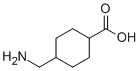
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
Effect of Tranexamic Acid on Intraoperative Blood Loss in Patients Undergoing Aquablation
CTID: NCT06710327
Phase: Phase 4 Status: Not yet recruiting
Date: 2024-11-29
Effect of Tranexamic Acid and Calcium Dobesilate for Bleeding of Endometrial Origin
CTID: NCT06707051
Phase: Phase 2/Phase 3 Status: Completed
Date: 2024-11-27
Tranexamic Acid Use on Pain, Mobility and Bleeding Following Total Hip and Total Knee Arthroplasty
CTID: NCT06208267
Phase: N/A Status: Not yet recruiting
Date: 2024-11-26
Topical Tranexamic Acid Use on Granulating Wounds Following Mohs Micrographic Surgery
CTID: NCT04541303
PhaseEarly Phase 1 Status: Completed
Date: 2024-11-21
Use of Tranexamic Acid in Reduction of Post-Op Complications in Mohs Micrographic Surgery
CTID: NCT04630886
PhaseEarly Phase 1 Status: Terminated
Date: 2024-11-19
View More
Massive Transfusion in Children-2: A Trial Examining Life Threatening Hemorrhage in Children
CTID: NCT06070350
Phase: Phase 3 Status: Recruiting
Date: 2024-11-13
Prevent Postpartum Hemorrhage in Women with Von Willebrand Disease: the VWD-WOMAN Trial
CTID: NCT04344860
Phase: Phase 3 Status: Completed
Date: 2024-11-13
The Effect of Local and Systemic Tranexamic Acid on Edema and Ecchymosis in Orbital Surgery
CTID: NCT06450392
Phase: Phase 4 Status: Recruiting
Date: 2024-11-12
Tranexamic Acid in Patients for Caesarian Delivery.
CTID: NCT05759156
Phase: N/A Status: Completed
Date: 2024-11-07
Tranexamic Acid for the Prevention of Postpartum Hemorrhage in Pregnant Women with Placenta Previa
CTID: NCT05811676
Phase: Phase 3 Status: Recruiting
Date: 2024-10-31
Effect of TXA on Reducing Bruising After Filler Injection
CTID: NCT06665594
Phase: Phase 2 Status: Not yet recruiting
Date: 2024-10-30
Efficacy and Safety of Retrograde Intraarticular Injection, Topical Soaking of Tranexamic Acid (TXA), or Placebo in Femoral Neck Fractured Patients Undergoing Cementless Bipolar Hemiarthroplasty
CTID: NCT06289478
Phase: N/A Status: Recruiting
Date: 2024-10-30
Preoperative Tranexamic Acid (TXA) to Prevent Bleeding in Patients Undergoing Major Colorectal Surgery
CTID: NCT06657924
Phase: Phase 2 Status: Not yet recruiting
Date: 2024-10-26
A Pilot Study Comparing Anti-Inflammatory Effects Of TXA Versus EACA In Pediatric Congenital Heart Surgery
CTID: NCT02656472
Phase: Phase 4 Status: Completed
Date: 2024-10-24
Prophylactic Tranexamic Acid To Reduce Blood Loss During Caesarean Delivery.
CTID: NCT06604325
Phase: N/A Status: Recruiting
Date: 2024-10-24
Comparison of Chitosan, Ankaferd and Tranexamic Acid in Dental Extraction in Liver Pre-Transplant Children
CTID: NCT06457360
PhaseEarly Phase 1 Status: Completed
Date: 2024-10-18
EFFECTS of TRANEXAMIC ACID in TUMESCENT SOLUTION for LIPOSUCTION PROCEDURES
CTID: NCT06648265
Phase: Phase 4 Status: Completed
Date: 2024-10-18
Window of Opportunity Study of Topical Tranexamic Acid for Cutaneous Squamous Cell Carcinoma
CTID: NCT06644079
Phase: Phase 2 Status: Not yet recruiting
Date: 2024-10-16
The Effect of Local Application of Tranexamic Acid Versus Placebo on Postoperative Complications in Plastic Surgery.
