱溶温度为600℃。质量扫描范围为150 ~ 1000 μ m,扫描时间为0.25 s/次。锁质量为588.8691 amu,使用频率为每5次扫描一次。每个测试化合物形成的谷胱甘肽加合物的相对量是用峰面积比估计的。与gsh衍生加合物相关的质谱峰面积除以内标拉贝他洛尔的面积。 放射配体结合试验[1] 根据Kunapuli等人的方法,从CHO细胞中表达的人食欲素2受体(hOX2R)和食欲素1受体(hOX1R)的Ile408-Val变体制备膜。将CHO/OX2R和CHO/OX1R细胞与PBS/1 mM EDTA分离,1000g离心10分钟。将细胞颗粒在冰冷的20 mM Hepes, 1 mM EDTA, pH 7.4中用Polytron均质,在4℃下20000g离心20分钟。这个过程重复了两次。以5 mg膜蛋白/mL重悬于实验缓冲液(20 mM Hepes, 125 mM NaCl, 5 mM KCl, pH 7.4)中。加入牛血清白蛋白至终浓度为1%,等分液保存于- 80°C。利用自动化Tecan液体处理系统和moser等人描述的Packard unfilter -96进行放射性配体结合试验。实验在96孔微滴板上进行,室温下,最终测定量为1.0 mL,在含125 nM NaCl和5 mM KCl的20 mM Hepes缓冲液(pH 7.4)中进行。用DMSO配制待测化合物溶液,用DMSO连续稀释,10种溶液浓度相差3倍,各20 μL。非特异性结合(NSB)采用高亲和力配体(终浓度为1 μM)测定,总结合(TB)采用DMSO(终浓度为2%)测定。将受体溶液(30pm终值,通常为2−10 μg膜)和氚化配体(~ 80 Ci/ mol)添加到测试化合物中。OX2R受体采用0.15 nM的化合物18 (KD = 0.3 nM)。OX1R受体采用0.7 nM的化合物19 (KD = 3 nM)。使用化合物20在0.03 nM (KD = 0.03 nM)浓度下进行OX1R测定,结果相同;然而,在这种情况下,首先在化合物中加入920 μL的膜,然后再加入60 μL的热配体。室温孵育3小时(化合物20 20小时)后,样品通过Packard GF/B过滤器过滤(预先浸泡在0.2% PEI,聚乙烯西格玛P-3143中),并用1ml 20 mM Hepes冷缓冲液(pH 7.4)洗涤5次。滤板真空干燥后,加入50 μL Packard Microscint-20,用Packard TopCount测定结合放射性(CPM bound)。 放射性配体结合[2] 瞬时表达人OX2受体的HEK293细胞的细胞膜与[3H]-EMPA在Krebs实验缓冲液(8.5 mM HEPES, 1.3 mM CaCl2, 1.2 mM MgSO4, 118 mM NaCl, 4.7 mM KCl, 4 mM NaHCO3, 1.2 mM KH2PO4, 11 mM葡萄糖,pH 7.4)中孵育,总实验体积为0.25 mL,最终DMSO浓度为1%。室温孵育90分钟后,通过GF/B 96孔玻璃纤维板快速过滤,用Tomtec细胞收集机用5 × 0.25 mL的ddH2O洗涤,终止反应。结合放射性是用Lablogic SafeScint通过液体闪烁来测定的,并在微-液体闪烁计数器上检测。非特异性结合被确定为在拮抗剂EMPA达到10 μM饱和浓度的情况下仍然存在。在[3H]-EMPA (0.4 nM - 15 nM)浓度范围内,膜(2 μg蛋白/孔)孵育,进行饱和度研究。使用SafeScint和Beckman LS 6000液体闪烁计数器测定放射性配体浓度。用1.5 nM浓度的[3H]-EMPA和一系列浓度的测试化合物如3 (Suvorexant / MK-4305)孵育膜(2 μg蛋白/孔)进行竞争结合。 通过将相同的细胞膜(2 μg蛋白/孔)添加到含有1% DMSO和1.5 nM辐射配体的Krebs缓冲液的孔中,在不同的时间点上共3小时,测定了辐射配体的结合动力学。通过预平衡膜和[3H]-EMPA测定90 min的解离动力学;然后在不同的时间点加入饱和浓度的冷EMPA (100 μM),以防止放射性配体与受体分离时重新结合。 OX1R和OX2R放射性配体结合实验:将表达重组人OX1R或OX2R的细胞制备的膜制剂与放射性标记的食欲素配体及不同浓度的 Suvorexant (MK-4305) 在25°C下孵育90分钟。通过过滤分离结合态和游离态放射性配体,测量放射性强度以计算Ki值 [1] 钙动员实验:将表达人OX1R或OX2R的细胞加载钙敏感荧光染料,与 Suvorexant (MK-4305) 预孵育30分钟。随后加入食欲素-A,随时间测量荧光强度,以确定抑制钙内流的IC50值 [2] |
| 细胞实验 |
基于 MDS Pharma 对 170 种酶、受体和离子通道的脱靶筛选,体外研究表明 MK-4305 具有清晰的辅助特征(对 OX2R 的选择性>10000 倍)。
FLIPR测定[1] 为了测量细胞内钙,将表达食欲素1受体Ile408-Val变体或人食欲素2受体的中国仓鼠卵巢(CHO)细胞生长在Iscove修饰的DMEM中,该DMEM含有2 mM l-谷氨酰胺、0.5 g/mL G418、1%次黄嘌呤胸腺嘧啶补充剂、100 U/mL青霉素、100 ug/mL链霉素和10%热灭活胎牛血清。将细胞以20000个/孔的速度接种到涂有聚d-赖氨酸的Becton-Dickinson黑色384孔透明底无菌板中。所有试剂均来自GIBCO-Invitrogen Corp.。种板在37°C和6% CO2下孵育过夜。α -6,12人食欲素- a作为激动剂,在1%牛血清白蛋白(BSA)中配制0.5 mM原液,在实验缓冲液(含20 mM HEPES和2.5 mM probenecid, pH 7.4)中稀释,最终浓度为0.3 - 2 nM,用于实验。在DMSO中配制10 mM的原液,然后在384孔板中稀释和移液,首先在DMSO中,然后在分析缓冲液中。实验当天,用100 μL实验缓冲液洗涤细胞3次,然后在60 μL含有1 μM Fluo-4AM酯、0.02% pluronic酸和1% BSA的实验缓冲液中(37°C, 6% CO2)孵育60分钟。然后抽吸染料上样液,用100 μL缓冲液洗涤细胞3次。然后在每个孔中留下30 μL相同的缓冲液。在荧光成像板读取器内,以15 μL的体积向板中加入待测化合物,孵育5 min,最后加入激动剂15 μL。每孔以1 s间隔1 min和6 s间隔4 min测量荧光,并将每个荧光峰的高度与0.3−2 nM ala -6,12 orexin-A用缓冲液代替拮抗剂诱导的荧光峰高度进行比较。对于每种拮抗剂,确定IC50值(抑制50%激动剂反应所需的化合物浓度)。[1] 功能性肌醇磷酸和ERK1/2磷酸化测定[2] 以25 000个细胞/孔密度稳定表达人食欲素-2受体的CHO细胞播种24 h后,在96孔板上进行基于细胞的肌醇磷酸和ERK1/2磷酸化功能测定;完整的分析细节见辅助信息。 重组细胞系培养:稳定表达人OX1R或OX2R的细胞在标准培养条件(37°C、5% CO2)下的生长培养基中培养。每2-3天传代一次,以维持对数生长期 [1] 钙动员细胞实验:将融合生长的细胞分离,重悬于实验缓冲液中,在37°C下加载荧光染料60分钟。洗涤后,将细胞接种到96孔板中,用 Suvorexant (MK-4305) 处理30分钟。加入食欲素-A作为刺激物,使用酶标仪检测荧光,评估钙通量 [2] 选择性筛选实验:将表达多种受体、离子通道或酶的细胞与10 μM浓度的 Suvorexant (MK-4305) 孵育。使用靶点特异性实验测量结合或功能活性,评估脱靶相互作用 [2] |
