| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 500mg |
|
||
| 5g |
|
||
| 50g |
|
||
| Other Sizes |
|
| 靶点 |
Selective inhibitor of neuronal nitric oxide synthase (nNOS). Competitive inhibition with a Ki of 20 μM. Shows low activity against inducible NOS (iNOS, <25% inhibition at 500 μM) and endothelial NOS (eNOS, <15% inhibition at 500 μM). [1]
2-Thiouracil does not target a specific enzyme or receptor. Its mechanism of action involves nucleophilic addition to quinonoid intermediates in the melanin biosynthesis pathway, specifically: dopaquinone (forming the 6-S-thiouracil-DOPA adduct), 5,6-indolequinone from DHI (forming C-2 and C-3 substituted adducts), and the o-indolequinone from DHICA (forming a C-4 substituted adduct). The provided document does not include IC50, Ki, or EC50 values for these interactions. [2] 2-Thiouracil exerts its effects through multiple mechanisms. Its primary classical target is thyroid peroxidase (TPO), the enzyme responsible for iodination of thyroglobulin in thyroid hormone synthesis . By inhibiting TPO, the compound blocks the production of thyroxine (T4) and triiodothyronine (T3), thereby reducing thyroid hormone levels. In the context of melanoma targeting, 2-thiouracil acts as a substrate for tyrosinase, an enzyme involved in melanin biosynthesis. It undergoes nucleophilic attack on transient quinonoid intermediates in the melanin pathway—primarily dopaquinone and 5,6-indolequinones—leading to covalent incorporation into growing melanin polymers . Additionally, 2-thiouracil has been identified as a selective inhibitor of neuronal nitric oxide synthase (nNOS), antagonizing tetrahydrobiopterin-dependent enzyme activation and dimerization . |
|---|---|