CTID: NCT06270407
Phase: Phase 4 Status: Recruiting
Date: 2024-10-16
Tranexamic Acid for Reduction of Transfusion in Abdominal Surgery
CTID: NCT06414031
Phase: Phase 3 Status: Recruiting
Date: 2024-10-15
Effect of TXA on Blood Loss in Reverse Total Shoulder Arthroplasty
CTID: NCT06638749
Phase: Phase 3 Status: Withdrawn
Date: 2024-10-15
Tranexamic Acid as an Intervention in Placenta Previa
CTID: NCT05688111
Phase: N/A Status: Completed
Date: 2024-10-15
Burn Study- Tranexamic Acid Versus Thrombin in Split Thickness Skin Graft
CTID: NCT06379724
Phase: Phase 4 Status: Recruiting
Date: 2024-10-08
Extended Oral Tranexamic Acid After Total Knee Arthroplasty
CTID: NCT06618820
Phase: Phase 3 Status: Enrolling by invitation
Date: 2024-10-01
Effect of Tranexamic Acid (TXA) on Reduction of Postoperative Blood Transfusion
CTID: NCT03063892
Phase: Phase 4 Status: Recruiting
Date: 2024-09-19
Evaluation of Tranexamic Acid in Myelodysplastic Syndromes and Acute Myeloid Leukemia
CTID: NCT06599762
Phase: N/A Status: Not yet recruiting
Date: 2024-09-19
Use of Tranexamic Acid After Vaginal Delivery With Episiotomy a RCT Placebo Control Trail
CTID: NCT05448456
Phase: Phase 4 Status: Completed
Date: 2024-09-19
Von Willebrand Factor in Pregnancy (VIP) Study
CTID: NCT04146376
Phase: Status: Recruiting
Date: 2024-09-19
Tranexamic Acid Versus Dexmedetomidine for Improving Surgical Field Quality During Spine Surgeries
CTID: NCT06587620
Phase: N/A Status: Completed
Date: 2024-09-19
Tranexamic Acid in Reducing Blood Loss in Patients with Pelvic Tumors Undergoing Hemipelvectomy Surgery
CTID: NCT03128866
PhaseEarly Phase 1 Status: Recruiting
Date: 2024-09-19
The Use of Tranexamic Acid in the Treatment of Symptomatic Subdural Hematoma
CTID: NCT05713630
Phase: Phase 3 Status: Not yet recruiting
Date: 2024-09-19
Postpartum Hemorrhage Reduction With Oral Tranexamic Acid: a Clinical Trial
CTID: NCT06025916
Phase: Phase 4 Status: Recruiting
Date: 2024-09-19
Comparing Intramyometrial Tranexamic Acid and Oxytocin for Blood Loss in Cesarean Section
CTID: NCT06010368
Phase: Phase 3 Status: Not yet recruiting
Date: 2024-09-05
Effect on Post-operative Pain of Tranexamic Acid Injection During Shoulder Surgery
CTID: NCT05302986
Phase: Phase 3 Status: Recruiting
Date: 2024-09-03
Prophylactic Endobronchial Tranexamic Acid to Reduce Bleeding in Transbronchial Cryobiopsy
CTID: NCT06266546
Phase: Phase 3 Status: Active, not recruiting
Date: 2024-08-21
A Randomized Control Trial Assessing the Effect of Topical Tranexamic Acid on Risk of Hematoma in Breast Surgery
CTID: NCT05441592
Phase: Phase 4 Status: Recruiting
Date: 2024-08-20
Tranexamic Acid During Colonic Endoscopic Resection Procedures
CTID: NCT05345613
Phase: Phase 4 Status: Recruiting
Date: 2024-08-07