| 动物实验 |
Mice Rat Sleep Assay[1]
Adult male Sprague−Dawley rats (450−600 g; Taconic Farms, Germantown, NY) were subcutaneously implanted with telemetric physiologic monitors (model F50-EEE or 4ET SI; Data Sciences International, Arden Hills, MN) that were used to simultaneously record both the electrocorticogram (ECoG) and electromyogram (EMG) activities of the rat. For placement of the 4ET SI, animals were anesthetized with isoflurane and electrodes for recording ECoG signals and EMG signals were placed. Position of the wires are based on the following coordinates. Channel 1 wire. From Lambda AP +2, ML +2 −2. Channel 2 wires From BREGMA AP +1.5 ML +3.2 (hole 1) AP −10.5 (hole2). Channel 3 wires From BREGMA AP −3.0 ML +1.5, −3.5. EMG lead placement was in neck muscle. An incision was made ∼3−5 cm in length midline on the dorsal thorax to form a pocket on the left and right side of midline, and the telemetry module was placed with a saddlebag placement method. The animals were given a single dose of antibiotic (gentomycin, 5.8 mg/kg) and an analgesic (buprenorphine, 0.1 mL) within 3 h following surgery. The animals were allowed to recover from surgery for at least two weeks prior to recording. Throughout these experiments, animals were housed individually in plastic cages (19 in. × 101/2 in. × 8 in.; Lab Products, Seaford, DE) and were provided water and food ad libitum. Lights were on a 12 h light: 12 h dark cycle with lights off at 4:00 a.m. and on at 4:00 p.m. ECoG and EMG signals were collected simultaneously from all animals using Dataquest ART software system, digitally sampled at 500 Hz, and stored on a PC for off-line analysis. The hydrochloride salt of compound 10 (458 mg) was dissolved in 70.2 mL of a 20% aqueous solution of TPGS and administered by oral gavage at 10 mpk of the free-base equivalent to four rats, 5 h into their active period (09:00 or ZT 17:00). For 3 (Suvorexant / MK-4305), the free-base (1.27 g) was suspended in 70.2 mL of a 20% aqueous solution of TPGS and dosed as above. Recordings were started just prior to compound administration and were collected for 23 h. The experiments were based on a standard crossover design with two animals receiving compound for one week and the complementary group receiving vehicle, followed by a week of reversed administration. All animals were exposed to two days administration of orally gavaged vehicle prior to initiation of experimental drug administration to allow for habituation. For baseline sleep measurements, continuous recordings were collected for two days to get average sleep behaviors for each animal over contiguous days prior to drug and vehicle administration. During the drug administration studies, recordings were collected each day prior to, during, and following drug administration. Recordings were begun prior to compound administration so that the exact time of administration was recorded within the raw data file as artifactual noise which was caused by removing the implanted transmitter from the receptive field of the receiver during administration. This