| 体外研究 (In Vitro) |
重组大鼠 nNOS 与 TU 孵育后,通过 L-[14C]瓜氨酸生成测定法测得,其酶活性受到浓度依赖性抑制,IC50 为 50 ± 5 μM。[1]
在测定条件下,1 mM 的 TU 可使重组 nNOS 的 NO 生成(通过硝酸盐/亚硝酸盐积累测量)抑制 51 ± 2%。[1] 在 100 μM 浓度下,TU 在缺乏 L-精氨酸和四氢生物蝶呤 (BH4) 的条件下,可抑制约 60% 的 nNOS 产生的 H2O2。这种抑制对氧合酶结构域活性具有特异性,因为在相同浓度下,TU 不影响 nNOS 的细胞色素 c 还原酶活性(还原酶结构域功能)。[1] 低温度 SDS-PAGE 实验证明,TU (500 μM) 拮抗了 BH4 诱导的 nNOS 二聚化,减少了在 1 μM BH4 存在下形成的 320 kDa 二聚体条带量。当 BH4 浓度提高到 10 μM 时,这种对二聚化的抑制作用被消除。[1] - 与 DOPA/酪氨酸的反应: 在 TU(3 mM,2倍摩尔过量)存在下,由酪氨酸酶催化氧化 DOPA(1.5 mM)时,反应过程发生显著改变。除产生少量 DHI 外,还形成了一组复杂的产物。主要产物被鉴定为 6-S-硫代尿嘧啶-多巴。其他峰(III-VIII)对应于 DHI-TU 加合物。[2] - 与 DHI 的反应: 在 TU(3 mM)存在下,由酪氨酸酶氧化 DHI(1.5 mM)时,DHI 的正常寡聚化模式几乎被完全抑制。取而代之地,分离并鉴定出了新产物:C-3 位取代的 1:1 DHI-TU 加合物(化合物 1)、C-2 位取代的 1:1 加合物(化合物 2)、通过 C-4 和 C-4' 位连接的对称二聚体(化合物 3),以及含两个 TU 单元的三聚体(化合物 4)。化合物 1-4 的总分离产率约为 20%。[2] - 与 DHICA 的反应: 在 TU(3 mM)存在下,由酪氨酸酶氧化 DHICA(1.5 mM),导致形成 DHICA-TU 结合物。分离出了 C-4 位取代的 1:1 加合物(化合物 5)、含一个 TU 单元的二聚体(化合物 6)以及含一个 TU 单元的三聚体(化合物 7)。[2] - TU-吲哚加合物的反应性: 加合物 1、2 和 5 经酪氨酸酶酶促氧化后,平稳分解为灰白色物质,在 400-600 nm 范围内表现为无特征吸收,无明显的不溶性黑色素样色素散射,表明其黑色素生成能力较差。[2] 体外研究表明,2-硫代尿嘧啶衍生物,特别是作为配体与钌(II)形成配合物后,对癌细胞系表现出强效的细胞毒性作用。用钌(II)-2-硫代尿嘧啶配合物处理HepG2肝细胞癌细胞后,检测到磷脂酰丝氨酸暴露增加、线粒体跨膜电位丧失、PARP裂解、DNA片段化、染色质凝集和胞质收缩。机制研究表明,这些配合物通过靶向NF-κB和Akt/mTOR信号通路抑制肝癌干细胞。在黑色素生成研究中,当在酪氨酸酶存在下与多巴共孵育时,2-硫代尿嘧啶显著改变了正常的黑色素生物合成,促进了加成产物的形成,包括6-S-硫代尿嘧啶-多巴和各种DHI-TU加合物。 |
| 体内研究 (In Vivo) |
- 掺入黑色素瘤黑色素: 将 [2-¹⁴C] 标记的 2-硫代尿嘧啶 腹腔注射到荷 B16 黑色素瘤小鼠体内后,大部分掺入发生在 30 分钟内。注射后 30 分钟,比掺入量为 25,000 cpm/mg 黑色素;90 分钟为 23,000 cpm/mg;180 分钟为 23,000 cpm/mg。TU 给药导致提取的黑色素的 A₃₅₀ 吸光度显著降低(从对照组的 0.071 ± 0.005 降至 180 分钟时的 0.043 ± 0.004),表明色素结构发生改变。对肿瘤黑色素进行化学降解后,PTCA(吡咯-2,3,5-三羧酸)的产率在 TU 注射后也降低(从对照组的 1125 ± 62 ng/mg 降至 180 分钟时的 589 ± 40 ng/mg)。[2]
