| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 50mg |
|
||
| 100mg |
|
||
| Other Sizes |
|

| 靶点 |
The biological effects of valine involve multiple intracellular targets. Its primary targets include the mTOR (mechanistic Target of Rapamycin) pathway, involved in cell growth and metabolism regulation; HDAC6 (Histone Deacetylase 6) , for which valine acts as a specific ligand参与 DNA repair processes; and key metabolic enzymes such as BCAT (Branched-Chain Amino Acid Aminotransferase) , VARS (Valine--tRNA Ligase) , and mitochondrial Propionyl-CoA carboxylase.
|
|---|---|
| 体外研究 (In Vitro) |
体外研究表明,缬氨酸能显著改善线粒体功能。它能上调参与线粒体生物合成和动力学的基因表达,并增强电子传递链复合物I、II和IV的活性。此外,L-缬氨酸是GLP-1分泌的强效刺激剂,它通过调节ATP敏感性钾通道(K_ATP)和电压门控钙通道(CaV)来促进激素释放。L-缬氨酸还能通过激活PI3K/Akt信号通路和抑制精氨酸酶,对多重耐药菌表现出抑制活性。
|
| 体内研究 (In Vivo) |
缬氨酸的功效已在动物模型中得到广泛验证。例如,口服L-缬氨酸(1 g/kg)可有效提高雄性小鼠血浆中活性GLP-1的水平。在啮齿动物肝损伤模型中,补充缬氨酸(1.7 g/kg/天)显著恢复了肝功能指标,降低了氧化应激,并改善了肝脏组织病理学。此外,膳食缬氨酸通过改善肠绒毛形态、增强肠道屏障功能和抑制GCN2介导的炎症反应来改善肠道健康。
|
| 酶活实验 |
为了评估含缬氨酸前药与靶蛋白的结合亲和力,分子对接是主要的研究方法。以缬氨酸酯前药与PepT1受体的结合为例,具体流程如下:蛋白准备:从PDB数据库获取人PepT1(例如SLC15A1)的晶体结构,添加氢原子并优化侧链。
配体准备:使用ChemDraw绘制缬氨酸酯前药的分子结构,并在MOE或Schrödinger软件中生成三维构象,然后进行能量最小化。 对接框定义:在肽转运蛋白的底物结合口袋内定义结合位点,使用大小足以覆盖整个结合腔的网格框。 分子对接计算:使用AutoDock Vina或Glide进行标准对接计算。 结果分析与评分:基于对接能量(ΔG_binding)和氢键相互作用,比较前药系列与母体化合物的结合亲和力差异,选择最佳结合构象。 |
| 细胞实验 |
缬氨酸体外细胞检测方案如下:
细胞培养:将GLUTag细胞(一种小鼠肠道内分泌细胞系)培养于含10%胎牛血清的高糖DMEM培养基中,置于37℃、5% CO₂培养箱中培养。 药物处理:当细胞汇合度达到70-80%时,将培养基更换为含有不同浓度L-缬氨酸(例如0-50 mM)的Krebs-Ringer缓冲液。 抑制剂干预:为研究特定机制,在缬氨酸处理前,将细胞预先与通道抑制剂(例如10 µM硝苯地平阻断CaV通道,或250 µM二氮嗪开放K_ATP通道)孵育。 样品收集:收集细胞培养上清液,用于ELISA法测定GLP-1水平。同时,裂解细胞以提取总蛋白,用于Western Blot分析信号通路蛋白(例如PI3K/Akt)。 数据分析:使用微孔板读数仪测量吸光度,并使用GraphPad Prism软件进行统计分析和EC50计算。 |
| 动物实验 |
动物准备:选择8-12周龄的雄性C57BL/6J小鼠,禁食过夜,可自由饮水。
给药:通过灌胃给予L-缬氨酸(1 g/kg),阳性对照组给予葡萄糖(2 g/kg)。 给药后观察:分别于给药后0、15、30、60和120分钟,从眼眶后静脉丛采集血样,置于含有DPP-4抑制剂的EDTA抗凝管中。 样本检测:通过离心(4℃,3000 rpm,10分钟)分离血浆。采用商业化的ELISA试剂盒定量检测活性GLP-1和胰岛素浓度。 数据分析:计算每个时间点GLP-1和胰岛素浓度的平均值±标准误(SEM),计算曲线下面积(AUC),并使用单因素方差分析(ANOVA)比较差异。 |