information allowed a direct measure of drug/vehicle administration time during offline analysis and was not included in the data analysis. Following the completion of data collection, all data were scored with automated sleep stage analysis software, Somnologica. Assignment of sleep stages was made in general accord with those described by J. M. Monti’s group.Sleep/wake stages were assigned based upon a combination of level of movement within the field of the radio frequency receiver over which individually housed rats were caged, EMG activity, and ECoG frequencies over 10 s epochs. Active wake was assigned to the epoch when movement of the animal was detected over the receiver or when there was an active EMG signal over the epoch and the ECoG frequencies consisted of low-voltage high frequency activity. An epoch was scored as light sleep when there was no movement activity, the EMG was moderately activ,e and the ECoG consisted of either theta or theta activity mixed with less than 50% of the epoch showing delta activity. Delta sleep was scored when there was no gross movement, reduced EMG activity, and the ECoG consisted of more than 50% delta wave activity (i.e., 0.5 to 4 Hz). Rapid eye movement (REM) sleep was scored when there was no movement or EMG activity and the ECoG consisted of primarily theta activity. Results of staging were grouped into 30 min periods following drug administration and the number of entries into each stage and the duration of minutes spent in each stage were calculated. The results for all four animals were averaged by treatment, or vehicle, over seven administration nights and the results were statistically compared based upon a mixed ANOVA analysis. Ex Vivo Occupancy Assay[1] Transgenic rats expressing human OX2R were dosed intravenously by infusion over a 30 min period or orally with 3 (Suvorexant / MK-4305) at doses of 0.1−2.0 mg/kg in 25% hydroxypropyl-β-cyclodextrin and then sacrificed. Samples of brain were quickly removed and frozen for use in the ex vivo occupancy assay, while a second set of tissue samples, a plasma sample, and CSF were frozen for LCMS determination of drug levels. For the ex vivo assay, approximately 60 mg of cord or brain was homogenized in 67 volumes of ice-cold assay buffer (20 mM HEPES, 120 mM NaCl, 5 mM KCl, pH7.4) and centrifuged at 21000g for 1 min. The pellets were resuspended in ice-cold buffer at a concentration of 10 mg tissue/mL and 100 μL aliquots were rapidly distributed to tubes with 0.5 mL rof oom temperature buffer containing 200 pM compound A. At 2, 4, 6, 8, 10, 12, and 15 min following membrane addition, incubations were terminated by filtration of three tubes over glass fiber filters. A parallel set of incubations performed in the presence of 1 μM of an unlabeled, potent DORA (OX2R Ki = 1.0 nM) was used to determine nonspecific radioligand binding at each time point. Radioactivity on the filters was determined by liquid scintillation counting and compound A rates of association were determined by linear regression. Receptor occupancy in a drug treated animal is calculated as: % occupancy = (1 − (slopedrug/slopevehicle)) × 100. The concentrations of