在荷黑色素瘤小鼠的体内研究中,注射放射性标记的2-硫代尿嘧啶导致该药物快速并选择性地掺入肿瘤色素中。这种掺入显著降低了350 nm处的吸光度值以及黑色素标志物吡咯-2,3,5-三羧酸的产量。在肝细胞癌异种移植模型(C.B-17 SCID小鼠接种HepG2细胞)中,钌(II)-2-硫代尿嘧啶配合物(2或4 mg/kg,腹腔注射,每日一次,连续21天)抑制了肿瘤生长,根据具体配合物和剂量的不同,肿瘤抑制率为53.6%-65.4%。治疗动物在体重或重要器官(肝、肾、肺、心)方面未见显著变化,表明全身毒性可耐受。对黑色素性黑色素细胞的研究表明,更亲脂性的丙基硫氧嘧啶类似物比2-硫代尿嘧啶本身表现出更高的细胞蓄积性和毒性。 |
| 酶活实验 |
主要的 NOS 活性测定中,将重组 nNOS 在含有 Tris-HCl 缓冲液 (pH 7)、NADPH、氯化钙、BH4、钙调蛋白、FAD、FMN 和 L-[U-14C]精氨酸的反应混合物中于 37°C 孵育 15 分钟。反应用冰冷的 HEPES/EDTA 缓冲液终止。然后将混合物通过阳离子交换柱以保留未反应的精氨酸。形成的 [14C]瓜氨酸用水洗脱,并通过液体闪烁计数法定量,以确定酶活性。[1]
NO 生成也可以通过测量其代谢物硝酸盐和亚硝酸盐来评估。将重组 nNOS 与包括 L-精氨酸在内的反应组分一起孵育,使用商业比色测试试剂盒定量培养基中积累的硝酸盐/亚硝酸盐。[1] nNOS 产生的 H2O2 使用硫氰酸亚铁法测定。[1] nNOS 的细胞色素 c 还原酶活性通过在缺乏钙和钙调蛋白的条件下,分光光度法监测 NADPH 依赖的细胞色素 c 还原来测量。[1] 本文献未描述针对 2-硫代尿嘧啶 的经典酶活性实验(如抑制常数、IC50)。实验侧重于 TU 与酶促生成的黑色素生物合成途径中醌类中间体的化学相互作用。使用蘑菇酪氨酸酶(3900 units/mg)在 pH 7.0 条件下催化 DOPA、DHI 和 DHICA 在有或无 TU 存在下的氧化反应。[2] 为研究2-硫代尿嘧啶与黑色素生物合成酶的相互作用,使用酪氨酸酶催化的氧化实验。方案包括将2-硫代尿嘧啶与多巴或5,6-二羟基吲哚在酪氨酸酶存在下共孵育。反应混合物在适当的缓冲条件(通常pH 6.8-7.4)和25-37°C下维持。可通过紫外-可见分光光度法监测反应进程,测量350 nm处吸光度的变化(硫代尿嘧啶掺入的特征)。将生成的黑色素产物进行化学降解,产生吡咯标志物(PDCA和PTCA),然后通过HPLC定量,以评估2-硫代尿嘧啶的掺入程度。对于甲状腺过氧化物酶抑制研究,可通过在存在不同浓度2-硫代尿嘧啶的条件下,测量甲状腺球蛋白的碘化或愈创木酚氧化来评估酶活性。 |
| 细胞实验 |
细胞毒性评估:将癌细胞系(如HepG2肝细胞癌细胞)以适当密度(通常5 × 10³个/孔)接种于96孔板中,培养过夜。然后用不同浓度的2-硫代尿嘧啶或其配合物(如1-100 μM)处理细胞24-72小时。使用MTT法或CCK-8法评估细胞活力。凋亡检测可使用Annexin V/PI染色后流式细胞术分析。其他终点包括线粒体膜电位测定以及通过Western blot评估PARP裂解。对于黑色素细胞蓄积研究,将黑色素性和非黑色素性黑色素细胞与浓度范围为40 ng至40 μg/10⁶细胞的2-硫代尿嘧啶共孵育,然后使用GC-MS分析细胞内药物浓度。
|
| 动物实验 |
- 动物: 使用无特定病原体的雌性 C57Bl/6 小鼠(6 周龄)。[2]
- 肿瘤模型: 小鼠皮下接种 B16 黑色素瘤细胞。接种后两周,待肿瘤形成后,小鼠静脉注射 10 μCi 的 [2-¹⁴C] 标记的 2-硫代尿嘧啶。[2] - 样本采集: 在指定时间点(30、90 和 180 分钟),通过 CO₂ 吸入法处死动物。剥离肿瘤周围组织,称重并迅速冷冻。[2] - 黑色素提取与分析: 肿瘤在 1% 乙酸-乙醇(30:70,v/v)中匀浆并离心。通过蔗糖密度梯度超速离心纯化黑素体。将干燥的黑色素等分试样溶解于 Soluene 350 中,并在液体闪烁计数器中进行计数。黑色素含量的分光光度测定采用 Ito 等人(1993)的方法。