| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 10 mM * 1 mL in DMSO |
|
||
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
| 靶点 |
P2X7 Receptor
P2X7 Receptor (purinergic receptor P2X subtype 7) - Ki value: ~1.6 nM (determined by [³H]A-804598 competitive binding assay in rat brain cortex membranes); - IC50 for inhibiting ATP-induced P2X7-mediated responses: ~8.3 nM (inhibition of ATP-induced ethidium bromide (EtBr) uptake, a marker of P2X7-mediated pore formation, in human embryonic kidney (HEK) 293 cells stably expressing human P2X7 (HEK-hP2X7)); - No significant binding to other P2 receptor subtypes (P2X1, P2X2, P2X3, P2X4, P2X5, P2Y1, P2Y2, P2Y4, P2Y6) at concentrations up to 10 μM, indicating high selectivity for P2X7[1] [1] |
|---|---|
| 体外研究 (In Vitro) |
以浓度依赖性方式,与 A-804598(0.1-10 μM;1 小时)预孵育可大大减少 BzATP 诱导的细胞损失。 3 μM A-804598 显示了针对 BzATP 诱导的细胞毒性的最高保护效果[2]。
为了补充P2X7敲除的结果,我们试图测试P2X7拮抗剂(a-804598)在小鼠小胶质细胞中的作用。在暴露于BzATP之前,将原代小胶质细胞与不同浓度的A-804598一起孵育1小时。通过CCK-8测定法测量细胞活力,并通过共聚焦显微镜检查小胶质细胞形态。如图6a所示,单独使用BzATP会导致约40%的小胶质细胞损失,而与A-804598预孵育会以浓度依赖的方式显著减轻BzATP诱导的细胞损失。三种微摩尔A-804598对BzATP诱导的细胞毒性表现出最大的保护作用(图6b)。此外,我们验证了A-804598对活化小胶质细胞的保护作用。LPS预处理的小胶质细胞首先暴露于不同浓度的A-804598,然后用BzATP处理。正如预期的那样,经预处理的小胶质细胞在BzATP的作用下表现出变形虫形态和显著的细胞损失(图6a)。然而,共聚焦显微镜和CCK-8测定的结果表明,BzATP引起的细胞损失可以通过与A-804598预孵育以浓度依赖的方式抵消(图6c)。综上所述,我们的研究结果揭示了P2X7拮抗剂A-804598对失活和活化小胶质细胞中BzATP诱导的细胞毒性的保护作用,进一步证明了P2X9在ATP诱导的小胶质细胞死亡中的介导作用。[2] 1. P2X7受体结合亲和力与选择性: - 在大鼠大脑皮层膜制备物中,[³H]A-804598以高亲和力结合P2X7受体:平衡解离常数(Kd)为~2.1 nM,最大结合容量(Bmax)为~11.2 fmol/mg蛋白。未标记的A-804598可竞争性置换[³H]A-804598,Ki值为~1.6 nM。 - 在针对15种其他受体家族(包括肾上腺素能、胆碱能、GABA能、阿片受体)和离子通道的结合实验中,10 μM A-804598对相应放射性配体的置换率<50%,证实无脱靶结合[1] 2. 抑制P2X7介导的功能反应: - 在HEK-hP2X7细胞中:A-804598呈剂量依赖性抑制ATP(1 mM)诱导的EtBr摄取(小孔形成),IC50为~8.3 nM;100 nM时抑制率超过95%。其还可抑制ATP诱导的细胞内钙([Ca²⁺]i)升高,IC50为~7.9 nM,与P2X7拮抗作用一致。 - 在大鼠原代小胶质细胞中:100 nM A-804598可完全阻断ATP(5 mM)诱导的EtBr摄取,证实其在天然细胞中对P2X7的抑制作用[1] 3. P2X7抑制作用的可逆性: - 将HEK-hP2X7细胞中10 nM A-804598洗脱后,ATP诱导的[Ca²⁺]i升高在30分钟内恢复至处理前水平的~90%,表明其与P2X7的结合具有可逆性[1] [1] |