Tranexamic Acid To Reduce Bleeding in Patients Treated With New Oral Anticoagulants Undergoing Dental Extraction
CTID: NCT03413891
Phase: Phase 4 Status: Terminated
Date: 2024-08-01
Effect of TXA Oral Sol 5% in Patients Treated With DOACs or VKA and Undergoing a Single or Multiple Tooth Extraction
CTID: NCT06143787
Phase: Phase 3 Status: Recruiting
Date: 2024-08-01
Early Tranexamic Acid and Modulating the Inflammatory Response in Sepsis
CTID: NCT04910464
Phase: Phase 3 Status: Recruiting
Date: 2024-07-31
California Prehospital and In Hospital Antifibrinolytic Therapy Via TXA
CTID: NCT03469947
Phase: Phase 3 Status: Completed
Date: 2024-07-30
Fractional Erbium YAG Laser vs Intradermal and Systemic Tranexamic Acid
CTID: NCT06522984
Phase: N/A Status: Recruiting
Date: 2024-07-26
Tranexamic Acid for Anaemia Trial
CTID: NCT06519422
Phase: Phase 3 Status: Not yet recruiting
Date: 2024-07-25
A Randomized Controlled Trial of Inhaled Tranexamic Acid for the Treatment of Pulmonary Hemorrhage in Cancer Patients
CTID: NCT05053867
Phase: Phase 3 Status: Recruiting
Date: 2024-07-24
Prevention of Postpartum Hemorrhage With Tranexamic Acid (Phase 2)
CTID: NCT05370820
Phase: Phase 2 Status: Recruiting
Date: 2024-07-24
Pharmacokinetics and Safety of Commonly Used Drugs in Lactating Women and Breastfed Infants
CTID: NCT03511118
Phase: Status: Recruiting
Date: 2024-07-24
Development of Products Based on Secretom From Stem Cell Conditioned Medium for Melasma Therapy
CTID: NCT06516419
Phase: N/A Status: Recruiting
Date: 2024-07-24
Comparing Tranexamic Acid Versus Ecbolics in Preventing Hemorrhage During and After Cesarean Section
CTID: NCT06060327
Phase: N/A Status: Completed
Date: 2024-07-23
Use of Tranexamic Acid in Blepharoplasties
CTID: NCT04724772
Phase: Phase 3 Status: Withdrawn
Date: 2024-07-23
Efficacy of Topical Tranexamic Acid in Reducing Blood Loss During CS in Patients With Placenta Previa
CTID: NCT06515535
Phase: N/A Status: Not yet recruiting
Date: 2024-07-23
Different Methods of Tranexamic Acid Application in Controlling Peri Operative Bleeding in Gynecomastia Surgery
CTID: NCT06509256
PhaseEarly Phase 1 Status: Not yet recruiting
Date: 2024-07-19
ITCH Trial: Protocol for a Randomized, Double Blind Placebo-controlled Trial
CTID: NCT04742205
Phase: Phase 2/Phase 3 Status: Completed
Date: 2024-07-16
Minimize Menorrhagia in Women With Von Willebrand Disease
CTID: NCT02606045
Phase: Phase 3 Status: Terminated
Date: 2024-07-01
Effects of Oral Tranexamic Acid Following Total Knee Arthroplasty
CTID: NCT06479161
Phase: Phase 2 Status: Recruiting
Date: 2024-06-28
Evaluating Postoperative Non-Opioid Pain Management Utilizing Local Anesthetics Coupled With Modulated Coagulation
CTID: NCT04814433
Phase: Phase 4 Status: Terminated