[2] 研究2-硫代尿嘧啶抗肿瘤活性的代表性体内方案采用小鼠异种移植模型。雌性C.B-17 SCID小鼠皮下注射HepG2细胞(约5 × 10⁶个细胞于0.1 mL PBS中)建立肿瘤。当肿瘤体积达到约100-150 mm³时,将动物随机分为治疗组(每组n=5-8只)。2-硫代尿嘧啶衍生物(如Ru(II)配合物)以2或4 mg/kg的剂量腹腔注射给药,每日一次,连续21天。阿霉素(2 mg/kg)作为阳性对照,单独溶剂作为阴性对照。每3-4天使用卡尺测量肿瘤尺寸,肿瘤体积计算公式为:体积 =(长×宽²)/2。研究结束时,切除肿瘤、称重并进行组织病理学检查。整个研究过程中监测体重作为全身毒性指标。对于黑色素瘤靶向研究,将放射性标记的2-硫代尿嘧啶注射到荷黑色素瘤小鼠体内,通过评估350 nm处黑色素吸收和化学降解后PTCA产量分析掺入肿瘤色素的程度。 |
| 药代性质 (ADME/PK) |
吸收、分布和排泄
药物向胎儿的传递:在胎儿中出现的时间 10 分钟;达到胎儿/母体浓度平衡的时间 40 分钟;胎儿/母体浓度比 1.0 /摘自表格/ /在大鼠中/ (35)S 标记的积累速度更快,且从较小剂量开始积累的程度更大,表明存在可饱和的转运机制。使用 (14)C-硫尿嘧啶进行的类似研究表明,母兔和胎儿的甲状腺组织中均存在放射性积累…… ……无论使用大剂量 (39 μmol) 还是小剂量 (1.2 μmol) 的 (35)S-硫尿嘧啶,与血浆相比,甲状腺组织中总放射性和未代谢的 (35)S-硫尿嘧啶的积累量均显著高于大鼠血浆中的水平。 硫尿嘧啶可经大鼠胃肠道吸收……大鼠静脉注射 5 mg 硫尿嘧啶后,3 小时后从尸体中回收的硫尿嘧啶仅占 30%,其余仅占痕量。 24 小时后。 有关 2-硫尿嘧啶(共 7 种代谢物)的更多吸收、分布和排泄(完整)数据,请访问 HSDB 记录页面。 代谢/代谢物 在雌性 Holtzman 大鼠的均质化肝脏制备物中,28-35% 的硫尿嘧啶在 3 小时内被代谢。硫尿嘧啶的分解途径推测如下:尿嘧啶;β-脲基丙酸,后者进一步代谢为 β-丙氨酸;氨和二氧化碳…… 采用高效液相色谱法 (HPLC) 评估了硫尿嘧啶在测定条件下的稳定性。将 TU (500 μM) 与高浓度 BH4 (最高达 100 μM) 孵育,并未导致明显的分解,证实了其在酶活性测定过程中的稳定性。[1] 2-硫代尿嘧啶的药代动力学数据显示,这类抗甲状腺药物通常半衰期较短,主要经尿液排泄。其吸收、分布、代谢和排泄性质因个体因素和具体衍生物而异。亲脂性更强的类似物6-丙基-2-硫代尿嘧啶(丙基硫氧嘧啶)比母体化合物表现出更高的细胞穿透能力。在暴露于高浓度(40 μg/10⁶细胞)丙基硫氧嘧啶的黑色素性黑色素细胞中,观察到显著的药物蓄积,这与其相比2-硫代尿嘧啶更强的毒性相一致。该化合物可使用灵敏的分析方法如LC-MS/MS在生物体液中检测,最近的方法已在牛尿中实现10 μg/L的检出限。 |
| 毒性/毒理 (Toxicokinetics/TK) |
2-硫代尿嘧啶被归类为危险物质,具有潜在致癌性。根据国际癌症研究机构的评估,2-硫代尿嘧啶可致癌。根据OSHA危险通报标准,它属于致癌性类别2,即怀疑会致癌。已知的不良反应包括血液学异常,且该化合物被怀疑具有致畸性和致癌性。在动物研究中,给予2-硫代尿嘧啶可诱导动脉硬化。然而,当以Ru(II)配合物形式存在时,全身毒性似乎可耐受,治疗小鼠未见显著体重变化或主要器官损伤。安全注意事项包括使用前获取特别指导、穿戴适当的个人防护装备、存放在上锁区域、并通过经批准的废物处理方案进行处置。该化合物严格限于科研使用,未经适当授权不得用于人类治疗应用。
|
| 参考文献 |
|
| 其他信息 |
根据世界卫生组织国际癌症研究机构 (IARC) 的说法,硫尿嘧啶可能致癌。