| 体内研究 (In Vivo) |
在疾病末期的腰脊髓中,A-804598慢性治疗(腹腔注射;30 mg/kg;每周五次)可降低LC3B-II和SQSTM1/p62的表达[3]。
众所周知,在疾病进展过程中,当自噬通量受损时,SQSTM1/p62自噬底物与LC3B-II一起在ALS小鼠的腰椎脊髓中积累(Zhang等人,2011)。因此,我们测量了SOD1-G93A小鼠腰椎脊髓中LC3B-II和SQSTM1/p62蛋白的水平,该小鼠在体内通过用血脑渗透剂a-804598对雌性SOD1-G933A小鼠进行慢性治疗来药理学抑制P2X7受体,证明在啮齿动物中口服或腹腔注射剂量后达到脑浓度(Able等人,2011;Iwata等人,2016),从发病前到疾病末期以30mg/Kg的剂量给药。我们发现,虽然与野生型相比,经赋形剂处理的SOD1-G93A小鼠的LC3B-II(图5A)和SQSTM1/p62(图5B)的蛋白质水平在终末期都有所增加,但在A-804598处理的ALS小鼠中,与赋形剂相比,LC3B-II的蛋白质含量似乎没有变化,而SQSTM1/p62被抑制到基础水平(图5A、B)。如图所示,在雌性SOD1-G93A小鼠中施用A-804598时,行为评分(图5C)、疾病发作(图5D)和存活率(图5E)均不受影响。[3] |
| 酶活实验 |
ATP敏感的P2X7受体定位在免疫来源的细胞上,包括中枢神经系统中的外周巨噬细胞和神经胶质细胞。P2X7受体的激活导致细胞内钙浓度的快速变化,促炎细胞因子IL-1β的释放,以及在长时间接触激动剂后,质膜中细胞溶解孔的形成。基因敲除研究和最近描述的选择性拮抗剂的数据表明,P2X7受体激活在炎症和疼痛中起作用。虽然存在几种物种选择性P2X7拮抗剂,但A-804598代表了一种结构新颖、具有竞争力和选择性的拮抗剂,对大鼠(IC50=10 nM)、小鼠(IC50=9 nM)和人类(IC50=11 nM)P2X7受体具有同等的高亲和力。A-804598还有效地阻断了激动剂刺激的IL-1β和Yo-Pro从天然表达人P2X7受体的分化THP-1细胞中的摄取释放。A-804598被氚化([3H]A-804598;8.1Ci/mmol),用于研究1321N1细胞中表达的重组大鼠P2X7受体。[3H]A-804598标记了一类高亲和力结合位点(Kd=2.4 nM,表观Bmax=0.56 pmol/mg)。在未转染的1321N1细胞中没有观察到特异性结合。P2X拮抗剂抑制[3H]A-804598结合的药理学特征与其阻断P2X7受体功能激活的能力相关(r=0.95,P<0.05)。这些数据表明,A-804598是迄今为止描述的哺乳动物P2X7受体最有效和最具选择性的拮抗剂之一,[3H]A-804598则是一种高亲和力的拮抗剂放射性配体,可特异性标记大鼠P2X7接收器[1]。
1. [³H]A-804598竞争性结合实验(大鼠大脑皮层膜): - 膜制备:解剖大鼠大脑,将皮层在冰浴缓冲液(50 mM Tris-HCl,pH 7.4,1 mM EDTA)中匀浆,4℃下100,000×g离心20分钟。沉淀用相同缓冲液重悬,-80℃保存备用。 - 孵育体系:200 μL反应混合物含膜蛋白(50 μg)、[³H]A-804598(0.5-10 nM)、未标记A-804598(0.1 nM-10 μM,用于竞争曲线)或缓冲液(用于总结合)。非特异性结合通过加入10 μM未标记P2X7拮抗剂(oxATP)测定。混合物在25℃孵育60分钟。 - 分离与检测:使用细胞收集器将结合与游离放射性配体通过预浸泡在0.5%聚乙烯亚胺中的玻璃纤维滤膜快速过滤分离。滤膜用冰浴缓冲液洗涤3次,干燥后与闪烁液混合,通过液体闪烁计数器计数放射性。