| 药代性质 (ADME/PK) |
吸收、分布和排泄
支链氨基酸 (BCAA) 通过钠依赖性主动转运从小肠吸收。多种疾病和异常生理状态会改变血液和组织中 BCAA 的浓度,包括糖尿病、肝功能障碍、饥饿、蛋白质能量营养不良、酒精中毒和肥胖。这些以及其他一些情况有时会导致血浆 BCAA 池发生剧烈变化。/氨基酸/ 虽然溶解在体液中的游离氨基酸仅占体内总氨基酸的一小部分,但它们对于人体蛋白质的营养和代谢控制至关重要。……虽然血浆是最容易获得的样本,但大多数氨基酸在组织细胞内的细胞内池中浓度更高。通常,大型中性氨基酸,例如亮氨酸和苯丙氨酸,与血浆中的浓度接近平衡。其他氨基酸,特别是谷氨酰胺、谷氨酸和甘氨酸,其细胞内浓度比血浆浓度高10到50倍。饮食变化或病理状况可导致血浆和组织中各种游离氨基酸浓度发生显著变化。摄入后,蛋白质在胃酸的作用下变性,并被胃蛋白酶水解成较小的肽。进食后胃酸增多会激活胃蛋白酶。蛋白质和肽随后进入小肠,在那里,肽键被多种酶水解。这些特定的水解酶来源于胰腺,包括胰蛋白酶、糜蛋白酶、弹性蛋白酶和羧肽酶。由此产生的游离氨基酸和小肽混合物随后通过各种载体系统运输到黏膜细胞,每个载体系统靶向特定的氨基酸、二肽和三肽,且每个系统仅靶向有限范围的肽底物。吸收的肽在细胞内水解后,游离氨基酸随后通过黏膜细胞内的其他特定载体系统分泌到门静脉血流中,或在细胞内进一步代谢。吸收的氨基酸进入肝脏,部分被吸收利用;其余部分进入体循环,被外周组织利用。即使在无蛋白饮食中,蛋白质仍会继续分泌到肠道,粪便氮丢失(即以细菌形式随粪便丢失的氮)可占必需氮丢失量的25%。在这些饮食条件下,作为蛋白水解酶成分分泌到肠道的氨基酸以及脱落的黏膜细胞中的氨基酸是维持肠道细菌生物量的唯一氨基酸来源。其他氨基酸丢失途径包括尿液排泄以及皮肤和毛发脱落。这些丢失量小于上述途径,但仍会对需求量的估算产生显著影响,尤其是在疾病状态下。 /氨基酸/ 有关L-缬氨酸(8种类型)的吸收、分布和排泄的更完整数据,请访问HSDB记录页面。 代谢/代谢物 肝脏 支链氨基酸(BCAA)——亮氨酸、异亮氨酸和缬氨酸——与其他大多数必需氨基酸不同,负责其分解代谢的酶主要位于肝外组织。每种酮酸都会经历由支链氨基转移酶(BCAT)催化的可逆氨反应,生成α-酮异己酸(KIC,源自亮氨酸)、α-酮-β-甲基戊酸(KMV,源自异亮氨酸)和α-酮异戊酸(KIV,源自缬氨酸)。这些酮酸随后在位于线粒体膜上的多酶系统——支链酮酸脱氢酶(BCKADs)的催化下发生不可逆的氧化脱羧反应。这些氧化反应的产物进一步转化为乙酰辅酶A、丙酰辅酶A、乙酰乙酸和琥珀酰辅酶A。因此,支链氨基酸(BCAAs)既具有生酮作用,又具有生糖作用。氨基酸的脱氨产物一旦进入三羧酸循环(TCA循环,也称为柠檬酸循环或克雷布斯循环)或糖酵解途径,其碳骨架便可用于生物合成途径,特别是葡萄糖和脂肪的合成。氨基酸的碳骨架最终生成葡萄糖还是脂肪取决于其进入的途径。如果以乙酰辅酶A的形式进入,则只能生成脂肪或酮体。然而,其他氨基酸的碳骨架可以通过某种方式进入这些代谢途径,使其碳原子可用于糖异生。这是经典营养学中将氨基酸描述为生酮氨基酸或生糖氨基酸(即能够产生酮体[或脂肪]或葡萄糖)的基础。一些氨基酸在降解过程中会同时产生这两种产物,因此被认为是生酮氨基酸和生糖氨基酸。/氨基酸/ ... 采用核磁共振分析鉴定富含星形胶质细胞(APC)的原代培养物在U-(13)C-缬氨酸分解代谢过程中产生的(13)C标记代谢物,这些代谢物随后释放到培养基中。结果表明,APC (1) 能有效清除培养基中的缬氨酸;(2) 将缬氨酸降解为琥珀酰辅酶A,琥珀酰辅酶A是三羧酸循环(TCA循环)的成员; (3) 将缬氨酸代谢产物及其衍生物,例如 U-(13)C-2-氧代异戊酸、U-(13)C-3-羟基异丁酸、U-(13)C-2-甲基丙二酸、[U-(13)C]异丁酸和 [U-(13)C]丙酸,以及几种三羧酸循环依赖性代谢物(包括乳酸)释放到细胞外环境中。 ……采用灌注法,用 (15)N 标记的亮氨酸、异亮氨酸或缬氨酸,研究了培养的小脑星形胶质细胞中支链氨基酸 (BCAA) 的代谢。