Date: 2024-06-26
A Study to Evaluate LB1148 for Return of Gastrointestinal Function and Adhesions in Subjects Undergoing Bowel Resection
CTID: NCT02836470
Phase: Phase 2 Status: Completed
Date: 2024-06-21
Tranexamic Acid to Prevent OpeRation in Chronic Subdural Hematoma
CTID: NCT03582293
Phase: Phase 3 Status: Recruiting
Date: 2024-06-20
Efficacy of Tranexamic Acid (TXA) in Humerus ORIF
CTID: NCT05802238
Phase: Phase 2 Status: Recruiting
Date: 2024-06-14
A Study to Evaluate LB1148 for Return of Bowel Function in Subjects Undergoing Bowel Resection
CTID: NCT05470387
Phase: Phase 3 Status: Terminated
Date: 2024-06-13
TRanEXamic Acid to Decrease Heavy Menstrual Bleeding in Individuals Anticoagulated for Venous Thromboembolism Pilot Study
CTID: NCT06452342
Phase: Phase 4 Status: Not yet recruiting
Date: 2024-06-12
Role of TXA in Patients Undergoing Breast Free Flap Reconstruction
CTID: NCT06428682
Phase: Phase 4 Status: Recruiting
Date: 2024-06-05
Evaluations of the Effects of Tranexamic Acid and Chlorhexidine Gel on Alveolar Osteitis Incidence
CTID: NCT06435832
Phase: N/A Status: Active, not recruiting
Date: 2024-05-30
Intravenous Tranexamic Acid and Intramyometrial Desmopressin Effect on Blood Loss During Laparoscopic Myomectomy.
CTID: NCT05517590
Phase: N/A Status: Completed
Date: 2024-05-24
Effect of Topical Tranexamic Acid on Postoperative Drainage in Minimally Invasive Transforaminal Lumbar Interbody Fusion (MI-TLIF)
CTID: NCT06427850
Phase: N/A Status: Not yet recruiting
Date: 2024-05-24
Protest Trial: TXA vs Saline
CTID: NCT06422273
Phase: Phase 3 Status: Enrolling by invitation
Date: 2024-05-20
Tranexamic Acid in Vaginal Reconstructive Surgery
CTID: NCT06419075
Phase: Phase 4 Status: Not yet recruiting
Date: 2024-05-17
Nebulized Tranexamic Acid in Sinus Surgery
CTID: NCT04905901
Phase: Phase 3 Status: Recruiting
Date: 2024-05-17
Tranexamic Acid to Improve Same-day Discharge Rates After Holmium Laser Enucleation of the Prostate (HoLEP)
CTID: NCT05082142
Phase: Phase 4 Status: Completed
Date: 2024-05-16
A Trial of a Hospital Policy of Tranexamic Acid Use to Reduce Transfusion in Major Non-cardiac Surgery
CTID: NCT04803747
Phase: Phase 4 Status: Active, not recruiting
Date: 2024-05-16
TXA in Spinal Fusion
CTID: NCT04272606
Phase: Phase 2 Status: Active, not recruiting
Date: 2024-05-14
Evaluating the Effect of Tranexamic Acid on the Clinical Outcomes in Patients With Traumatic Brain Injury
CTID: NCT06330935
Phase: Phase 1/Phase 2 Status: Recruiting
Date: 2024-05-10
Impact of Topical Tranexamic Acid in Breast Reconstruction
CTID: NCT05807074
Phase: Phase 4 Status: Recruiting
Date: 2024-05-09
ACid Tranexamic or Terlipressin for Initial Emergency Treatment of Mild to seVere hEmoptysis: a Randomized Trial.