硫尿嘧啶是一种核碱基类似物,是尿嘧啶的一种衍生物,其中 C-2 位的氧代基团被硫代基团取代。它既是一种抗甲状腺药物,也是一种代谢产物。它是一种硫代羰基化合物,也是一种核碱基类似物。它在功能上与尿嘧啶相关。 已有报道称在眼虫 (Euglena gracilis) 中发现了 2-硫尿嘧啶,并有相关数据。 硫尿嘧啶是一种含硫尿嘧啶。作为一种已知的抗甲状腺药物和高选择性的一氧化氮合酶 (NOS) 抑制剂,硫尿嘧啶还能与酪氨酸酶催化酪氨酸氧化产生的多巴醌共价结合,从而选择性地在过度活跃的黑色素生成细胞中新合成的黑色素中积累,并为定位黑色素瘤细胞提供了一种方法。 (NCI04) 存在于芸薹属和十字花科植物的种子中。硫脲嘧啶曾被用作抗甲状腺药物、冠状动脉扩张剂和治疗充血性心力衰竭的药物,但其用途已被其他药物广泛取代。已知它会导致血液疾病,并被怀疑具有致畸性和致癌性。 作用机制 抗甲状腺药物主要通过干扰碘的有机化来抑制甲状腺激素的形成。这意味着它们会干扰碘离子的氧化,但由于对甲状腺碘氧化系统的了解尚不完全,因此难以阐明其详细机制。 /抗甲状腺药物/ 硫酰胺衍生物不会对甲状腺产生永久性影响,但会抑制激素的合成和分泌,直至疾病过程中出现自发缓解。/硫酰胺衍生物/ 治疗用途 抗代谢药;抗甲状腺药;血管扩张剂 据报道,硫脲嘧啶在人类医学中曾用作抗甲状腺药物,并用于治疗心绞痛和充血性心力衰竭……然而,没有证据表明硫脲嘧啶目前在美国用于这些用途。 治疗甲状腺功能亢进;心绞痛;充血性心力衰竭 /中国:美国以前的用途/ 药物(兽用):甲状腺抑制剂;用于治疗甲状腺功能亢进症和促进增重 药物警告 抗甲状腺药物治疗的主要缺点是停药后复发率高。/抗甲状腺药物/ 患者应立即报告咽喉痛或发热,这通常是粒细胞缺乏症的先兆。如果发现轻度粒细胞减少,可能是甲状腺毒症的征兆,也可能是这种危险药物反应的首发症状。此时需要谨慎并频繁进行白细胞计数。硫胺素衍生物:服用这些药物的妇女不应母乳喂养婴儿。抗甲状腺药物:在妊娠的哪个阶段开始治疗才会对胎儿产生影响(胎儿甲状腺肿和甲状腺功能减退)尚不明确,但在某些情况下,用药直到妊娠三分之二才开始。甚至更晚的母体用药也可能导致这种情况。 /抗甲状腺药物/ 有关 2-硫尿嘧啶(共 6 条)的更多药物警告(完整)数据,请访问 HSDB 记录页面。 2-硫尿嘧啶 是一种已上市的抗甲状腺药物,也是一种正在研究用于检测和靶向转移性黑色素瘤的药物。其抗甲状腺和靶向黑色素瘤的特性与其对甲状腺碘过氧化物酶、髓过氧化物酶、嗜酸性粒细胞过氧化物酶和酪氨酸酶等酶的抑制作用有关。[1] 鉴于甲状腺中存在 NOS 活性,新发现的 TU 作为选择性 nNOS 抑制剂的特性可能有助于理解其抗甲状腺作用。它也代表了一种潜在的先导化合物,可用于开发神经保护剂。[1] 该抑制作用与 L-精氨酸竞争。增加辅因子四氢生物蝶呤 (BH4) 的浓度可以逆转这种抑制作用,这表明该化合物与 BH4 结合位点存在相互作用或邻近关系。硫脲部分中的硫原子对于活性至关重要,因为尿嘧啶(缺乏硫)和硫脲均无效。[1] - 选择性掺入机制: 2-硫代尿嘧啶 通过在活跃的黑色素生成过程中与反应性醌类中间体发生化学反应而蓄积在黑色素瘤中。与半胱氨酸等巯基化合物主要在多巴醌的 5-位反应不同,TU 几乎只在 6-位加成,形成 6-S-TU-多巴。TU 还能与来自 DHI 的下游吲哚醌(在 C-2 和 C-3 位)以及来自 DHICA 的下游吲哚醌(在 C-4 位)反应。由此产生的 TU-吲哚加合物随后可通过氧化共聚作用掺入到生长的黑色素聚合物中。[2] - 对黑色素结构的影响: TU 的掺入显著改变了所得黑色素的性质,导致 A₃₅₀ 吸光度降低,降解产物(PTCA 和 PDCA)产率改变,与正常真黑色素相比,其溶解度和颜色也发生变化。[2] - 临床潜力: TU 及相关硫脲嘧啶化合物因其在黑色素瘤组织中的选择性蓄积能力,已被研究作为用于诊断成像(如使用放射性碘)和潜在硼中子俘获治疗(BNCT)的黑色素瘤定位剂。[2] - 分离鉴定的关键化合物: 该研究分离并鉴定了几种新型 TU-吲哚加合物,包括化合物 1(C-3 位取代的 DHI-TU)、化合物 2(C-2 位取代的 DHI-TU)、化合物 3(2 的 4,4'-双吲哚基二聚体)、化合物 4(含两个 TU 单元的 DHI 三聚体)、化合物 5(C-4 位取代的 DHICA-TU)、化合物 6(5 的 3,4'-双吲哚基二聚体)和化合物 7(含一个 TU 单元的 DHICA 三聚体)。提供了这些化合物的完整 NMR 和 MS 表征数据。[2] |