为了模拟谷氨酸能突触活动期间的条件,一些细胞培养物暴露于谷氨酸脉冲(50 μM;每 2 分钟一次,持续 10 秒;总共 75 分钟)……来自 (15)N-亮氨酸、(15)N-异亮氨酸或 (15)N-缬氨酸的 (15)N 掺入细胞内谷氨酸的比例约为 40-50%,不同支链氨基酸 (BCAA) 之间的差异很小。有趣的是,暴露于外源性谷氨酸后,来自 (15)N-缬氨酸的谷氨酸标记百分比并未降低,这与来自 (15)N-亮氨酸或 (15)N-异亮氨酸的标记百分比显著降低形成鲜明对比。这表明,在重复暴露于谷氨酸期间,只有缬氨酸相关的转氨作用被上调。研究表明,缬氨酸可能作为一种重要的氨基酸,在神经元和星形胶质细胞之间运输,而这种功能可能在突触活动期间上调。本研究检测了健康人和肝硬化患者肝脏中缬氨酸代谢途径关键酶的活性,包括支链氨基转移酶、支链α-酮酸脱氢酶复合物、甲基丙烯酰辅酶A (MC-CoA) 水合酶(巴豆酰辅酶A酶)和3-羟基异丁酰辅酶A (HIB-CoA) 水解酶。与缺乏支链氨基转移酶的大鼠肝脏不同,健康人肝脏中可检测到支链氨基转移酶活性,且肝硬化患者肝脏中该酶活性略有升高。健康人肝脏中支链α-酮酸脱氢酶复合物的总活性约为大鼠肝脏的1%,而该复合物的活性形式在健康肝脏和肝硬化肝脏中均占20%至30%。肝硬化仅显著降低了该酶的实际活性。这些结果表明,人肝脏对支链氨基酸和α-酮酸的分解代谢活性低于大鼠肝脏。与支链α-酮酸脱氢酶复合物相比,人肝脏中MC-CoA水合酶和HIB-CoA水解酶的活性非常高,提示这些酶在潜在毒性化合物MC-CoA(缬氨酸和异丁酸分解代谢的中间体)的分解代谢中发挥重要作用。肝硬化导致HIB-CoA水解酶活性显著降低,但对柠檬酸合酶活性无影响。这表明HIB-CoA水解酶活性的降低并非反映线粒体数量的普遍减少,而是可能导致细胞损伤,最终导致肝功能衰竭。缬氨酸的药代动力学特性主要体现在其提高前药口服生物利用度的能力上,而非其自身作为氨基酸的代谢。药物化学的一个重点是将缬氨酸作为前药设计中的“前体部分”,通过靶向肠道肽转运蛋白PepT1来增强口服吸收。例如,在大鼠口服阿昔洛韦的缬氨酸-缬氨酸二肽前药(VVACV)后,活性代谢物阿昔洛韦的血浆浓度-时间曲线下面积(AUC)比口服阿昔洛韦本身高出近6倍,显著提高了其全身暴露量。 |
| 毒性/毒理 (Toxicokinetics/TK) |
毒性概述
(适用于缬氨酸、亮氨酸和异亮氨酸) 这组必需氨基酸已被鉴定为支链氨基酸(BCAA)。由于人体无法合成这种碳原子排列,因此这些氨基酸必须从膳食中摄取。这三种化合物的分解代谢均始于肌肉,产生NADH和FADH2,它们可用于生成ATP。这三种氨基酸的分解代谢在前两步中使用相同的酶。每种氨基酸的第一步都是转氨反应,使用相同的BCAA氨基转移酶,以α-酮戊二酸为胺受体。该反应生成三种不同的α-酮酸,它们被相同的支链α-酮酸脱氢酶氧化,生成三种不同的辅酶A衍生物。随后,代谢途径发生分支,产生许多中间产物。缬氨酸的主要产物是丙酰辅酶A,它是琥珀酰辅酶A的生糖前体。异亮氨酸的分解代谢最终产生乙酰辅酶A和丙酰辅酶A;因此,异亮氨酸既是生糖物质又是生酮物质。亮氨酸产生乙酰辅酶A和乙酰乙酰辅酶A,因此严格来说是一种生酮物质。许多遗传性疾病与支链氨基酸(BCAA)的分解代谢异常有关。最常见的缺陷是支链α-酮酸脱氢酶缺乏。由于三种氨基酸各只有一种脱氢酶,因此所有三种α-酮酸都会积累并随尿液排出。这种疾病被称为枫糖尿症,因为患者的尿液具有特征性的枫糖浆气味。这些病例通常伴有严重的智力障碍。遗憾的是,由于这些是必需氨基酸,因此不能在饮食中严格限制其摄入量。最终,患者寿命较短,并伴有发育异常。主要的神经系统问题是由于中枢神经系统髓鞘形成不良所致。 相互作用……高亮氨酸饮食会抑制低蛋白饮食喂养的大鼠的生长,而补充异亮氨酸和缬氨酸则可以阻止这种生长抑制。 支链氨基酸(BCAA)与其他大中性氨基酸(LNAA,尤其是色氨酸和酪氨酸)竞争膜转运已被证实。虽然支链氨基酸(BCAA)并非神经递质的直接前体,但它们可以影响某些大中氨基酸(LNAA)跨血脑屏障的转运,从而影响中枢神经系统中某些神经递质的浓度。 雄性Wistar大鼠急性暴露于百草枯(3、5、10、20 mg/kg,皮下注射)后,特定脑区运动行为模式和单胺水平的变化……结果表明,5、10和20 mg/kg剂量的百草枯显著降低了大鼠的运动、刻板和旋转行为。