| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|
| 靶点 |
Topoisomerase II
DNA topoisomerase II [2][3] MDM2 (indirect target, sensitized by miR-181b) [3] |
|---|---|
| 体外研究 (In Vitro) |
Teniposide 是一种拓扑异构酶 II 抑制剂。 Teniposide (VM-26, 0.15–45 mg/L) 的 IC50 为 0.35 mg/L,并以剂量依赖性方式抑制 Tca8113 细胞增殖。当暴露于 5 mg/L 的替尼泊苷时,Tca8113 细胞会发生凋亡。 Tca8113 细胞中的细胞被替尼泊苷 (5.0 mg/L) 阻滞在 G2/M 期[2]。当细胞具有高水平的 miR-181b 时,替尼泊苷对源自患者的原代培养神经胶质瘤细胞具有活性,其 IC50 为 1.3±0.34 μg/mL。与对照细胞相比,替尼泊苷处理的细胞表现出活力降低,并且当MDM2被抑制时,IC50从5.86±0.36μg/mL下降至2.90±0.35μg/mL。通过 MDM2 的介导,替尼泊苷还降低表达高水平 miR-181b 的神经胶质瘤细胞的活力[3]。
在口腔鳞状细胞癌(OSCC)细胞中,替尼泊苷(VM-26)以浓度依赖方式发挥抗增殖活性,浓度≥10 μM时细胞活力显著降低。它触发凋亡,表现为caspase-3激活、PARP切割和DNA片段化。该药物还在部分OSCC细胞系中诱导G2/M期细胞周期阻滞,在另一些细胞系中诱导S期阻滞[2] - 在胶质瘤细胞中,替尼泊苷(VM-26)具有抗肿瘤活性,且miR-181b过表达可增强其疗效。miR-181b靶向MDM2,下调其表达并促进p53蓄积,协同增强替尼泊苷诱导的细胞死亡[3] - 替尼泊苷(VM-26)可捕获DNA拓扑异构酶II,稳定酶-DNA切割复合物并阻止DNA链连接,导致DNA双链断裂,进而引发细胞周期阻滞或凋亡[2][3] |
| 体内研究 (In Vivo) |
替尼泊苷(0.5 mg/kg,腹腔注射)可显着提高微核嗜多染红细胞 (MNPCE) 的频率,这一发现由于显着抑制骨髓而与骨髓毒性直接相关。替尼泊苷(24 mg/kg,腹腔注射)可显着降低 BrdU 标记精子的频率。此外,替尼泊苷(12、24 mg/kg,腹腔注射)可显着诱导雄性小鼠生殖细胞中的二体精子[1]。
在雄性小鼠中,腹腔注射替尼泊苷(VM-26)对体细胞(骨髓细胞)和生殖细胞(精原细胞)均产生非整倍体效应。它以剂量依赖方式引起数目染色体异常,包括非整倍体和多倍体。治疗组的染色体畸变频率显著高于对照组[1] |
| 酶活实验 |
DNA拓扑异构酶II活性检测:将纯化的DNA拓扑异构酶II与超螺旋质粒DNA在反应缓冲液中于37°C孵育。加入系列浓度(1-20 μM)的替尼泊苷(VM-26),混合物孵育45分钟。加入SDS和蛋白酶K终止反应,随后在55°C孵育1小时。通过1%琼脂糖凝胶电泳分离DNA产物,溴化乙锭染色。通过测量超螺旋DNA条带强度,定量拓扑异构酶II介导的DNA松弛抑制效果,证实该药物可稳定酶-DNA切割复合物[2][3]
|
| 细胞实验 |
OSCC细胞抗增殖、凋亡及细胞周期检测:将OSCC细胞以4×10³个细胞/孔接种到96孔板中,用1、5、10、20 μM的替尼泊苷(VM-26)处理24、48、72小时。采用比色法检测细胞活力。凋亡检测时,用10和20 μM药物处理细胞48小时后,用膜联蛋白V-FITC/PI染色并通过流式细胞术分析;通过蛋白质印迹法检测caspase-3和PARP的表达。采用碘化丙啶染色和流式细胞术评估细胞周期分布[2]
- 胶质瘤细胞增敏检测:将胶质瘤细胞用miR-181b模拟物或阴性对照模拟物转染24小时,随后用5-15 μM的替尼泊苷(VM-26)处理48小时。采用四唑盐比色法检测细胞活力。通过蛋白质印迹法检测MDM2和p53蛋白水平,通过RT-PCR定量MDM2 mRNA表达[3] |
| 动物实验 |
Aneugenic effect mouse model: Male ICR mice (8-10 weeks old) were randomly divided into control and treatment groups (n=6 per group). Teniposide (VM-26) was dissolved in a suitable solvent and administered intraperitoneally at doses of 5, 10, 20 mg/kg. Control mice received an equal volume of solvent. Mice were sacrificed 24 hours after a single dose for bone marrow sampling, and 35 days after dosing for spermatogonia sampling. Chromosomal preparations were made from bone marrow cells and spermatogonia, and numerical chromosome abnormalities were analyzed by microscopic observation [1]
|
| 药代性质 (ADME/PK) |
Absorption, Distribution and Excretion
4% to 12% of the dose is excreted unchanged in the urine. Radioactive material is excreted in the feces within 72 hours after administration, accounting for 0% to 10% of the administered dose. 10.3 mL/min/m² In 11 patients who received teniposide at a dose of 100–150 mg/m², administered 1.5–3 hours before tumor resection, teniposide was detected in brain tumor tissue at concentrations of 0.05–1.12 μg/g tissue. In three patients, lower drug concentrations were observed in adjacent normal brain tissue (< 0.9 μg/g tissue), while in the remaining patients, it was undetectable (< 0.05 μg/g tissue). Teniposide was detected in one patient who died three days after receiving a cumulative intravenous dose of 576 mg teniposide. The highest concentrations of