drug required to achieve 90% receptor occupancy were derived by nonlinear curve fitting using Prism software. Rat sleep study: Male Sprague-Dawley rats were surgically implanted with electroencephalogram (EEG) and electromyogram (EMG) electrodes for sleep monitoring. After recovery, rats were acclimated to recording cages for 5 days. Suvorexant (MK-4305) was dissolved in a vehicle consisting of polyethylene glycol 400 and water (1:1 v/v) and administered orally via gavage at doses of 3, 10, or 30 mg/kg. EEG/EMG signals were recorded continuously for 24 hours post-administration, and sleep stages were scored manually [1] Dog sleep study: Beagle dogs were implanted with EEG/EMG electrodes and allowed to recover for 2 weeks. Suvorexant (MK-4305) was formulated as an oral suspension and administered at 1, 3, or 10 mg/kg. Sleep parameters were recorded for 12 hours post-dosing, and data were analyzed for TST, NREM, and REM sleep durations [1] Cynomolgus monkey sleep disruption study: Monkeys were implanted with EEG electrodes and subjected to sleep disruption via intermittent noise exposure. Suvorexant (MK-4305) was administered orally at 0.3, 1, or 3 mg/kg 1 hour before the disruption period. EEG recordings were analyzed for wake time, TST, and sleep architecture [2] Rat sleep deprivation reversal study: Rats were sleep-deprived for 6 hours using a gentle handling method. Suvorexant (MK-4305) was administered orally at 10 mg/kg immediately after deprivation. EEG/EMG recordings were performed for 18 hours post-dosing to assess sleep recovery [2] |
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
Under fasting conditions, the median peak concentration (Tmax) of sovoraxen is 2 hours. Co-administration with a high-fat meal has no effect on AUC or Cmax, but may delay Tmax by approximately 1.5 hours. The mean absolute bioavailability of 10 mg is 82%. Approximately 66% is excreted in feces, and 23% in urine. The mean volume of distribution is approximately 49 liters. Metabolism/Metabolites Sovoraxen is primarily metabolized by the cytochrome P450 3A4 enzyme (CYP3A4), with a smaller contribution from CYP2C19. The main circulating metabolites are sovoraxen and a hydroxysovoraxen metabolite, the latter of which is not expected to have pharmacological activity. There is a possibility of drug interactions with drugs that inhibit or induce CYP3A4 activity. Biological Half-Life The mean half-life is approximately 12 hours. In rats, the oral bioavailability of Suvorexant (MK-4305) was 45% at a dose of 10 mg/kg, with a peak plasma concentration (Cmax) of 1.2 μg/mL 1 hour after administration. The elimination half-life (t1/2) was 3.8 hours and the volume of distribution (Vd) was 12 L/kg [1] In dogs, the oral bioavailability was 72% (10 mg/kg dose), Cmax was 2.5 μg/mL (0.8 hours), t1/2 was 5.2 hours, and Vd was 8.5 L/kg [1] In cynomolgus monkeys, the oral bioavailability of Suvorexant (MK-4305) was 68% (3 mg/kg dose), Cmax was 1.8 μg/mL (1 hour), t1/2 was 6.5 hours, and the total clearance (CL) was 1.2 mL/min/kg [2] Suvorexant (MK-4305) is mainly metabolized by cytochrome P450 3A4. (CYP3A4) is present in liver microsomes, but the major active metabolite was not detected [2] |