皮质和下丘脑中去甲肾上腺素 (NE) 水平显著降低,纹状体中多巴胺 (DA) 及其酸性代谢物水平也显著降低……腹腔注射 L-缬氨酸(200 mg/kg)可显著降低中等剂量 (5 mg/kg) 百草枯的毒性,但高剂量 (20 mg/kg) 则无此作用…… 非人类毒性值 大鼠腹腔注射 LD50 为 5390 mg/kg 缬氨酸作为一种天然氨基酸,通常被认为可安全用于日常服用,但过量摄入或特定病理情况下,其潜在毒性仍需引起关注。动物毒性研究表明,大鼠急性腹腔注射 D-缬氨酸的 LD50 为 6,093 mg/kg,其毒性水平与氯化钠(食盐)相当。然而,长期摄入高浓度缬氨酸(饲料中缬氨酸含量为0.74%-0.79%)会促进脂肪生成并抑制脂肪酸氧化,从而加速非酒精性脂肪肝(NAFLD)的发展,并诱发肝损伤。在罕见的遗传性疾病ECHS1缺乏症中,缬氨酸代谢受损会导致有毒代谢产物的积累,从而加剧癫痫和神经系统损伤。 |
| 参考文献 | |
| 其他信息 |
治疗用途
支链氨基酸 (BCAA) 是具有兴奋作用的必需氨基酸。它们促进肌肉生长和组织修复,并且是青霉素生物合成途径的前体。 它们被用作膳食补充剂,也是许多用于治疗肝病的制剂的成分。 富含 BCAA 的蛋白质或氨基酸混合物,在某些情况下,单独使用 BCAA,已被用于治疗多种代谢性疾病。这些氨基酸因其能够减少大脑对芳香族氨基酸的吸收,并提高慢性肝病和肝性脑病患者体内 BCAA 的低水平而备受关注。它们也用于败血症和其他异常患者的肠外营养。 药物警告 本研究旨在评估枫糖尿症 (MSUD) 患者在长期观察期间对限制 BCAA 含量饮食的依从性。研究组由7名年龄在1.5至18岁之间的儿童组成。营养评估基于每3至4个月采集一次的3至4天的膳食记录。……除钙以外,推荐每日食物清单中大多数营养素的能量和含量均符合推荐摄入量(RDI)。对患有枫糖尿症(MSUD)的儿童进行膳食分析显示,他们缺乏铁、锌、铜、维生素B1、B2、烟酸和维生素C(通常低于RDI的90%)。/支链氨基酸/ 研究人员测量了5888名新生儿和20名年龄在1至20岁之间、疑似患有代谢紊乱的受试者的氨基酸水平,发现了一例由缬氨酸中间代谢紊乱引起的“枫糖尿症”。研究人员从患者出生后的第一天起就追踪其血清和尿液中的缬氨酸浓度。该患者还表现出频繁的低血糖发作。经过18个月的早期复合维生素、矿物质和微量元素治疗后,部分受损的酶系统得以恢复,血清和尿液中的缬氨酸和葡萄糖水平也恢复正常。同样的治疗方法对一岁以上的患者效果显著降低,这支持了维生素和矿物质仅在“枫糖尿症”发作后立即服用才有效的结论…… 药效学L-缬氨酸是一种具有兴奋作用的支链必需氨基酸(BCAA)。它能促进肌肉生长和组织修复,并且是青霉素生物合成途径的前体。缬氨酸是三种支链氨基酸之一(另外两种是亮氨酸和异亮氨酸),它们可以增强能量、提高耐力,并有助于肌肉组织的恢复和修复。这类氨基酸还可以降低升高的血糖水平,并促进生长激素的分泌。补充缬氨酸时,应始终以 2:1:2 的毫克比例与异亮氨酸和亮氨酸一起服用。缬氨酸是蛋白质中一种必需氨基酸,对婴儿的最佳生长、儿童的生长以及成人的氮平衡至关重要。L-缬氨酸缺乏会影响身体发育,导致神经系统疾病和贫血。它在制药和食品工业中有着广泛的应用。 |
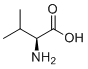
| 分子式 |
C5H11NO2
|
|---|---|
| 分子量 |
117.14
|
| 精确质量 |
117.078
|
| CAS号 |
72-18-4
|
| 相关CAS号 |
D-Valine;640-68-6;L-Valine-15N;59935-29-4;L-Valine-13C5,15N;202407-30-5;L-Valine-1-13C;81201-85-6;L-Valine-13C5;55443-52-2;L-Valine-13C5,15N,d8;1994261-62-9;L-Valine-13C5,15N,d2;201417-09-6;L-Valine-2-13C;73834-52-3;L-Valine-1-13C,15N;87019-54-3;L-Valine-d;77257-03-5;L-Valine-15N,d8
|
| PubChem CID |
6287
|
| 外观&性状 |
White to off-white solid powder
|
| 密度 |
1.1±0.1 g/cm3
|
| 沸点 |
213.6±23.0 °C at 760 mmHg
|
| 熔点 |
315ºC
|
| 闪点 |
83.0±22.6 °C
|
| 蒸汽压 |
0.1±0.9 mmHg at 25°C
|