Kd、Bmax和Ki值通过非线性回归计算[1] 2. ATP诱导的EtBr摄取实验(HEK-hP2X7细胞): - 细胞以5×10⁴个/孔接种于96孔板,过夜培养。培养基替换为含EtBr(5 μM)和A-804598(0.1 nM-1 μM)的汉克平衡盐溶液(HBSS)。预孵育10分钟后,加入ATP(1 mM)诱导小孔形成。 - 使用酶标仪在激发光540 nm、发射光620 nm条件下,每2分钟测量一次荧光强度,持续30分钟。计算EtBr摄取速率(荧光升高斜率),通过浓度-抑制曲线推导IC50[1] 3. ATP诱导的[Ca²⁺]i升高实验(HEK-hP2X7细胞): - 细胞在含0.02%普朗尼克酸的HBSS中,用钙敏感染料Fluo-4 AM(4 μM)37℃负载30分钟。洗涤后,细胞与A-804598(0.1 nM-1 μM)预孵育10分钟。 - 加入ATP(1 mM),在激发光488 nm、发射光525 nm条件下测量荧光强度20分钟。以峰值荧光强度量化[Ca²⁺]i升高,通过非线性回归计算IC50[1] [1] |
| 细胞实验 |
细胞毒性测定[2]
细胞类型: 小胶质细胞 测试浓度: 0.1, 0.3, 1, 3, 10 μM 孵育时间:1 小时 实验结果:在失活和活化的小胶质细胞中均能免受 BzATP 诱导的细胞毒性。 1. HEK-hP2X7细胞培养及P2X7功能验证: - 人胚胎肾细胞HEK 293稳定转染人P2X7 cDNA,在完全培养基(DMEM + 10%胎牛血清 + 选择性抗生素)中培养。当融合度达80%-90%时,每2-3天传代一次。 - 功能实验(EtBr摄取、[Ca²⁺]i升高)前,细胞以5×10⁴个/孔接种于96孔板,过夜培养以确保贴壁。实验前将培养基替换为HBSS,排除血清干扰[1] 2. 大鼠原代小胶质细胞分离及EtBr摄取实验: - 对1-3日龄大鼠幼崽实施安乐死,解剖大脑。将皮层组织剪碎,用胰蛋白酶(0.25%)37℃消化15分钟,吹打成单细胞悬液。 - 细胞接种于T75培养瓶,在DMEM + 10%胎牛血清中培养7-10天。通过37℃下200 rpm振荡培养瓶2小时分离小胶质细胞,离心收集后以1×10⁵个/孔接种于96孔板。 - 24小时后,小胶质细胞与100 nM A-804598预孵育10分钟,随后加入ATP(5 mM)+ EtBr(5 μM),通过测量荧光强度评估EtBr摄取抑制情况[1] [1] |
| 动物实验 |
动物/疾病模型:成年 B6 .Cg-Tg (SOD1-G93A) 1Gur/J 雌性小鼠 [3]
剂量: 30 mg/kg 给药途径:腹腔注射 (ip);每周五次 实验结果: SQSTM1/p62 表达降低。 100 日龄(发病前)的 SOD1-G93A 小鼠被随机分为载体处理组和中枢神经系统穿透性 P2X7 特异性拮抗剂 A-804598 处理组(Donnelly-Roberts 等,2009;Catanzaro 等,2014;Iwata 等,2016),A-804598 通过腹腔注射给药,剂量为 30 mg/kg,每周五次,直至疾病终末期。由于对药物治疗的反应存在性别差异(Pizzasegola 等,2009),并且 P2X7 拮抗剂 Brilliant Blue G 仅延长了雌性 SOD1-G93A 小鼠的生存期(Bartlett 等,2017;Sluyter 等,2017),因此我们选择研究雌性小鼠。[3] |
| 参考文献 |
|
| 其他信息 |
ATP敏感性P2X7受体定位于免疫来源的细胞,包括外周巨噬细胞和中枢神经系统(CNS)的神经胶质细胞。P2X7受体的激活会导致细胞内钙离子浓度快速变化,释放促炎细胞因子IL-1β,并且在激动剂作用持续一段时间后,会在细胞膜上形成细胞溶解孔。基因敲除研究和近期报道的选择性拮抗剂的数据表明,P2X7受体的激活在炎症和疼痛中发挥作用。虽然目前已存在多种物种选择性P2X7受体拮抗剂,但A-804598是一种结构新颖的竞争性选择性拮抗剂,对大鼠(IC50 = 10 nM)、小鼠(IC50 = 9 nM)和人(IC50 = 11 nM)的P2X7受体均具有同等的高亲和力。 A-804598 还能有效阻断激动剂刺激的 IL-1β 释放以及分化的 THP-1 细胞对 Yo-Pro 的摄取,这些细胞天然表达人 P2X7 受体。