| 毒性/毒理 (Toxicokinetics/TK) |
Hepatotoxicity
In multiple clinical trials, suvorexin was well tolerated, with serum ALT elevations occurring in 0% to 5% of cases, typically in higher dose groups, and resolving spontaneously without dose adjustment. No clinically observable liver injury was reported in suvorexin's registration trials. Suvorexin has a limited market presence, but even with overdose, it has not been found to be associated with clinically observable liver injury. Probability Score: E (Unlikely to cause clinically observable liver injury). Pregnancy and Lactation Effects ◉ Overview of Use During Lactation Data from two women indicate that suvorexin levels in breast milk are extremely low. If the mother needs to take suvorexin, breastfeeding does not need to be discontinued. If suvorexin is used, infant sedation should be monitored, especially in newborns or preterm infants. Until more data are available, alternative medications may be preferred, especially during breastfeeding of newborns or preterm infants. ◉ Effects on breastfed infants No relevant published information was found as of the revision date. ◉ Effects on lactation and breast milk No relevant published information was found as of the revision date. Protein binding Suvoresin binds extensively (>99%) to human plasma proteins and does not preferentially distribute to erythrocytes. It binds to human serum albumin and α1-acid glycoprotein. Suvoresin (MK-4305) has a plasma protein binding rate of >99% in rat, dog and human plasma [1] In a 14-day repeated-dose toxicity study in rats, oral administration of suvoresin (MK-4305) up to 300 mg/kg/day did not cause significant changes in body weight, hematological or clinical chemical parameters. No histopathological abnormalities were observed in major organs [1] In dogs, repeated administration at a dose of 100 mg/kg/day for 14 days resulted in mild sedation, but no other adverse reactions were observed. No evidence of hepatotoxicity or nephrotoxicity was detected [2] Suvorexant (MK-4305) does not inhibit or induce major CYP450 isoenzymes at therapeutic concentrations, suggesting a low likelihood of drug interactions [2] |
| 参考文献 | |
| 其他信息 |
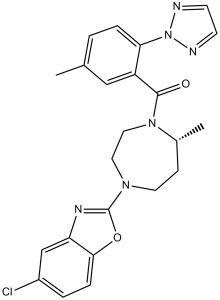
Suvorexant is an aromatic amide formed by the condensation of the carboxyl group of 5-methyl-2-(2H-1,2,3-triazol-2-yl)benzoic acid with the secondary amino group of 5-chloro-2-[(5R)-5-methyl-1,4-diazacycloheptane-1-yl]-1,3-benzoxazole. It is an orexin receptor antagonist used to treat insomnia. It has both central nervous system depressant and orexin receptor antagonistic effects. It belongs to the 1,3-benzoxazole class, triazole class, diazacycloheptane class, aromatic amide class, and organochlorine class. Suvorexant is a Category IV controlled substance under the U.S. Drug Enforcement Administration (DEA). Category IV substances are less likely to be abused compared to Category III substances. It is a sedative. Suvorexant is a selective dual orexin receptor OX1R and OX2R antagonist that promotes sleep by reducing wakefulness and arousal. It has been approved for the treatment of insomnia.