CTID: NCT04961528
Phase: Phase 3 Status: Recruiting
Date: 2024-05-03
The Efficacy of Tranexamic Acid in the Treatment of Lichen Planus Pigmentosus and Erythema Dyschromicum Perstans
CTID: NCT04233749
Phase: Phase 2 Status: Recruiting
Date: 2024-05-02
Indian Trial of Tranexamic Acid in Spontaneous Intracerebral Haemorrhage
CTID: NCT05836831
Phase: Phase 4 Status: Recruiting
Date: 2024-04-30
Topical TRanexamic Acid vs. Placebo on Acute Postoperative Pain Following DRF Fixation
CTID: NCT06384456
Phase: Phase 4 Status: Not yet recruiting
Date: 2024-04-26
Tranexamic Acid to Prevent Heavy Bleeding After Childbirth in Women at Higher Risk
CTID: NCT05562609
Phase: Phase 3 Status: Recruiting
Date: 2024-04-25
Tranexamic Acid and Depot-Medroxyprogesterone Acetate for Perimenopausal Irregular Uterine Bleeding
CTID: NCT04710017
Phase: N/A Status: Completed
Date: 2024-04-24
Do Patients Who Received Tranexamic Acid in Vaginal Hysterectomy Loose Les Blood, Comparing to Patients Who Did Not?
CTID: NCT05921071
Phase: N/A Status: Recruiting
Date: 2024-04-23
Tranexamic Acid to Reduce Blood Loss in Spine Trauma Surgery
CTID: NCT02314988
Phase: Phase 2/Phase 3 Status: Recruiting
Date: 2024-04-15
Rate of Tranexamic Acid Administration on Blood Pressure (RateTXA) Study.
CTID: NCT06356948
Phase: Phase 4 Status: Recruiting
Date: 2024-04-10
Tranexamic Acid in Adult Spinal Deformity Surgery
CTID: NCT03553186
Phase: Phase 3 Status: Recruiting
Date: 2024-04-08
TRANexamic Acid to Reduce Bleeding in BURN Surgery
CTID: NCT03113253
Phase: Phase 4 Status: Completed
Date: 2024-04-04
Tranexamic Acid to Reduce Contraceptive-related Bleeding Side Effects
CTID: NCT06000423
Phase: Phase 4 Status: Recruiting
Date: 2024-04-03
Tranexamic Acid to Prevent Bleeding After Endoscopic Resection of Large Colorectal Polyps: A Pilot Project
CTID: NCT04559880
Phase: Phase 4 Status: Completed
Date: 2024-04-03
Tranexamic Acid to Improve Arthroscopic Visualization in Shoulder Surgery
CTID: NCT04594408
Phase: Phase 4 Status: Completed
Date: 2024-04-02
The Effects of Tranexamic Acid on Joint Inflammation and Cartilage Health in Anterior Cruciate Ligament Injured Patients
CTID: NCT03552705
Phase: Phase 2 Status: Recruiting
Date: 2024-03-28
TXA in Anticoagulated Patients Study
CTID: NCT04560010
Phase: Phase 4 Status: Terminated
Date: 2024-03-26
Tranexamic Acid Use to Reduce Blood Transfusion in Pediatric Cancer Patients Undergoing Limb Salvage Procedure
CTID: NCT04410042
Phase: Phase 3 Status: Terminated
Date: 2024-03-15
Tranexamic Acid During Excisional Burn Surgery
CTID: NCT05507983
Phase: Phase 3 Status: Recruiting
Date: 2024-03-13
Effectiveness Of Tranexamic Acıd Application Wıth Different Doses On Bleeding, Edema And Physician Comfort In Orthognathıc Surgery Patients
CTID: NCT06301204
Phase: N/A Status: Completed
Date: 2024-03-08
Vasopressin Versus Tranexamic Acid for Control of Blood Loss Related to A
READY 2: A PHASE 3, OPEN LABEL, SINGLE ARM STUDY ON THE USE OF CUSA-081 FOR DYSFUNCTIONAL CENTRAL VENOUS ACCESS DEVICES (CVADs)
CTID: null
Phase: Phase 3 Status: Prematurely Ended
Date: 2022-02-14
Topical use of tranexamic acid for optimisation of wound healing in a novel, acute wound model
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2021-08-30
The effect of tranexamic acid on seroma formation after mastectomies
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2021-08-30
MULTICENTER, DOUBLE BLIND CLINICAL TRIAL TO EVALUATE THE ANTIHEMORRAGIC EFFECT OF THE TOPICAL TRANEXAMIC ACID DURING PACEMAKER IN ANTICOAGULATED PATIENTS
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2021-06-17
ACTIVE : ACid tranexamic or Terlipressin for Initial emergency treatment of mild to seVere hEmoptysis
CTID: null
Phase: Phase 3 Status: Trial now transitioned
Date: 2021-04-02
Study of the noninferiority of an oral versus intravenous administration of tranexamic acid in total hip arthroplasty
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2021-03-15
Evaluation of intra articular Tranexamic acid for reduction of total blood loss in total hip arthroplasty
CTID: null
Phase: Phase 2 Status: Trial now transitioned
Date: 2021-01-21
Intramuscular tranexamic acid for the treatment of symptomatic mild traumatic brain injury in older adults: a randomised, double-blind, placebo-controlled trial.