A-804598 经氚标记([3H]A-804598;8.1 Ci/mmol)后,用于研究在 1321N1 细胞中表达的重组大鼠 P2X7 受体。[3H]A-804598 标记了一类高亲和力结合位点(Kd=2.4 nM,表观 Bmax=0.56 pmol/mg)。在未转染的 1321N1 细胞中未观察到特异性结合。P2X 受体拮抗剂抑制 [3H]A-804598 结合的药理学特征与其阻断 P2X7 受体功能激活的能力呈正相关(r=0.95,P<0.05)。这些数据表明,A-804598 是迄今为止报道的哺乳动物 P2X7 受体最有效且选择性最高的拮抗剂之一,[3H]A-804598 是一种高亲和力拮抗剂放射性配体,可特异性标记大鼠 P2X7 受体。[1]
背景:ATP 门控 P2X7 是一种非选择性阳离子通道,参与多种细胞功能以及病理生理过程,包括神经性疼痛、免疫反应和神经炎症。尽管 P2X7 在小胶质细胞中大量表达,但其在神经炎症中的作用仍不清楚。 方法:从 P0-2 日龄 C57BL/6 野生型或 P2X7 基因敲除 (P2X7-/-) 小鼠幼崽的皮层中分离出原代小胶质细胞。我们使用脂多糖、脂多糖联合IFNγ或IL-4联合IL-13诱导小胶质细胞极化为促炎或抗炎状态。通过RNA测序和定量实时PCR检测静息或激活状态下小鼠和人小胶质细胞中P2rx7的表达水平。采用细胞计数试剂盒-8(CCK-8)和免疫细胞化学方法检测小胶质细胞死亡,并使用P2X7激动剂BzATP或P2X7拮抗剂A-804598,通过Luminex多重分析或ELISA检测野生型或P2X7-/-小胶质细胞的分泌情况。采用Western blot分析P2X7信号通路。 结果:首先,我们证实P2rx7在小鼠和人原代小胶质细胞中组成型表达。此外,在促炎和抗炎条件下,小鼠小胶质细胞中P2rx7 mRNA水平均下调。第二,P2X7激动剂BzATP可导致小鼠小胶质细胞死亡,而P2X7基因敲除或A-804598在基础状态和促炎状态下均能抑制这种效应,提示P2X7在BzATP诱导的小胶质细胞死亡中起介导作用。第三,BzATP诱导的IL-1家族细胞因子(包括IL-1α、IL-1β和IL-18)的释放,在P2X7-/-小胶质细胞中被阻断,在促炎状态下的小胶质细胞中也被A-804598阻断,而其他细胞因子/趋化因子的释放则与P2X7激活无关。这些发现支持P2X7在IL-1家族细胞因子释放中的特异性作用。最后,研究发现 P2X7 的激活与 AKT 和 ERK 通路相关,这可能是 P2X7 在小胶质细胞中发挥作用的潜在机制。 结论:这些结果表明,P2X7 介导 BzATP 诱导的小胶质细胞死亡和 IL-1 家族细胞因子的特异性释放,表明 P2X7 在神经炎症中发挥重要作用,并暗示靶向 P2X7 可能用于治疗神经炎症性疾病。[2] 自噬和炎症在肌萎缩侧索硬化症 (ALS) 的发病机制中起着决定性作用。ALS 是一种成人起病的神经退行性疾病,其特征是上运动神经元和下运动神经元 (MN) 的退化和最终丧失,这会使小胶质细胞处于激活状态,从而维持神经炎症并形成神经退行性变的恶性循环。鉴于胞外ATP通过P2X7受体构成神经元向小胶质细胞传递的警报信号,且该信号与肌萎缩侧索硬化症(ALS)相关,并且P2X7受体影响免疫细胞的自噬,我们研究了超氧化物歧化酶1(SOD1)-G93A小鼠原代小胶质细胞中P2X7受体的激活是否能直接诱导自噬。我们发现,P2X7受体通过mTOR通路增强自噬标志物微管相关蛋白1轻链3(LC3)-II的表达,同时调节抗炎性M2型小胶质细胞标志物的表达。我们还发现,在SOD1-G93A小鼠小胶质细胞中,短暂刺激P2X7受体后,自噬靶标SQSTM1/p62的表达降低,但在持续刺激后则升高。 P2X7拮抗剂A-804598和自噬/磷脂酰肌醇-3-激酶抑制剂wortmannin (WM)可阻止这些效应。此外,在SOD1-G93A小鼠中,长期体内使用A-804598治疗可降低疾病终末期腰椎脊髓中SQSTM1/p62的表达。这些数据表明,自噬通量的调节是P2X7激活ALS小胶质细胞的一种新机制,值得在ALS的进一步研究中加以考虑。