Sovresen is an orexin receptor antagonist. Its mechanism of action is as an orexin receptor antagonist, a P-glycoprotein inhibitor, and a cytochrome P450 3A inhibitor. Sovresen is an orexin receptor antagonist used to treat insomnia and sleep disorders. Sovresen treatment has been associated with rare, transient elevations of serum enzymes, but no clinically significant cases of liver injury have been found. Sovresen is an orally bioavailable orexin receptor antagonist that antagonizes orexin receptor type 1 (OX1R) and orexin receptor type 2 (OX2R) for the treatment of insomnia. After oral administration, sovresen targets and binds to orexin receptors OX1R and OX2R. It blocks the binding of the neuropeptides orexin A and orexin B to OX1R and OX2R, thereby inhibiting orexin signaling-induced arousal. Drug Indications Suvorexant is indicated for the treatment of insomnia characterized by difficulty falling asleep and/or difficulty maintaining sleep. FDA Label Mechanism of Action Suvorexant is a dual antagonist of orexin receptors OX1R and OX2R. It exerts its pharmacological action by inhibiting the binding of the neuropeptides orexin A and B (also known as hypothalamic secretin 1 and 2). These neuropeptides are produced by neurons in the lateral hypothalamus. These neurons control the brain's arousal centers, are active during wakefulness, especially during movement, and cease firing during sleep. Suvorexant reduces arousal and wakefulness by inhibiting the strengthening effects of the arousal system, rather than directly promoting sleep. Despite a deeper understanding of the biological basis of sleep control in the brain, few new mechanisms for treating insomnia have been discovered in recent years. A notable exception is the use of orexin receptor antagonists to inhibit the excitatory neuropeptides orexin A and B. This article describes how we worked to investigate the poor oral pharmacokinetics of a leading high-throughput screening (HTS)-derived diazepam-based orexin receptor antagonist, ultimately discovering compound 10 with a 7-methyl substitution on the diazepam core. Despite exhibiting good potency, improved pharmacokinetics, and excellent in vivo efficacy, compound 10 forms an active metabolite during microsomal incubation. Based on mechanistic hypotheses and in vitro bioactivation experiments, we substituted the fluoroquinazoline ring in compound 10 with chlorobenzoxazole to obtain compound 3 (MK-4305). MK-4305 is a potent dual orexin receptor antagonist currently undergoing a phase III clinical trial for the treatment of primary insomnia. [1] Orexin receptor antagonists are a novel treatment for insomnia that directly targets sleep/wake regulation. Several such compounds have entered clinical development, including dual orexin receptor antagonists suvorexin and amorexin. In this study, we analyzed several orexin-2 (OX₂) selective antagonist radioligands [³H]-EMPA using equilibrium and kinetic studies. Furthermore, using CHO cells stably expressing the OX2 receptor, we investigated the effects of some compounds on inositol phosphate accumulation and ERK-1/2 phosphorylation by incubating with different agonists for 30 min and 5 