CTID: null
Phase: Phase 3 Status: GB - no longer in EU/EEA
Date: 2020-12-17
PEROPERATIVE TRANEXAMIC ACID IN BARIATRIC FAST TRACK SURGERY TO REDUCE
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2020-11-05
A multi-centre randomised, double-blind, placebo-controlled trial of pre-hospital treatment with tranexamic acid for severely injured
CTID: null
Phase: Phase 3 Status: Completed
Date: 2020-11-03
TRAnexamic Acid for Preventing blood loss following a cesarean delivery in women with placenta pREVIA: a multicenter randomised, double blind placebo controlled trial (TRAAPrevia)
CTID: null
Phase: Phase 3 Status: Trial now transitioned
Date: 2020-03-12
Heavy menstrual bleeding in premenopausal women treated with direct oral anticoagulants - the MEDEA study
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2019-11-12
Pharmacokinetics of Tranexamic Acid after oral, intramuscular or intravenous administration: a prospective, randomised, cross-over trial in healthy volunteers.
CTID: null
Phase: Phase 2 Status: Completed
Date: 2019-08-21
Pharmacokinetics of intramuscular tranexamic acid in trauma patients: a clinical trial
CTID: null
Phase: Phase 2 Status: Completed
Date: 2019-08-01
Different dosing of Tranexamic Acid in patients undergoing elective total hip or knee arthroplasty. A randomized, controlled, double-blinded clinical trial.
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2019-02-18
Study of thromboelastography during tranexamic acid treatment in preventing bleeding in patients with haematological malignancies presenting severe thrombocytopenia (TTRAP-bleeding)
CTID: null
Phase: Phase 2 Status: Completed
Date: 2019-02-12
PeriOperative ISchemic Evaluation-3 (POISE-3) Trial
CTID: null
Phase: Phase 3 Status: Ongoing, Completed
Date: 2018-12-11
Efficacy and safety of levocetirizine alone or in combination with tranexamic acid in the treatment of spontaneous chronic urticaria. Multicentric controlled randomized study in cross-over, double-blind.
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2018-12-04
TRANSFUSION OF RED BLOOD CELLS, TRANEXAMIC ACID AND FIBRINOGEN CONCENTRATE FOR SEVERE TRAUMA HEMORRHAGE AT PRE-HOSPITAL PHASE OF CARE. A PILOT TRIAL.