teniposide were found in the spleen, prostate, heart, large intestine, liver, and pancreas. It is currently unclear whether teniposide is excreted into breast milk. Excretion routes: Renal: 4% to 12% of the dose is excreted unchanged teniposide. In a study of tritium-labeled teniposide in adults, 44% of the radiolabeled substance (parent compound and metabolites) was recovered in urine within 120 hours after administration. Fecal excretion: 0% to 10% of the dose. For more complete data on absorption, distribution, and excretion of teniposide (6 items in total), please visit the HSDB record page. Metabolism/Metabolites In isolated human liver formulations, cytochrome P450 mixed-function isoenzymes catalyze (side chain) E-ring metabolism to O-demethylated metabolites and catechol metabolites. This metabolism is subsequently attributed primarily to CYP3A4 activity, and secondarily to CYP3A5. Peroxidase-mediated O-demethylation of teniposide has also been reported. It has been reported that in children taking teniposide, the main metabolites in serum and urine are hydroxy acids formed by the opening of the lactone ring; cis isomers have also been detected, possibly degradation products formed during storage. Aglycones formed from the partial loss of glucosinolates were not detected (Evans et al., 1982). In other studies using high doses of teniposide, no hydroxy acids were found in plasma or urine, and the concentration of teniposide did not change after incubation with glucuronidase in these samples, indicating that little or no formation of the proposed glucuronide metabolites was observed (Holthuis et al., 1987). However, in another study, 6% of teniposide was excreted unchanged in urine within 24 hours, and another 8% was excreted as an unidentified aglycone glucuronide. In general, the effects of teniposide in mammalian cells in vitro occur without exogenous metabolic activation. Several teniposide metabolites have been identified, but their mutagenic properties have not been investigated. The known metabolites of teniposide include teniposide catechol. Biological half-life 5 hours Terminal half-life: 5 hours. Note: Following intravenous infusion, plasma teniposide concentrations show a biexponential decrease. Plasma clearance follows a multiphasic pattern. Half-lives of 4 hours and 10 to 40 hours were observed after distribution. |
| 毒性/毒理 (Toxicokinetics/TK) |
Drug Interactions
Intravenous administration of cyclosporine (5 mg/kg body weight, 2 hours later, followed by intravenous administration of 30 mg/kg body weight, 48 hours later) increased the AUC of teniposide by 50%, due to its reduced clearance. Conversely, concomitant administration of phenytoin increased the clearance of teniposide from 13 mL/min/m² in the control group to 32 mL/min/m². Genetic Toxicity: Teniposide (VM-26) exhibited dose-dependent aneuploidy toxicity in male mice, inducing chromosomal number abnormalities in somatic cells (bone marrow) and germ cells (spermatogonia). The highest dose (20 mg/kg) resulted in a significant increase in the incidence of aneuploidy and polyploidy [1] |
| 参考文献 |
|
| 其他信息 |