min, respectively. EMPA, suvorexin, amorexin, and TCS-OX-29 all bound to the OX2 receptor with moderate to high affinity (pK(I) values ≥7.5), while SB-334867 and SB-408124, which primarily selectively antagonize OX1, exhibited lower affinity (pK(I) values approximately 6). Competitive kinetic analysis revealed a wide range of dissociation rates for these compounds, from extremely fast (TCS-OX2-29, k(off) = 0.22 min⁻¹) to extremely slow (almorexant, k(off) = 0.005 min⁻¹). Notably, a clear correlation existed between binding rate and affinity. In cell-based experiments, the rapidly acting antagonists EMPA and TCS-OX2-29 exhibited reversible antagonism against orexin A agonist activity. However, both suvorexant and almorexant resulted in concentration-dependent inhibition of the maximum orexin A response, which was more pronounced with shorter agonist incubation times. Analysis based on a semi-equilibrium model indicated that the antagonists dissociated more slowly in cellular systems than in membrane-bound systems; in this case, almorexant effectively acted as a pseudo-irreversible antagonist. [2] Sovresen (MK-4305) is a first-in-class dual orexin receptor antagonist (DORA) designed for the treatment of insomnia. Its mechanism of action is to block the binding of orexin peptides to OX1R and OX2R, which are key regulators of the sleep-wake cycle[1]. Unlike benzodiazepines, sovresen (MK-4305) did not cause respiratory depression or physical dependence in preclinical studies. It maintains normal sleep structure by increasing non-rapid eye movement (NREM) sleep without interfering with rapid eye movement (REM) sleep[2]. The high selectivity of sovresen (MK-4305) to orexin receptors minimizes off-target effects, which contributes to its good safety profile in preclinical evaluations[1]. |
| 分子式 |
C23H23CLN6O2
|
|
|---|---|---|
| 分子量 |
450.9207
|
|
| 精确质量 |
450.16
|
|
| CAS号 |
1030377-33-3
|
|
| 相关CAS号 |
|
|
| PubChem CID |
24965990
|
|
| 外观&性状 |
Typically exists as solid at room temperature
|
|
| LogP |
4.9
|
|
| tPSA |
80.3
|
|
| 氢键供体(HBD)数目 |
0
|
|
| 氢键受体(HBA)数目 |
6
|
|
| 可旋转键数目(RBC) |
3
|
|
| 重原子数目 |
32
|
|
| 分子复杂度/Complexity |
664
|
|
| 定义原子立体中心数目 |
1
|
|
| SMILES |
C[C@@H]1CCN(CCN1C(=O)C2=C(C=CC(=C2)C)N3N=CC=N3)C4=NC5=C(O4)C=CC(=C5)Cl
|
|
| InChi Key |
JYTNQNCOQXFQPK-MRXNPFEDSA-N
|
|
| InChi Code |
InChI=1S/C23H23ClN6O2/c1-15-3-5-20(30-25-8-9-26-30)18(13-15)22(31)29-12-11-28(10-7-16(29)2)23-27-19-14-17(24)4-6-21(19)32-23/h3-6,8-9,13-14,16H,7,10-12H2,1-2H3/t16-/m1/s1
|
|
| 化学名 |
[(7R)-4-(5-chloro-1,3-benzoxazol-2-yl)-7-methyl-1,4-diazepan-1-yl]-[5-methyl-2-(triazol-2-yl)phenyl]methanone
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|---|
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2177 mL | 11.0884 mL | 22.1769 mL | |
| 5 mM | 0.4435 mL | 2.2177 mL | 4.4354 mL | |
| 10 mM | 0.2218 mL | 1.1088 mL | 2.2177 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
A multi-center, double-blind, randomized, parallel design study to compare the effectiveness of suvorexant versus placebo on sleep pressure and circadian rhythm in insomniacs with hypertension: The Super 1 study
CTID: UMIN000023389
Phase: Status: Complete: follow-up complete
Date: 2016-08-12