CTID: null
Phase: Phase 2 Status: Completed
Date: 2018-09-27
Tranexamic acid use to reduce de blood transfusion in femur fracture patients. Clinical randomized trial with placebo control
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2018-07-12
Tranexamic Acid to Prevent Operation in Chronic Subdural Hematoma
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2018-05-24
Reduction of bleeding during abdominal plastids with Exacyl®
CTID: null
Phase: Phase 4 Status: Completed
Date: 2018-05-14
Tranexamic Acid to reduce bleeding in patients treated with new oral anticoagulants undergoing dental extraction (EXTRACT-NOAC)
CTID: null
Phase: Phase 4 Status: Prematurely Ended
Date: 2018-01-03
TRAnexamic Acid for Preventing postpartum hemorrhage following a Cesarean Delivery :a multicenter randomised, double blind placebo controlled trial (TRAAP2)
CTID: null
Phase: Phase 3 Status: Completed
Date: 2017-11-23
Randomized, placebo-controlled, double-blind trial to evaluate the efficacy and safety of topical application of tranexamic acid for saving blood losses in patients subjected to prosthetic knee surgery.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2017-11-13
A randomised controlled trial of topical intranasal tranexamic acid versus placebo to reduce the need for nasal packing in patients presenting to the Emergency Department with spontaneous epistaxis.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2017-02-20
Randomized, double-blind, 3-arm trial to evaluate the impact of blood loss following administration of three tranexamic acid regimens in patients undergoing bone or soft tissue sarcomas.
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2017-02-09
TREATT: TRial to EvaluAte Tranexamic acid therapy in Thrombocytopenia
CTID: null
Phase: Phase 3 Status: GB - no longer in EU/EEA
Date: 2016-08-23
Human intestinal ischemia and reperfusion
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2016-06-06
Single centre randomised controlled trial to assess the effect of the addition of twenty-four hours of oral tranexamic acid post-operatively to a single intra-operative intravenous dose of tranexamic acid on calculated blood loss following primary hip and knee arthroplasty.
CTID: null
Phase: Phase 4 Status: Completed
Date: 2016-03-10
TIME OF ADMINISTRATION OF TRANEXAMIC ACID TO PREVENT BLEEDING IN TOTAL KNEE ARTHROPLASTY
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2016-03-10
Tranexamic acid to reduce blood loss in hemorrhagic caesarean delivery: a multicenter randomized double blind placebo
CTID: null
Phase: Phase 3 Status: Prematurely Ended
Date: 2016-02-02
Effects of Tranexamic Acid on on Intra- and Postoperative bleeding, Blood transfusion and Coagulation in Children Undergoing Craniosynostosis Surgery
CTID: null
Phase: Phase 4 Status: Completed
Date: 2015-08-05
Etude ESPER : Epargne Sanguine au cours de la pose d’une Prothèse de hanche totale grâce à l’Exacyl® chez le patient traité par Rivaroxaban
CTID: null
Phase: Phase 4 Status: Completed
Date: 2015-03-25
Tranexamic acid in major vascular surgery (Tranex-AAA)
CTID: null
Phase: Phase 4 Status: Completed
Date: 2015-01-05
The Effectiveness and Population Pharmacokinetics and Pharmacogenomics of a Reduced Dose of Tranexamic Acid for Craniosynostosis Surgery: A multicenter study. The TXA Study.
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2014-11-24
TRAnexamic Acid for Preventing postpartum hemorrhage
CTID: null
Phase: Phase 3 Status: Completed
Date: 2014-08-21
A ONE YEAR STUDY ON THE USE OF TRANEXAMIC ACID IN BENIGN PROSTATIC HYPERPLASIA SURGERY
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2014-05-07
Prevention of bleeding and edema in bi-maxillary orthognathic surgery; the effectiveness of tranexamic acid on intraoperative bleeding in orthognathic surgery
CTID: null
Phase: Phase 4 Status: Completed
Date: 2014-04-24
Topical application of tranexamic acid compared to the intravenous administration in total hip arthroplasty with the direct anterior approach. A prospective, randomized, clinical trial.
CTID: null
Phase: Phase 2 Status: Ongoing
Date: 2013-12-19
STOP-AUST: The Spot sign and Tranexamic acid On Preventing ICH growth – AUStralasia Trial
CTID: null
Phase: Phase 2 Status: Completed
Date: 2013-12-10
Evaluation of intra articular Tranexamic acid for reduction of total blood loss in total knee unilateral arthroplasty
CTID: null
Phase: Phase 4 Status: Completed
Date: 2013-10-30
Evaluation of postoperative administration of tranexamic acid on reducing blood loss after hip prothesis surgery.