According to state or federal labeling requirements, teniposide may cause developmental toxicity. Teniposide is a semi-synthetic derivative of podophyllotoxin with antitumor activity. Teniposide inhibits DNA synthesis by forming a complex with topoisomerase II and DNA. This complex induces double-strand DNA breaks and prevents topoisomerase II from binding for repair. The accumulation of DNA breaks prevents cells from entering the mitotic phase of the cell cycle, ultimately leading to cell death. Teniposide primarily acts on the G2 and S phases of the cell cycle. Teniposide is a topoisomerase inhibitor. The mechanism of action of teniposide is as a topoisomerase inhibitor. Teniposide is a semi-synthetic derivative of podophyllotoxin with antitumor activity. Teniposide forms a ternary complex with topoisomerase II and DNA, leading to dose-dependent DNA single-strand and double-strand breaks, DNA-protein crosslinking, inhibition of DNA strand reconnection, and cytotoxicity. This drug acts on late S phase or early G1 phase of the cell cycle. (NCI04)
Teniposide is a semi-synthetic derivative of podophyllotoxin with antitumor activity. Teniposide inhibits DNA synthesis by forming a complex with topoisomerase II and DNA. This complex induces double-strand DNA breaks and prevents topoisomerase II from binding for repair. The accumulation of DNA breaks prevents cells from entering the mitotic phase of the cell cycle, ultimately leading to cell death. Teniposide primarily acts on the G2 and S phases of the cell cycle. Drug Indications Teniposide is used to treat refractory acute lymphoblastic leukemia. Mechanism of Action The mechanism of action of teniposide appears to be related to the inhibition of type II topoisomerase activity, as teniposide does not intercalate into DNA or bind strongly to DNA. Teniposide binds to DNA topoisomerase II and inhibits its activity. The cytotoxic effect of teniposide is related to the relative number of double-strand DNA breaks generated intracellularly, reflecting the stability of the topoisomerase II-DNA intermediate. It is an inhibitor of DNA topoisomerase II: Teniposide is a DNA topoisomerase II poison that has been shown to promote DNA breaks, particularly with a strong affinity for the C or T at the -1 position. Most mutational events reported in mammalian cells, including point mutations, chromosomal deletions and crossing over, and aneuploidy, can be explained by this activity. Teniposide does not inhibit bacterial topoisomerases and therefore may not cause bacterial mutations through the same mechanisms as in mammalian cells. Unlike many other DNA topoisomerase II inhibitors, teniposide does not bind to DNA, either covalently or intercalally. Instead, it appears to interact directly with DNA topoisomerase II. …The drug appears to exert its cytotoxic effects by damaging DNA, thereby inhibiting or altering DNA synthesis. Studies have shown that teniposide can induce single-strand DNA breaks; the drug can also induce double-strand DNA breaks and DNA-protein crosslinks. …Teniposide appears to have cell cycle specificity, inducing G2 phase arrest and preferentially killing cells in the late G2 and S phases. Therapeutic