[3] 1. A-804598是首个报道的高亲和力、选择性P2X7受体拮抗剂,可放射性标记为[³H]A-804598,这是一种用于定量分析组织(例如脑、脊髓、免疫器官)中P2X7受体分布和密度的工具。[1] 2. A-804598 对 P2X7 具有高度选择性(在 10 μM 浓度下对其他 P2 受体或脱靶效应无活性),因此适用于研究 P2X7 特异性功能,例如小胶质细胞活化、细胞因子释放和孔道形成,而不会干扰其他嘌呤能信号通路[1] 3. [³H]A-804598 结合试验可用于筛选新型 P2X7 配体:能够将 [³H]A-804598 从 P2X7 受体上置换下来的化合物可被鉴定为潜在的 P2X7 激动剂或拮抗剂,其 Ki 值反映了结合亲和力[1] [1] |
| 分子式 |
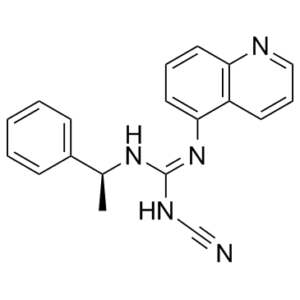
C19H17N5
|
|
|---|---|---|
| 分子量 |
315.38
|
|
| 精确质量 |
315.148
|
|
| 元素分析 |
C, 72.36; H, 5.43; N, 22.21
|
|
| CAS号 |
1125758-85-1
|
|
| 相关CAS号 |
|
|
| PubChem CID |
53325874
|
|
| 外观&性状 |
White to off-white solid powder
|
|
| LogP |
4.298
|
|
| tPSA |
73.1
|
|
| 氢键供体(HBD)数目 |
2
|
|
| 氢键受体(HBA)数目 |
3
|
|
| 可旋转键数目(RBC) |
5
|
|
| 重原子数目 |
24
|
|
| 分子复杂度/Complexity |
473
|
|
| 定义原子立体中心数目 |
1
|
|
| SMILES |
N([H])(/C(/N([H])C#N)=N/[C@@]([H])(C([H])([H])[H])C1C([H])=C([H])C([H])=C([H])C=1[H])C1=C([H])C([H])=C([H])C2=C1C([H])=C([H])C([H])=N2
|
|
| InChi Key |
PQYCRDPLPKGSME-AWEZNQCLSA-N
|
|
| InChi Code |
InChI=1S/C19H17N5/c1-14(15-7-3-2-4-8-15)23-19(22-13-20)24-18-11-5-10-17-16(18)9-6-12-21-17/h2-12,14H,1H3,(H2,22,23,24)/t14-/m0/s1
|
|
| 化学名 |
|
|
| 别名 |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| 存储方式 |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| 运输条件 |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| 溶解度 (体外实验) |
|
|||
|---|---|---|---|---|
| 溶解度 (体内实验) |
配方 1 中的溶解度: ≥ 2.5 mg/mL (7.93 mM) (饱和度未知) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。
例如,若需制备1 mL的工作液,可将100 μL 25.0 mg/mL澄清DMSO储备液加入到400 μL PEG300中,混匀;然后向上述溶液中加入50 μL Tween-80,混匀;加入450 μL生理盐水定容至1 mL。 *生理盐水的制备:将 0.9 g 氯化钠溶解在 100 mL ddH₂O中,得到澄清溶液。 配方 2 中的溶解度: 2.5 mg/mL (7.93 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液; 超声助溶. 例如,若需制备1 mL的工作液,可将 100 μL 25.0 mg/mL澄清DMSO储备液加入900 μL 20% SBE-β-CD生理盐水溶液中,混匀。 *20% SBE-β-CD 生理盐水溶液的制备(4°C,1 周):将 2 g SBE-β-CD 溶解于 10 mL 生理盐水中,得到澄清溶液。 View More