| 分子式 |
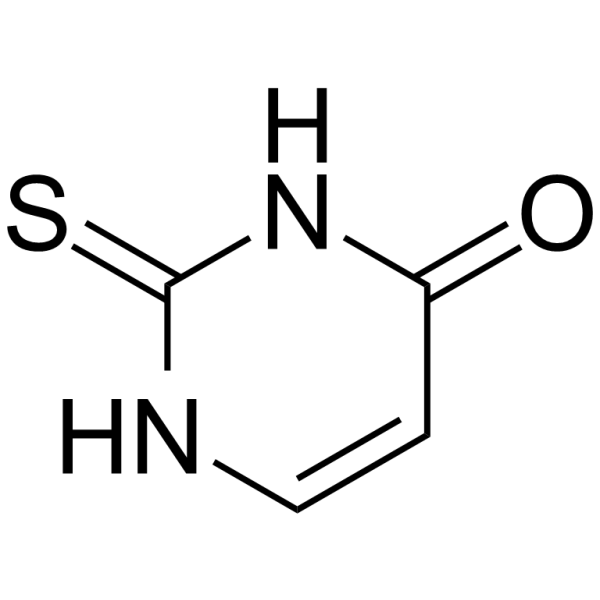
C4H4N2OS
|
|---|---|
| 分子量 |
128.15236
|
| 精确质量 |
128.004
|
| CAS号 |
141-90-2
|
| 相关CAS号 |
2-Thiouracil-13C,15N2
|
| PubChem CID |
1269845
|
| 外观&性状 |
White to off-white solid powder
|
| 密度 |
1.5±0.1 g/cm3
|
| 沸点 |
337.2ºC at 760mmHg
|
| 熔点 |
>300 °C(lit.)
|
| 闪点 |
157.7ºC
|
| 蒸汽压 |
5.45E-05mmHg at 25°C
|
| 折射率 |
1.678
|
| LogP |
-0.28
|
| tPSA |
80.74
|
| 氢键供体(HBD)数目 |
2
|
| 氢键受体(HBA)数目 |
2
|
| 可旋转键数目(RBC) |
0
|
| 重原子数目 |
8
|
| 分子复杂度/Complexity |
163
|
| 定义原子立体中心数目 |
0
|
| SMILES |
C1=CNC(=S)NC1=O
|
| InChi Key |
ZEMGGZBWXRYJHK-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C4H4N2OS/c7-3-1-2-5-4(8)6-3/h1-2H,(H2,5,6,7,8)
|
| 化学名 |
2-sulfanylidene-1H-pyrimidin-4-one
|
| 别名 |
2-thiouracil; Thiouracil; 141-90-2; Deracil; Antagothyroid;
|
| HS Tariff Code |
2934.99.9001
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
DMSO : ~50 mg/mL (~390.17 mM)
H2O : ~0.67 mg/mL (~5.23 mM) |
|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 3.75 mg/mL (29.26 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 37.5 mg/mL 澄清的 DMSO 储备液加入到400 μL PEG300中,混匀;再向上述溶液中加入50 μL Tween-80,混匀;然后加入450 μL 生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: ≥ 3.75 mg/mL (29.26 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 例如,若需制备1 mL的工作液,可将 100 μL 37.5 mg/mL 的澄清 DMSO 储备液加入到 900 μL 玉米油中并混合均匀。 请根据您的实验动物和给药方式选择适当的溶解配方/方案: 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 7.8034 mL | 39.0168 mL | 78.0336 mL | |
| 5 mM | 1.5607 mL | 7.8034 mL | 15.6067 mL | |
| 10 mM | 0.7803 mL | 3.9017 mL | 7.8034 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。