Use Antitumor drug; enzyme inhibitor; nucleic acid synthesis inhibitor Teniposide, used in combination with other approved anticancer drugs, is indicated for induction therapy of refractory childhood acute lymphoblastic (lymphoblastic) leukemia. /Included in the US product label/ Teniposide can be used as monotherapy or in combination therapy for the treatment of refractory non-Hodgkin lymphoma. /Not included in the US product label/ Teniposide can be used as monotherapy or in combination therapy for the treatment of refractory neuroblastoma. /Not included in the US product label/ Drug Warnings Because patients with Down syndrome and leukemia may be particularly sensitive to myelosuppressive chemotherapy, the initial dose of teniposide should be reduced in such patients. There is currently no experience with the use of teniposide in patients with impaired renal and/or hepatic function, therefore specific recommendations regarding dose adjustment cannot be made. However, the possibility of dose adjustment for such patients should be considered. The major and dose-limiting adverse reaction of teniposide is hematologic toxicity. Myelosuppression is dose-related and can be severe when teniposide is used in combination with other chemotherapy drugs to treat acute lymphoblastic leukemia (ALL). When using the teniposide doses and dosing regimens required for treating refractory ALL, early and severe myelosuppression is expected, with delayed recovery, as myeloproliferation is the anticipated endpoint of treatment. Patients receiving this drug may experience severe myelosuppression, leading to infection and bleeding. In pediatric patients receiving teniposide monotherapy, infection and bleeding occurred in approximately 12% and 5%, respectively. Pregnancy Risk Grade: D / Positive evidence of risk exists. Human studies, investigational data, or post-marketing data all indicate fetal risk. However, the potential benefits of using this drug may outweigh the potential risks. For example, this drug may be acceptable in life-threatening situations or when a patient has a serious illness for which safer drugs are unavailable or ineffective. / For more complete data on drug warnings for teniposide (18 of them), please visit the HSDB record page. Pharmacodynamics Teniposide is a phase-specific cytotoxic drug that acts on the late S phase or early G2 phase of the cell cycle. Teniposide inhibits cell mitosis by causing single-strand and double-strand DNA breaks and cross-linking between proteins and DNA. Teniposide (VM-26) is a semi-synthetic podophyllotoxin derivative with potent antitumor activity[2][3] - Mechanism of action: It exerts its antitumor effect by capturing DNA topoisomerase II, stabilizing the enzyme-DNA cleavage complex, and inducing DNA double-strand breaks, thereby leading to cell cycle arrest (G2/M phase or S phase) and apoptosis[2][3] - Sensitization pathway: miR-181b enhances the antitumor efficacy of teniposide (VM-26) in glioma cells by targeting MDM2, downregulating its expression, and promoting p53-mediated cell death[3] - Clinical application: It is used to treat a variety of cancers, including childhood acute lymphoblastic leukemia, glioma, and oral squamous cell carcinoma[2][3] - Genetic toxicity risk: The aneuploidy effect of germ cells suggests a potential genetic risk, which needs to be taken seriously in clinical applications[1] |