配方 3 中的溶解度: ≥ 2.5 mg/mL (7.93 mM) (饱和度未知) in 10% DMSO + 90% Corn Oil (这些助溶剂从左到右依次添加,逐一添加), 澄清溶液。 1、请先配制澄清的储备液(如:用DMSO配置50 或 100 mg/mL母液(储备液)); 2、取适量母液,按从左到右的顺序依次添加助溶剂,澄清后再加入下一助溶剂。以 下列配方为例说明 (注意此配方只用于说明,并不一定代表此产品 的实际溶解配方): 10% DMSO → 40% PEG300 → 5% Tween-80 → 45% ddH2O (或 saline); 假设最终工作液的体积为 1 mL, 浓度为5 mg/mL: 取 100 μL 50 mg/mL 的澄清 DMSO 储备液加到 400 μL PEG300 中,混合均匀/澄清;向上述体系中加入50 μL Tween-80,混合均匀/澄清;然后继续加入450 μL ddH2O (或 saline)定容至 1 mL; 3、溶剂前显示的百分比是指该溶剂在最终溶液/工作液中的体积所占比例; 4、 如产品在配制过程中出现沉淀/析出,可通过加热(≤50℃)或超声的方式助溶; 5、为保证最佳实验结果,工作液请现配现用! 6、如不确定怎么将母液配置成体内动物实验的工作液,请查看说明书或联系我们; 7、 以上所有助溶剂都可在 Invivochem.cn网站购买。 |
| 制备储备液 | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1708 mL | 15.8539 mL | 31.7078 mL | |
| 5 mM | 0.6342 mL | 3.1708 mL | 6.3416 mL | |
| 10 mM | 0.3171 mL | 1.5854 mL | 3.1708 mL |
1、根据实验需要选择合适的溶剂配制储备液 (母液):对于大多数产品,InvivoChem推荐用DMSO配置母液 (比如:5、10、20mM或者10、20、50 mg/mL浓度),个别水溶性高的产品可直接溶于水。产品在DMSO 、水或其他溶剂中的具体溶解度详见上”溶解度 (体外)”部分;
2、如果您找不到您想要的溶解度信息,或者很难将产品溶解在溶液中,请联系我们;
3、建议使用下列计算器进行相关计算(摩尔浓度计算器、稀释计算器、分子量计算器、重组计算器等);
4、母液配好之后,将其分装到常规用量,并储存在-20°C或-80°C,尽量减少反复冻融循环。
计算结果:
工作液浓度: mg/mL;
DMSO母液配制方法: mg 药物溶于 μL DMSO溶液(母液浓度 mg/mL)。如该浓度超过该批次药物DMSO溶解度,请首先与我们联系。
体内配方配制方法:取 μL DMSO母液,加入 μL PEG300,混匀澄清后加入μL Tween 80,混匀澄清后加入 μL ddH2O,混匀澄清。
(1) 请确保溶液澄清之后,再加入下一种溶剂 (助溶剂) 。可利用涡旋、超声或水浴加热等方法助溶;
(2) 一定要按顺序加入溶剂 (助溶剂) 。
P2X7 activation modulates microtubule-associated protein 1 light chain 3 (LC3)-II in superoxide dismutase 1 (SOD1)-G93A microglia.Front Cell Neurosci.2017 Aug 21;11:249. |
|---|
P2X7 induces autophagic flux in SOD1-G93A microglia.Front Cell Neurosci.2017 Aug 21;11:249. |
In vivochronic treatment with P2X7 antagonist A-804598 decreases SQSTM1/p62 expression in lumbar spinal cord of SOD1-G93A mice.Front Cell Neurosci.2017 Aug 21;11:249. |