CTID: null
Phase: Phase 4 Status: Completed
Date: 2013-08-08
The effect on blood loss with combined topical and intravenous tranexamic acid in cardiac surgery patients: a randomized double-blind placebo-controlled trial
CTID: null
Phase: Phase 4 Status: Ongoing
Date: 2013-04-24
Pilot study on efficacy of bilastine in preventing angioedema symptoms in patients with angioedema of unknown etiology (idiopathic angioedema, IAE)
CTID: null
Phase: Phase 2 Status: Prematurely Ended
Date: 2013-03-23
Tranexamic acid for the treatment of gastrointestinal haemorrhage: an international randomised, double blind placebo controlled trial
CTID: null
Phase: Phase 3 Status: Prematurely Ended, Completed
Date: 2013-02-22
Peroperative Tranexamic acid as prophylaxis of bleeding related to benign hysterectomy - a randomized, placebo-controlled trial
CTID: null
Phase: Phase 4 Status: Completed
Date: 2013-02-07
Tranexamic Acid for the treatment of significant traumatic brain injury: an international, randomised, double blind, placebo controlled trial.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2012-12-03
Tranexamic acid for hyperacute primary IntraCerebral Haemorrhage TICH-2
CTID: null
Phase: Phase 3 Status: Completed
Date: 2012-11-23
Efficacy of topical tranexamic acid versus intravenous administration to reduce blood transfussion rate in total knee arthroplasty surgery: phase III, unicentric, controlled,double blind, randomized non inferiority clinical trial.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2012-11-08
Ultra-early tranexamic acid after subarachnoid hemorrhage.
CTID: null
Phase: Phase 3 Status: Completed
Date: 2012-09-06
Prevention of postoperative bleeding in bone tumors surgery: A multicenter, randomized, parallel controlled clinical trial evaluating the efficacy of topic fibrin glue, topic tranexamic acid and normal haemostasia in patients undergoing massive bone resection.
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2012-07-09
Prevention of postoperative bleeding: A multicenter, randomized, parallel, controlled clinical trial, evaluating the efficacy of tranexamic acid and fibrin glue in patients undergoing interventions for sub-capital femoral fracture.
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2012-07-09
A Phase II, double-blind, parallel group, dose-selection study to compare antifibrinolytic MDCO-2010 vs. placebo and tranexamic acid in reducing blood loss in patients undergoing primary cardiac surgery
CTID: null
Phase: Phase 2 Status: Prematurely Ended
Date: 2012-04-03
Prevención del sangrado postoperatorio en artroplastia total de rodilla: Ensayo clínico multicéntrico, aleatorizado, abierto, paralelo de tres brazos de tratamiento, que evalúa la eficacia del ácido tranexámico endovenoso y tópico versus hemostasia habitual
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2011-11-30
International, prospective, double-blind, 3-arm comparative, randomized, placebo controlled phase IV study on the effect of counseling and either tranexamic acid or
CTID: null
Phase: Phase 4 Status: Completed
Date: 2011-03-01
Tranexamic acid for the treatment of postpartum haemorrhage: An international randomised, double blind, placebo controlled trial
CTID: null
Phase: Phase 3 Status: Completed
Date: 2011-02-17
ACIDO TRANEXAMICO FRENTE A PLACEBO PARA LA REDUCCION DE LAS PERDIDAS SANGUINEAS EN LA CIRUGIA DE REEMPLAZO TOTAL DE CADERA: ENSAYO CLINICO ALEATORIZADO
CTID: null
Phase: Phase 3 Status: Ongoing
Date: 2010-11-12
A randomis
e.querySelector("font strong").innerText = 'View More'
} else if(up_display === 'none' || up_display === '') {
icon_angle_down.style.display = 'none';