| 分子式 |
C32H32O13S
|
|
|---|---|---|
| 分子量 |
656.65
|
|
| 精确质量 |
656.156
|
|
| 元素分析 |
C, 58.53; H, 4.91; O, 31.67; S, 4.88
|
|
| CAS号 |
29767-20-2
|
|
| 相关CAS号 |
|
|
| PubChem CID |
452548
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| 密度 |
1.6±0.1 g/cm3
|
|
| 沸点 |
864.3±65.0 °C at 760 mmHg
|
|
| 熔点 |
274 - 277ºC
|
|
| 闪点 |
476.5±34.3 °C
|
|
| 蒸汽压 |
0.0±0.3 mmHg at 25°C
|
|
| 折射率 |
1.697
|
|
| LogP |
1.71
|
|
| tPSA |
189.07
|
|
| 氢键供体(HBD)数目 |
3
|
|
| 氢键受体(HBA)数目 |
14
|
|
| 可旋转键数目(RBC) |
6
|
|
| 重原子数目 |
46
|
|
| 分子复杂度/Complexity |
1090
|
|
| 定义原子立体中心数目 |
10
|
|
| SMILES |
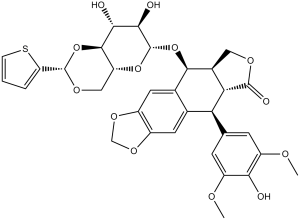
S1C([H])=C([H])C([H])=C1C1([H])OC([H])([H])[C@]2([H])[C@]([H])([C@@]([H])([C@]([H])([C@@]([H])(O2)OC2([H])C3=C([H])C4=C(C([H])=C3[C@@]([H])(C3C([H])=C(C(=C(C=3[H])OC([H])([H])[H])O[H])OC([H])([H])[H])[C@@]3([H])C(=O)OC([H])([H])C32[H])OC([H])([H])O4)O[H])O[H])O1
|
|
| InChi Key |
NRUKOCRGYNPUPR-QBPJDGROSA-N
|
|
| InChi Code |
InChI=1S/C32H32O13S/c1-37-19-6-13(7-20(38-2)25(19)33)23-14-8-17-18(42-12-41-17)9-15(14)28(16-10-39-30(36)24(16)23)44-32-27(35)26(34)29-21(43-32)11-40-31(45-29)22-4-3-5-46-22/h3-9,16,21,23-24,26-29,31-35H,10-12H2,1-2H3/t16-,21+,23+,24-,26+,27+,28+,29+,31+,32-/m0/s1
|
|
| 化学名 |
(5S,5aR,8aR,9R)-5-[[(2R,4aR,6R,7R,8R,8aS)-7,8-dihydroxy-2-thiophen-2-yl-4,4a,6,7,8,8a-hexahydropyrano[3,2-d][1,3]dioxin-6-yl]oxy]-9-(4-hydroxy-3,5-dimethoxyphenyl)-5a,6,8a,9-tetrahydro-5H-[2]benzofuro[6,5-f][1,3]benzodioxol-8-one
|
|
| 别名 |
VM26; NSC-122819; VM-26; HSDB 6546; NSC 122819; VM 26; HSDB-6546; NSC122819; HSDB6546; CCRIS 2058. Trade name: Vumon; Vehem. Abbreviations: EPT; PTG
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month 注意: 本产品在运输和储存过程中需避光。 |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (3.81 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 2.5 mg/mL (3.81 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL 澄清 DMSO 储备液加入到 900 μL 玉米油中并混合均匀。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.5229 mL | 7.6144 mL | 15.2288 mL | |
| 5 mM | 0.3046 mL | 1.5229 mL | 3.0458 mL | |
| 10 mM | 0.1523 mL | 0.7614 mL | 1.5229 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| Recruiting | Completed | Drug: Teniposide | HLH | Beijing Friendship Hospital | October 22, 2023 | Not Applicable |
| NCT00004916 | Completed | Drug: teniposide Drug: paclitaxel |
Lymphoma | Northwestern University | February 1999 | Phase 1 Phase 2 |
| NCT00186875 | Completed | Drug: methotrexate, teniposide, PEG-asparaginase Drug: L-asparaginase, erwinia asparaginase |
Acute Lymphoblastic Leukemia Lymphoma, Lymphoblastic |
St. Jude Children's Research Hospital |
November 2003 | Phase 2 |
| NCT00184041 | Completed | Drug: Daunorubicin, Vincristine, Prednisone, Methotrexate, PEG- Asparaginase, 6-Mercaptopurine, Cytoxan, Cytosine Arabinoside, VM-26 and 6-Thioguanine |
Acute Lymphoblastic Leukemia | St. Jude Children's Research Hospital |
July 2004 | Phase 2 |
|
|
|
|
|
|
|