| 折射率 |
1.461
|
| LogP |
0.2
|
| tPSA |
63.32
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
3
|
| 可旋转键数目(RBC) |
2
|
| 重原子数目 |
8
|
| 分子复杂度/Complexity |
90.4
|
| 定义原子立体中心数目 |
1
|
| SMILES |
CC(C)[C@@H](C(=O)O)N
|
| InChi Key |
KZSNJWFQEVHDMF-BYPYZUCNSA-N
|
| InChi Code |
InChI=1S/C5H11NO2/c1-3(2)4(6)5(7)8/h3-4H,6H2,1-2H3,(H,7,8)/t4-/m0/s1
|
| 化学名 |
L-valine
|
| 别名 |
ValineL-Valine NSC-76038 Valina EC 200-773-6 NSC76038NSC 76038
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
H2O : ~20 mg/mL (~170.72 mM)
|
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: 25 mg/mL (213.40 mM) in PBS (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶。 (<60°C).
请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 8.5368 mL | 42.6840 mL | 85.3679 mL | |
| 5 mM | 1.7074 mL | 8.5368 mL | 17.0736 mL | |
| 10 mM | 0.8537 mL | 4.2684 mL | 8.5368 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT06581991 | NOT YET RECRUITING | Dietary Supplement: Valine oral solution (50mg/ml) and isoleucine oral solution (25mg/ml) | Maple Syrup Urine Disease | Meta Healthcare Ltd | 2024-10 | Not Applicable |
| NCT06372314 | NOT YET RECRUITING | Drug: Total parenteral nutrition (TPN): this is total nutrition provided by central vein. | Stable Neonates Receiving Total Parenteral Nutrition (TPN) | The Hospital for Sick Children | 2024-08 | Not Applicable |
| NCT05705414 | NOT YET RECRUITING | Drug: Valine Drug: EEA |
End Stage Renal Disease | The University of Texas Health Science Center at San Antonio | 2024-09 | Early Phase 1 |
| NCT02305056 | TERMINATED | Drug: C13 N15 Valine | High-grade Glioma | University Hospital, Grenoble | 2014-09 | Phase 1 |
| NCT01495871 | WITHDRAWN | Dietary Supplement: Amino Acids Dietary Supplement: Placebo of inert compounds Dietary Supplement: Valine |
Traumatic Brain Injury | The University of Texas Medical Branch, Galveston | 2011-11 | Not Applicable |